|
# # # # Researchers have been developing vaccines for Parkinson’s in the hope of not only treating individuals currently affected by the condition, but also limiting the incidence of the condition in future generations. Some of these vaccines are being clinically tested and the results are encouraging. In today’s post, we will review clinical trial results recently published by a biotech company called Vaxxinity and what comes next for the field. # # # # |
 Lady Mary Wortley Montagu. Source: NationalTrust
Lady Mary Wortley Montagu. Source: NationalTrust
The return of Lady Mary Wortley Montagu (née Pierrepont) to England in 1721 represented a monumental – but little appreciated moment – in Western medical history.
Five years earlier, her husband Edward Wortley Montagu had been appointed Ambassador to the Ottoman empire, and they had moved to Constantinople (now Istanbul). In March 1717, the 27 year old Mary – whose only brother had died from smallpox – witnessed the practice of inoculation against smallpox called variolation, which she herself called “engrafting” when she wrote home about it in her letters.
 Source: Guardian
Source: Guardian
Variolation was the method of inoculation used to immunize individuals against smallpox (Variola) with material taken from a patient (or a recently variolated individual), in the hope that a mild infection would result and provide protection. Only 1–2% of those variolated died from the induced infection, but this was much better than the >30% who contracted smallpox naturally.
In 1718, Lady Mary Wortley Montagu asked that her five-year-old son, Edward be inoculated against smallpox, and after the procedure, wrote to her husband:
“The boy was engrafted last Tuesday, and is at this time singing and playing, and very impatient for his supper” (Source)
And before they left Turkey in 1721, she had her daughter Mary inoculated as well. Mary and her brother were the first English people to be immunized against a disease. Upon her return to London, she enthusiastically promoted the idea of variolation. Unfortunately she encountered a great deal of resistance from the medical establishment, and the idea didn’t really catch on.
This is very interesting, but what does this have to do with Parkinson’s?
Well, for quite a while now, researchers have been trying to develop a method of innoculating people against Parkinson’s. A vaccine for the condition.
How does a vaccine actually work?
A vaccine involves exposing the body’s immune system to a particular piece of a protein or disease-causing agent (think: a virus), and helping to prepare the defenses against an actual attack from said pathogen (or a disease-causing agent). The key feature of the vaccine is that it can’t cause disease itself. So, scientists often use the membrane of a virus, with all of the disease-causing machinery inside it removed.
By injecting in an inert portion of a virus, the immune system can become aware and start to keep a look out for that pathogen. And that way, when the pathogen eventually attacks, the immune cells of the body can get rid of it before it does too much damage or causes us to get sick.
But how can one make a vaccine for Parkinson’s? Does this mean that Parkinson’s is caused by a virus?
Scientists have learned a lot in the development of vaccines, and they can now make vaccines that allow us to target not just pathogens, but also rogue cells (such as cancer cells) or rogue proteins that are believed to be causing trouble.
 Immune cells attacking a cancer cell. Source: Lindau-nobel
Immune cells attacking a cancer cell. Source: Lindau-nobel
Interesting, but how is a vaccine being made for a neurodegenerative condition like Parkinson’s?
One of the big theories about how Parkinson’s progresses involves the idea that a toxic form of the Parkinson’s-associated protein, alpha synuclein, could be being passed from cell to cell.
And as this toxic version of alpha synuclein is absorbed by each new healthy cell, it starts causing trouble in that healthy cell and seeding new clustering (or aggregation) of the protein, which is believed to lead to the appearance of Lewy bodies in those previously healthy cells.
What are Lewy bodies?
Lewy bodies are dense circular clusters of alpha synuclein protein (and other proteins) that are found in specific regions of the brain in people with Parkinson’s (Click here for more on Lewy bodies).
 A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
The aggregated alpha synuclein protein, however, is not limited to just the Lewy bodies. In the affected areas of the Parkinsonian brain, aggregated alpha synuclein can be seen in the branches (or neurites; see black arrow in the image below) of cells – see the image below where alpha synuclein has been stained brown on a section of brain from a person who passed away with Parkinson’s.
