The great American baseball legend, Yogi Berra, once said: “In theory, there is no difference between theory and practice. But in practice, there is.”
Silly as it reads, there is a great deal of truth to that statement.
In science, we very quickly chase after a particular theory as soon as a little bit of evidence is produced that supports it. Gradually, these theories become our basic understanding of a situation, until someone points out the holes in the theory and we have to revise it.
A new theory of Parkinson’s disease has recently been proposed. In today’s post we will review what the theory is suggesting and what evidence there is to support it.

“I still say it’s only a theory”. Source: NewYorker
In the age of ‘alternative facts’, it is always important to remember that we don’t know as much as we think we do. In fact, much of our modern world still relies on a kind of faith rather than actual ‘facts’. For example, we take a particular type of medicine, because it has worked for some people in the past, not because it will definitely make us better.
And the same applies to our understanding of neurodegenerative conditions, like Parkinson’s disease. Based on all the evidence we have collected thus far, we have theories of how Parkinson’s disease may be progressing. But there are always exceptions to the rule, and these force us to refine or reconsider our theories.
Recently a refinement to our theory of Parkinson’s disease has been suggested.
Who has suggested it?
This is Prof Ole Isacson.

Source: Crunchbase
He’s a dude.
He is is a Professor of Neurology at Harvard Medical School, and Chief Scientific Officer of the Neuroscience Research Unit and Senior Vice President at the pharmaceutical company Pfizer.
And this is Dr Simone Engelender.

Source: Rappaport
She’s awesome as well.
She is Associate Professor of Molecular Pharmacology at the Rappaport Family Institute for Research in the Medical Sciences in Haifa, Israel.
Together they have proposed a new theory of Parkinson’s disease that has the research community talking:

Title: The Threshold Theory for Parkinson’s Disease.
Authors: Engelender S, Isacson O.
Journal: Trends Neurosci. 2017 Jan;40(1):4-14.
PMID: 27894611
The new theory proposes that Parkinson’s disease may actually be a ‘systemic condition’ (that is, affecting cells everywhere at the same time), but the clinical features – such as motor issues – only appear as certain thresholds are passed in the affected populations of neurons in the brain.
What does that mean?
Wait a minute. Let’s start at the beginning.
Before discussing what the new theory suggests, shall we first have a look at what the old theories proposed?
Ok, what did the old theory propose?
This is Prof Heiko Braak:

