|
# # # # At the end of each month, the SoPD writes a post which provides an overview of some of the major or interesting pieces of Parkinson’s-related research that were made available during April 2026. The post is divided into 10 parts based on the type of research:
# # # # |
So, what happened during April 2026?
In world news:
1st April – The basketball world was still recovering from the Duke vs UConn March Madness Elite Eight game: (coach Hurley’s response was a hilarious roller coaster of emotion (starting from 23 to 50 seconds in the video below) and his parents in the audience are probably still recovering (from 1.55 to 2.08 minutes – no lip-synching required!)):
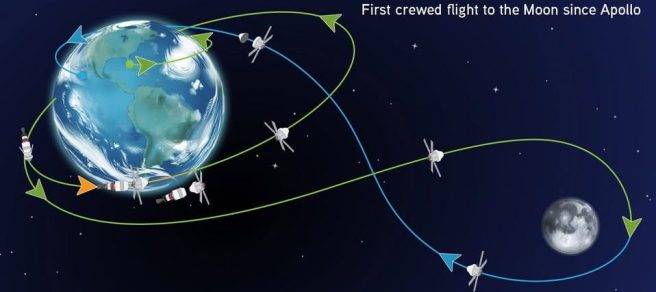
6th April – The crew of Artemis II breaks the record for the furthest humans have ever been from Earth, reaching a maximum distance of 252,757 miles (406,773 km) as they travel around the far side of the Moon (Click here to read more about this).
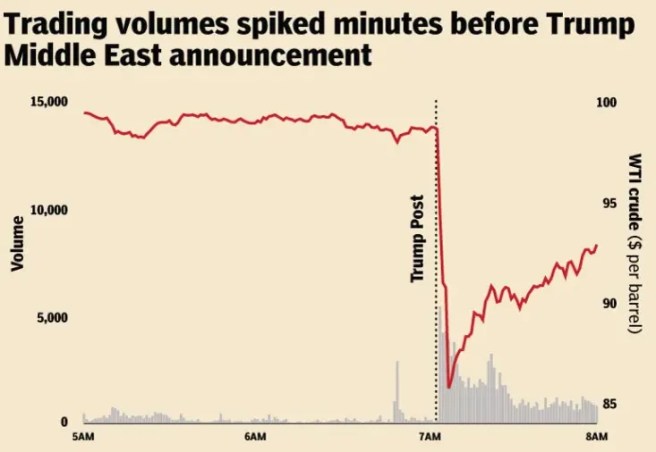
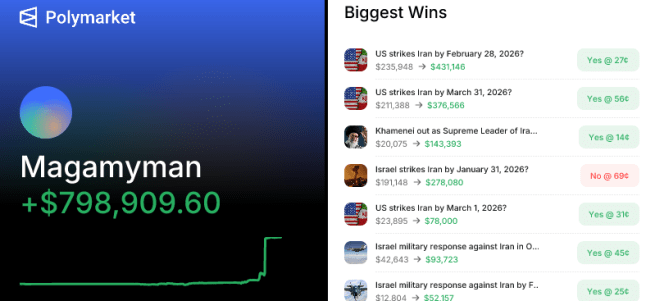
7th April – Traders placed bets worth around $950 million just hours ahead of the U.S. and Iran announced a two-week ceasefire (Click here to read more about this). And 10 days later (17th April) Deja vu: Traders placed bets worth about $760 million on a falling oil price just 20 minutes before U.S. and Iran announced on Friday that the Strait of Hormuz was open – well,… at least someone is winning…. right??? I wonder if they are getting tired of all that winning? (Click here to read more about this).
22nd April – During a Senate Finance Committee hearing, the US Health and Human Services Secretary, Robert F. Kennedy Jr, said “President Trump has a different way of calculating percentages. There’s two ways of calculating percentages. If you have a $600 drug and you reduce it to $10, that’s a 600% reduction” (Click here to read more about this). Six days later, Nature published an interesting news post on how the US Government has terminated more than 100 advisory committees to science agencies – 77 of them in the National Institutes of Health (Click here to read that piece)..
28th April – Japan Airlines announced that they will introduce humanoid robots to become baggage handlers in a trial run at Tokyo airport amid country’s surge in inbound tourism and worsening labour shortages/demographic issues (Click here to read more about this).
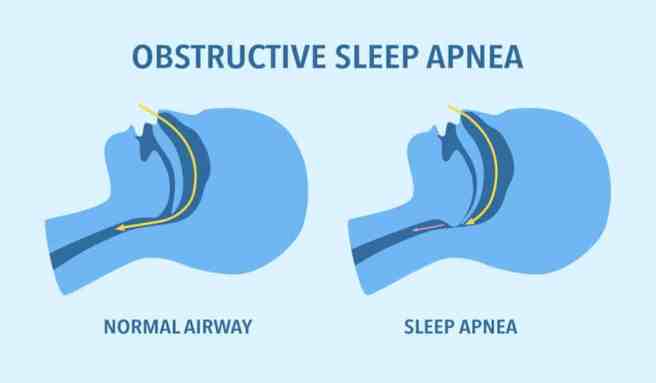
In the world of Parkinson s research, a great deal of new research and news was reported:
In April 2026, there were 1,003 research articles added to the Pubmed website with the tag word “Parkinson s” attached (compared to 5,177 for all of 2026). In addition, there was a wave to news reports regarding various other bits of Parkinson s research activity (clinical trials, etc).