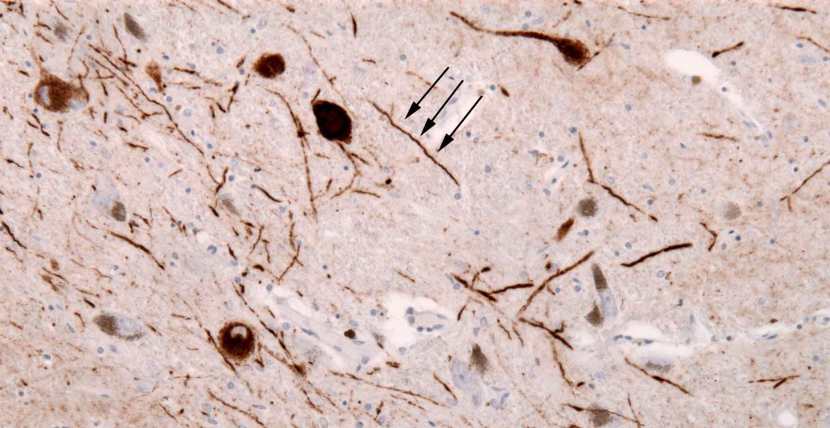
Examples of Lewy neurites (indicated by arrows). Source: Wikimedia
This aggregated form of alpha synuclein is believed to be toxic, which may be causing the loss of certain neurons in the brains of people affected by Parkinson’s. And it is this aggregated form of alpha synuclein that some researchers believe is being passed from cell to cell, seeding Lewy body pathology as it is being passed on.
Is there any evidence that alpha synuclein protein can be transferred between cells?
Back in the 1990s, there were a series of clinical trials of cell transplantation conducted on people with Parkinson’s. The idea was to replace the cells that have been lost to the condition (Click here to read a previous post about cell transplantation). Many of the individuals who were transplanted have now passed away by natural causes and their brains have been examined post-mortem.
One very interesting finding from the analysis of those brains is that some of the cells in the transplants have Lewy bodies in them (up to 10% of transplanted cells in one case – Click here to read the research report on that case).
 Above are photos of neurons from the post-mortem brains of people with Parkinson’s that received transplants. White arrows in the images above indicate Lewy bodies inside transplanted cells. Source: The Lancet
Above are photos of neurons from the post-mortem brains of people with Parkinson’s that received transplants. White arrows in the images above indicate Lewy bodies inside transplanted cells. Source: The Lancet
This finding suggested to researchers that somehow this neurodegenerative condition may be being passed on from the Parkinson’s affected brain to the healthy transplanted cells.
And researchers have proposed that the toxic form of the Parkinson’s-associated protein alpha synuclein may be the guilty party in this cell-to-cell transfer process. Being passed from one cell to another and seeding further protein aggregation as they get passed along (Click here to read more on this idea and the evidence for it).
So a vaccine for Parkinson’s is targeting this toxic form of alpha synuclein?
Exactly.
One way of dealing with this problem of cell-to-cell transfer of the toxic form of alpha synuclein is to grab it as it is being passed between the cells, and remove it from the body. Think of it as vacuuming up all of the toxic form of alpha synuclein that is flowing around outside of cells.
And using a vaccine approach, we can stimulate the body’s immune system to generate antibodies that target the toxic form of the alpha synuclein protein.
What are antibodies?
Antibodies are Y-shaped proteins that the immune system naturally and continuously produces to identify anything in the body that is ‘not self’ (that is, not a normally occurring part of you – think of viruses, bacteria, etc).

Monoclonal antibodies. Source: Astrazeneca
Antibodies act like alert flags for the immune system. When antibodies bind to something, they alert the immune system to investigate and potentially remove the bound object. Each antibody targets a very specific structure, while ignoring everything else.
In this fashion, antibodies are a very powerful method of removing items from the body that are causing trouble or not wanted. And researchers have adapted this natural system for Parkinson’s using immunotherapy approaches.
How does the immune system make antibodies?