Source – Memim.com
He’s pretty cool too. Nice guy.
Many years ago, Prof Braak – a German neuroanatomist – sat down and examined hundreds of postmortem brains from people with Parkinson’s disease.
He had collected brains from people at different stages of Parkinson’s disease – from just after being diagnosed to having had the condition for decades – and he was looking for any kind of pattern that might explain where and how the disease starts. His research led to what is referred to as the “Braak stages of Parkinson’s disease” – a six step explanation of how the disease spreads up from the brain stem and into the rest of the brain (Click here to read more about this).
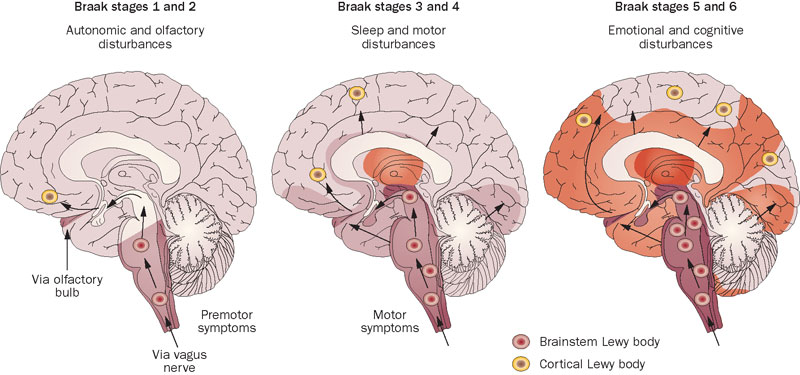
The Braak stages of PD. Source: Nature
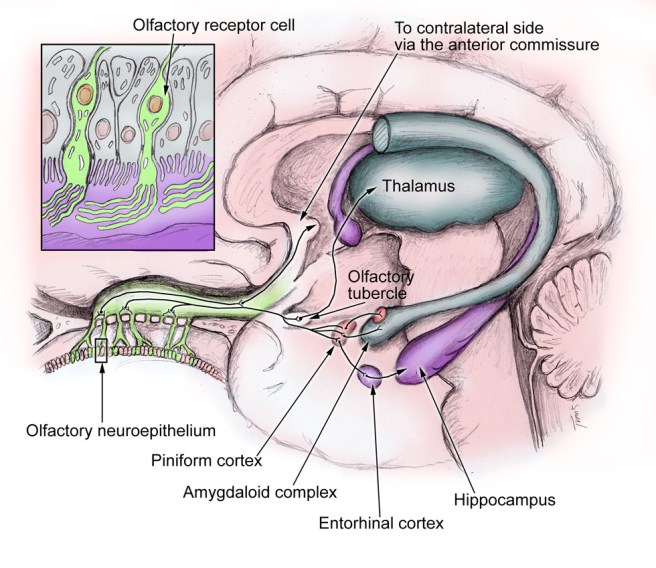
Braak’s results also led him to propose that Parkinson’s disease may actually begin in the brain stem (which connects the brain to the spinal cord) and the disease slowly works it’s way up into the brain.
That is the ‘ascending’ theory of Parkinson’s disease.
This idea has been further adapted by Braak and others with the discovery of Parkinson’s disease features in the gut (we have discussed this in previous posts – Click here and here to read those posts).
But how does the disease actually spread?
Good question.
The spread of the condition is believed to be due to the protein alpha synclein being passed between cells in some manner. This idea stemmed from the analysis of the brains of people with Parkinson’s disease who received cell transplantation therapy in the 1980-90’s. After those people passed away (due to natural causes), their brains were analysed and it was discovered that some of the cells in the transplants (1-5%) have Lewy bodies in them (Lewy bodies are one of the hallmarks of Parkinson’s disease, dense circular clusters of proteins including alpha synuclein). This suggests that the disease is passed on to the healthy transplanted cells in some way.