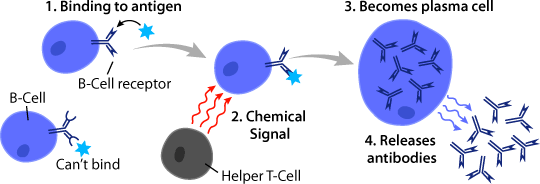
When a pathogen enters the body, and the immune system recognizes that it is “not self”, a class of white blood cells called B-cells are activated. Each B-cell has a receptor that will connect to only one antigen shape. An antigens are substances that trigger the body’s immune system.
When a particular B-cell receptor connects to its specific antigen, another white blood cell called a Helper T-cell releases chemicals that tell that B-cell to start dividing. This effort results in an army of B-cells with the perfect shaped receptor to detect the pathogen in your body.
 Source: Askabiologist
Source: Askabiologist
Now many of these B-cells quickly turn into plasma cells. Plasma cells are the cells that make and release antibodies. These antibodies attach to the same antigen as the original B-cell receptor. Plasma cells are extremely efficient at their task as they can make thousands of antibodies per second, and these freshly baked antibodies spread throughout the body, sticking a red flag on any other copies of that pathogen.
So vaccines involve a fragment of a pathogen or rogue protein being injected into the body and causing an immune response that results in antibodies being produced for that pathogen/rogue protein?
In a nut shell, yes.
And researchers have been applying this to the Parkinson’s protein alpha synuclein in the hope of clearing excess aggregating alpha synuclein.
Ok. Do these vaccines work?
Well, there have been two major clinical trial programs exploring alpha synuclein targeting vaccines for Parkinson’s. And recently one of the research teams published new data from their Phase 1 clinical studies.
This is the report here:
 Title: Target engagement and immunogenicity of an active immunotherapeutic targeting pathological α-synuclein: a phase 1 placebo-controlled trial.
Title: Target engagement and immunogenicity of an active immunotherapeutic targeting pathological α-synuclein: a phase 1 placebo-controlled trial.
Authors: Eijsvogel P, Misra P, Concha-Marambio L, Boyd JD, Ding S, Fedor L, Hsieh YT, Sun YS, Vroom MM, Farris CM, Ma Y, de Kam ML, Radanovic I, Vissers MFJM, Mirski D, Shareghi G, Shahnawaz M, Singer W, Kremer P, Groeneveld GJ, Yu HJ, Dodart JC.
Journal: Nat Med. 2024 Sep;30(9):2631-2640.
PMID: 38902546 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers were testing a vaccine that was produced by the biotech firm Vaxxinity. And over a 44-week, randomized, placebo-controlled, double-blind, single-center Phase 1 study, the scientists investigated the safety, tolerability and immunogenicity of UB-312.
Wait a minute. What does “immunogenicity” mean?
It is basically the ability of a substance or agent to cause an immune response in the body. So the researchers were looking for the ability of UB-312 to make the body produce antibodies against the aggregating form of alpha synuclein.
Got it. So what did they find?
The scientists wanted to investigate different doses of their vaccine UB-312, so they recruited 20 Parkinson’s patients and randomized them using a 7:3 (UB-312 : placebo) ratio into two groups:
The first group received 300μg of UB-312 in Week 1 of the study, and then they received ‘booster’ shots of 100 μg at weeks 5 and 13 of the 44 week study.
The second group received 300μg of UB-312 in Week 1 of the study, and then they received ‘booster’ shots of 300 μg at weeks 5 and 13 of the 44 week study.
The vaccine was found to be safe and well tolerated at both doses across the study. The adverse events that did occur were mostly mild and transient. After all three shots of UB-312, anti-alpha synuclein antibodies were found in the blood of five of the six participants in the first group (300/100/100 μg) and all seven participants in the second group (300/300/300 μg). Antibodies were found in the cerebrospinal fluid (the liquid that the brain sits in) collected from 5/13 patients who received all three UB-312 doses (curiously, only one participant in the higher dose regime had antibodies in the cerebrospinal fluid):
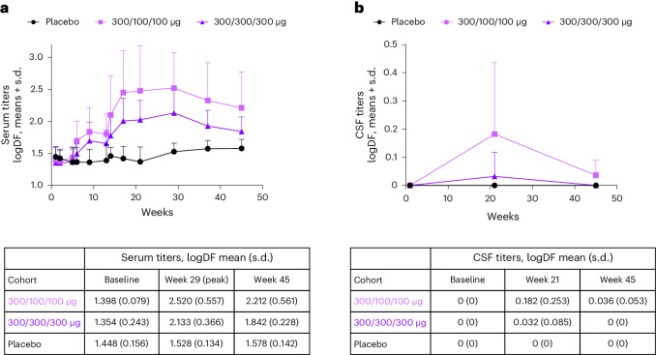 UB-312 or placebo were administered at weeks 1, 5 and 13. Serum (blood) measures in panel A. and cerebrospinal fluid (CSF) in panel B. Source: PMC
UB-312 or placebo were administered at weeks 1, 5 and 13. Serum (blood) measures in panel A. and cerebrospinal fluid (CSF) in panel B. Source: PMC
Despite the low level of antibodies in the cerebrospinal fluid, when the level of alpha synuclein was measured in the cerebrospinal fluid was measured at 45 weeks, the investigators found the low dose UB-312 group (300/100/100 μg) had a significant 19.8% decrease (P < 0.05) in alpha synuclein levels and the higher dose UB-312 group (300/300/300 μg) had a (non-significant) 15.2% decrease in levels, compared to their baseline measures before initiating treatment.
At the start of the study, 19 of the 20 participants tested positive for the alpha synuclein seeding assay test (Click here to read a previous SoPD post on this topic). Curiously, at the end of the study one participant in the high dose UB-312 group (300/300/300 μg) who started the study with a positive alpha synuclein seeding assay test result, was negative at the end of study.
Was their any change in the progression of Parkinson’s symptoms?
The investigators used the Montreal Cognitive Assessment (MoCA) and the Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS–UPDRS) to evaluate the progression of symptoms. The study was not really long enough or large enough to make a proper measure of disease progression, but the investigators did report that both the MoCA and UPDRS scores were generally stable during the study, with no statistical differences between the treatment and placebo groups.
Interesting. You said there were “two major clinical trial programs exploring alpha synuclein targeting vaccines for Parkinson’s”. If Vaxxinity is one of them, who is the other?
The other group is a Switzerland-based biotech company called AC Immune.
 And in 2021, the company announced that they would be purchase the Parkinson’s-associated vaccine assets from an Austrian biotech company called AFFiRiS.
And in 2021, the company announced that they would be purchase the Parkinson’s-associated vaccine assets from an Austrian biotech company called AFFiRiS.
Of specific interest to AC Immune in this deal was an alpha synuclein targeting vaccine called PD01A.
In 2020, the AFFiRiS team published their Phase 1 clinical trial results of PD01A. The results were published in the prestigous medical journal Lancet Neurology: Title: Safety and immunogenicity of the α-synuclein active immunotherapeutic PD01A in patients with Parkinson’s disease: a randomised, single-blinded, phase 1 trial.
Title: Safety and immunogenicity of the α-synuclein active immunotherapeutic PD01A in patients with Parkinson’s disease: a randomised, single-blinded, phase 1 trial.
Authors: Volc D, Poewe W, Kutzelnigg A, Lührs P, Thun-Hohenstein C, Schneeberger A, Galabova G, Majbour N, Vaikath N, El-Agnaf O, Winter D, Mihailovska E, Mairhofer A, Schwenke C, Staffler G, Medori R.
Journal: Lancet Neurol. 2020 Jul;19(7):591-600.
PMID: 32562684
In this report, the researchers outlined the results of their Phase I safety/tolerability studies for PD01A in people with recently diagnosed Parkinson’s. And I write ‘studies’ because the Phase I project involved 3 consecutive study extensions, resulting in most of the participants being followed for more than 3 years.