Photos of neurons from the post-mortem brains of people with Parkinson’s that received transplants. White arrows in the images above indicate lewy bodies inside transplanted cells. Source: The Lancet
So the research community has been working with the idea of an ‘ascending’ theory of Parkinson’s disease, and the spreading of the condition via the passing of alpha synuclein from cell to cell. And this theory has been fine,…
Why do I feel like there’s a ‘but’ coming?
Because there is a ‘but’ coming.
And it’s a big BUT.
But as Prof Isacson and Dr Engelender point out there are some holes in this theory.
Some big holes.
For example, in a 2008 study of 71 postmortem brains from people with Parkinson’s disease, 47% of the cases did not fit the predicted ‘Braak theory’ spread of alpha synuclein, and 7% of those cases did not have any cell loss in the dorsal motor nucleus (one of the first sites of damage in the Braak theory – Click here to read more).
Ok, so the theory is not perfect…what are Prof Isacson and Dr Engelender proposing instead?
They suggest that alpha synuclein accumulation starts at about the same time in nerve cells throughout the body, but the different groups of nerve cell differ in how much toxicity they can handle.
Some of these groups of cells can handle a lot (and more than half of the cells need to be lost before clinical features begin to appear), while others have a lower ‘threshold’ (only a few cells need to die before symptoms appear).
Prof Isacson and Dr Engelender argue that the nerve cells around the gut, for example, have a lower reserve (or total number), and, therefore, symptoms related to the gut become more obvious sooner as those cells die off or become less efficient. This lower threshold is in contrast to the more well known cell loss of the dopamine producing neurons in the midbrain, where approximately 50-70 percent of the dopamine neurons disappear before the classical motor features of Parkinson’s start to appear. Their theory suggests that this part of the brain has a larger reserve, and thus higher threshold.
Hence the reason why this is being called the ‘threshold theory’.
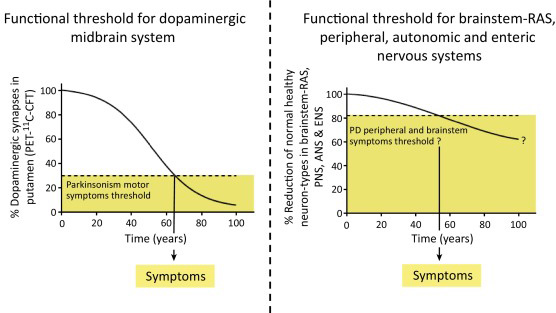
Some groups of cells may have a higher threshold in Parkinson’s disease. Source: Cell
Some cells may have a low threshold and only require a few cells to be lost before the clinical features associated with those cells begin to appear. These symptoms would obviously appear earlier than those features associated with a high threshold population of cells, which required substantial loss before symptoms appear.
This idea would explain differing results seen in research findings regarding, for example, vagotomies (the cutting of the vagus nerve to the gut – click here to read more about this). This new theory would suggest that the procedure might not have any impact at all on lowering the risk of Parkinson’s disease.
Both scientists insist that searching for treatments that slow or block the aggregation of alpha synuclein is still necessary.
“Instead of studying how proteins move from one neuron to another and searching for compounds that prevent the ‘spread’ of aggregated alpha-synuclein, we need to study why alpha-synuclein accumulates within neurons and how these neurons die in the disease, and search for compounds that prevent the general neuronal dysfunction,” – Dr Engelender
(Source: Science Daily)
So are there any problems with this new theory?
The new theory is a very interesting idea and deserves consideration. It solves some of the problems with the “ascending theory” discussed further above. But it also faces some of the same problems that the ascending theory has to deal with.
For example, in one large autopsy study which investigated 904 brains, the investigators blindly collected all of the brains that had alpha synuclein present in the groups of neurons that are affected in Parkinson’s disease (eg. the dorsal motor nucleus of vagus, substantia nigra, and/or basal forebrain nuclei.). They found that alpha synuclein was observed in 11.3% (or 106 cases). But when the researchers then looked at the clinical notes associated with those cases, only 32 (30%) had been diagnosed with a neurodegenerative disorder. The rest had demonstrated no clinical features.
Another study found that 8.3% of the aged control brains had alpha synuclein present in them. In addition, the presence of alpha synuclein is not specific to Parkinson’s disease – approximately 50% of people who die with Alzheimer’s disease have been found to have Lewy bodies. These results suggest that alpha synuclein aggregation can be present in both healthy and diseased brains. But if this is so, what role is alpha synuclein playing in Parkinson’s disease?
(You see the sort of problems we are dealing with in research when trying to come up with a theory of how something complicated is actually working?)
What does it all mean?
The central job of a scientist is to test hypotheses.
A hypothesis is a true or false statement (for example, hypothesis: the sun will come up tomorrow – easy to test as the sun either will or won’t come up; the statement is either true or false). In building one hypothesis on top of another hypothesis, we develop theories about how the world around us works.
Sometimes our hypotheses can unwittingly take us in a particular direction, depending on different variables. The danger in this process (one which must be met with discipline and control procedures) is that one can start to look for results that support a hypothesis or theory. It is a very human characteristic to become blind to any evidence to the contrary.
A new theory of Parkinson’s disease has been proposed. It suggests that rather than the condition starting in one location and progressively moving higher into the brain, Parkinson’s disease may actually start everywhere and it is the varying levels of tolerance between different types of cells that determines which cells die first.
It is certainly a new take of the available evidence and the research community is considering it. It will be interesting to see what kind of feedback results from this article, and we will post updates on that feedback as they become available.
The banner for today’s post was sourced from Sott
 A PhD viva examination. Source: Guardian
A PhD viva examination. Source: Guardian