The first study involved 24 individuals (45–65 years of age; all no more than 4 years since diagnosis) being recruited before February 2013. They were randomly assigned to one of two treatment groups based on dose:
- 15ug of PD01A (n=12)
- 75ug of PD01A (n=12)
They were given four ‘priming’ injections of PD01A (at baseline, 4, 8, & 12 weeks into the study) and then they were followed out to 52 weeks with assessments, before starting a further 39 week follow up period.
 Source: Alzheimersweekly
Source: Alzheimersweekly
After this initial study, an extension study was initiated that involved a re-randomisation of the participants (to either the 15ug or 75ug group) and a booster shot (another injection of the vaccine). The participants were then followed for a further 24 weeks of assessments.
And after this, the participants were invited to take part in a third extension study, which involved another booster shot, but this time they all received the 75ug injection and were followed for a further 52 weeks assessments.
As far as Phase I studies go, this was really long. But AFFiRiS – obviously aware of the safety issues with a first-in-human vaccine study – wanted to be safe and thorough.
And what did the results suggest?
First, the team found that the treatment is safe and well tolerated in the participants.
In addition, the vaccine also caused the immune system to start producing alpha synuclein targeting antibodies.
Did they reduce alpha synuclein levels in the body?
The concentration of total alpha synuclein protein in the cerebrospinal fluid did not change as a result of the vaccine treatment. But the researchers reported that at 26 weeks into the study, they observed a 51% reduction in cerebrospinal fluid levels of the aggregated form of alpha synuclein in the 75ug group.
The aggregated form of alpha synuclein makes up just a small percentage of the total levels of alpha synuclein – hence the reason there was no reduction in total levels.
How long did the vaccine effect last?
The researchers reported that two years into the study, the levels of alpha synuclein targeting antibodies had dropped back to near baseline levels, but the investigators found that the immune system could be rapidly reactivated after booster shots of the vaccine.
And what did the vaccine do for the Parkinson’s symptoms of the study participants?
Ok, so like the Vaxxinity study discussed above, it is important to remember that this is an open label study – meaning that all of the participants knew what they were being treated with. In such situations, there is an increased risk of having a placebo response to a treatment (this is a situation where an individual experiences benefits that may have no biological association with the treatment being administered).
Placebo responses are an issue in Parkinson’s trials as individuals with the condition appear to be more prone to experiencing them (Click here to read an old SoPD post on this topic).
In addition, we have to be careful in managing expectations. We do not want to be raising expectations, if in the long term a treatment does not go on to be approved by health regulators.
So, with all of that said: Regarding assessments of disease progression, the researchers wrote in their report that:
“DAT-SPECT examinations did not show statistically significant changes up to 91 weeks in study 1. MDS-UPDRS part 3 scores were generally stable across the studies”
What does any of that actually mean?
Two important details here:
- DAT-SPECT is a type of brain imaging technique (specifically, it is a type of single-photon emission computed tomography (SPECT) imaging technique that allows for the visualisation of dopamine transporter (DAT) levels in the brain). Dopamine transporter is a protein on the membrane of dopamine neurons involved in dopamine turnover. By labelling DAT, one can get an indication of how many dopamine terminals there are in the brain.
- The Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) is the most commonly used clinical assessment in clinical trials. Part 3 of the scale focuses on the motor features of the condition. There are 33 items assessed (in effect there is only 18 items, but several address which side of the body, or which limb is affected). An increase in UPDRS score indicates progression of Parkinson’s features, while a lower score suggests correction or improvement (Click here to read a previous SoPD post about the MDS-UPDRS).
Now remembering that this is just a small open-label study not designed to test efficacy of the vaccine, the researchers were reporting that they saw no deterioration in DAT levels (according to the DAT-SPECT brain imaging), and no change in clinical assessments of motor symptoms across multiple years of assessment.
They state “Mean MDS-UPDRS part 3 scores in the pooled 15 μg group were 11·9 (SD 8·2) at baseline and 12·5 (14·8) at the last visit. In the pooled 75 μg group, MDS-UPDRS part 3 scores were 12·3 (7·2) at baseline and 8·6 (7·7) at the last visit”
And they added that this stablisation of the clinical features of Parkinson’s was “in contrast to previously published data which reports a worsening in MDS-UPDRS part 3 scores in a similar population of patients with early Parkinson’s” (they cite this reference to support this statement).
So the vaccine slowed progression?
While the results may appear encouraging, please remember that all of the research discussed in today’s post is coming from small, open label Phase 1 studies not designed to test efficacy. Interpretation of the results must be handled with great caution. In both cases a larger, double-blind Phase 2 double blind study is what is really required to determine if there is any evidence of efficacy.
Currently all we can really take away from these studies is that the treatment was safe and well tolerated in individuals with Parkinson’s over multiple years, and that the vaccines can stimulate the production of anti-alpha synuclein antibodies.
When do the Phase 2 studies start?
Well, it already has in the case of AC Immune.
They are conducting the Phase 2 “VacSYn” trial, which is a multicenter, placebo-controlled, double-blind, randomized study of their vaccine ACI-7104.056 in 150 people with recently diagnosed Parkinson’s. The study involves a screening period of up to 8 weeks, a 74-week double-blind treatment period, and a 26-week post-treatment follow-up period (Click here to read more about this study). The study started in 2023 and is scheduled to complete in early 2028.
And what about Vaxxinity?
The company began a Phase 1b trial in May 2023 involving four patients with Multiple Systems Atrophy (a more rapidly progressing from of Parkinsonism) and four with Parkinson’s. They all agreed to receive three priming and five booster injections of 300 μg UB-312 over two years. The study is expected to finish in April 2025 (Click here to read more about this study), so hopefully later next year we will learn the results and the Phase 2 plans for UB-312.
So what does it all mean?
The “Father of Vaccination” Edward Jenner, was only 13 years old when Lady Mary died in 1762, but he would go on to develop a much safer technique of inoculation (using cowpox instead of smallpox). And it should be noted that there was considerable resistance to his findings from the medical establishment of the time. Good things take time it seems.
 Edward Jenner. Source: Historyofvaccines
Edward Jenner. Source: Historyofvaccines
But vaccines are a good thing. Despite a lot of strange opinions about vaccines, the simple fact is that a very large portion of us would not be here if not for the success of vaccination. Along with antibiotics, vaccines have been developed into a very powerful weapon in the arsenal protecting against the microbial world.
And now scientists are taking the knowledge that they have built up and applying it to conditions like Parkinson’s. It is rather remarkable how far humans have come in a very short space of time. The hope is of course that one day, there will be a vaccine that is given to everyone over a certain age that will protect them from Parkinson’s (and perhaps other neurodegenerative conditions like Alzheimer’s), and this will allow for the prevention, if not delayed onset, of these debilitating diseases..
# # # # # # # #
ADDENDUM: November 14th 2024
Today AC Immune provided interim results from their Phase 2 “VacSYn” trial of ACI-7104.056 in early Parkinson’s. The study is broken into two part:
Part A involves 30 individuals with recently diagnosed Parkinson’s randomized to receive ACI-7104.56 or placebo (at a ratio of 3:1). This part of the study is ongoing, but the investigators reported that ACI-7104.056 is again safe and well tolerated, and positive antibody responses have been effectively induced against alpha synuclein by week 6 (after 2 immunizations).
Based on the collection of further results to be reported in H1 2025, AC Immune will then decide whether to initiate Part B of the VacSYn trial which will involve up to 150 patients. The participants in this second Part will also be assessed for progression of motor and non-motor symptoms, using clinical, digital, imaging, and fluid biomarkers (Click here to read the press release).
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The author of this post is an employee of Cure Parkinson’s, so he might be a little bit biased in his views on research and clinical trials supported by the trust. That said, the trust has not requested the production of this post, and the author has not had any communication with the companies mentioned. He is sharing this post simply because it may be of interest to the Parkinson’s community.
The banner for today’s post was sourced from Vaxxinity.

