|
# # # # One of the most common questions I get from SoPD readers is what’s new with inflammasome research? Another version of this question is where are the clinical trials for NLRP3 inhibitors in Parkinson’s? Readers have become very enchanted by this new class of anti-inflammatory drugs as a potential future treatment for Parkinson’s – and there is preclinical evidence to support this vibe. But the clinical development of these experimental therapies has been slow. Recently, the pharmaceutical company Roche has initiated Phase 1b testing of their NLRP3 inhibitor (called Selnoflast) in people with Parkinson’s – the first in this class. In today’s post, we will discuss what the inflammasome is, how NLRP3 inhibitors work, and what the new clinical trial involves. # # # # |
 On the 21st September 2020, the website for an Ireland-based biotech company called Inflazome suddenly disappeared. In its place was a single page, that stated the large pharmaceutical company Roche had purchased the biotech firm and taken on all of its inflammasome-targeting intellectual property (Source).
On the 21st September 2020, the website for an Ireland-based biotech company called Inflazome suddenly disappeared. In its place was a single page, that stated the large pharmaceutical company Roche had purchased the biotech firm and taken on all of its inflammasome-targeting intellectual property (Source).
This was a big deal for folks who were watching the inflammasome research world. It suggested that the big players (pharma) were now interested in this space ($449 million interested in the case of Inflazome). And since then, there has been a rush of other pharma companies buying or developing inflammasome-targeting agents.
The Inflazome purchase was also interesting because the company was targeting Parkinson’s as one of their indications of interest.
And it would appear that Roche is now following up on this interest, having initiated a clinical trial program focused on inflammasomes in Parkinson’s.
Hang on a second. Remind me, what are inflammasomes?
Inflammasomes are multi-protein formations that are present inside of cells in your body. They detect pathogenic agents or stressors that have found their way inside of cells, and once these inflammasomes detect something that should not be there, they activate the release of highly pro-inflammatory messenger proteins (called cytokines) such as interleukin-1b (IL-1b) and IL-18.
These cytokines are released into the world outside of the cell and alert the immune system that something is not quite right.
 Source: Youtube
Source: Youtube
Think of inflammasomes as one of the amplifiers of the immune system.
There are difference types of inflammasomes and they vary based on what activates them. For example, the presence of RNA from a particular virus may activate one type of inflammasome, while a certain toxin will cause the assembly of a different inflammasome.
For the purpose of keeping things simple in today’s post, we are going to focus on one of the most well characterised inflammasomes.
It is called the NLRP3 inflammasome.
What does the NLRP3 part mean?
The name of this particular inflammasome is derived from one of the three core components: a protein called NLRP3.
 The NLRP3 inflammasome. Source: Twitter
The NLRP3 inflammasome. Source: Twitter
NLRP3 is an abbreviation that is a lot easier to say than “Nucleotide-binding domain, Leucine-Rich-containing family, Pyrin domain-containing-3“.
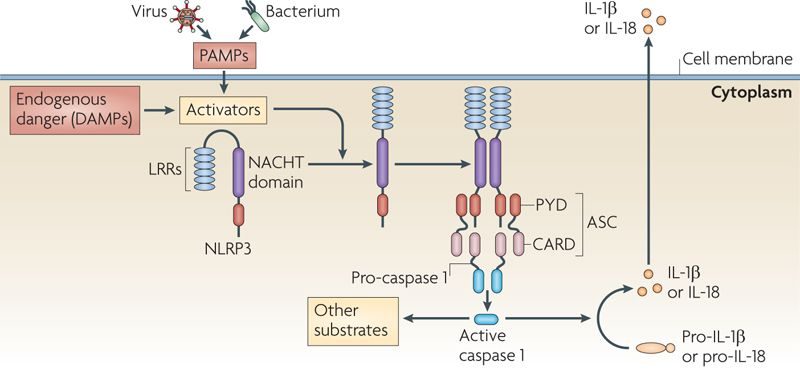
As I mentioned above, inflammasomes are multi-protein formations. Critically, inflammasomes are made up of three primary components:
- a sensor protein
- an adaptor protein
- a zymogen procaspase-1 (don’t panic about how complicated this last one sounds just yet, we’ll come back to it in a second!).
NLRP3 is a sensor protein.
It act as a “pattern-recognition receptor“. That is to say, NLRP3 is a protein that recognises a particular pattern in the shape or structure of certain ‘activators’ (think, a virus or stressor). These activator patterns are called pathogen-associated molecular patterns (or PAMPs). There are many different forms of sensors, which help to recognise all the various patterns of troublesome agents.
 Activation of NLRP3 inflammasome. Source: Nature
Activation of NLRP3 inflammasome. Source: Nature
When NLRP3 protein is first produced by a cell, it floats around in an inactive state waiting for PAMPs to interact with it and activate it.
NLRP3 can actually be activated by a wide range of agents, including:
- low intracellular potassium concentrations
- Viruses (such as influenza A and Hepatitis C)
- Bacteria (including neisseria gonorrhoeae)
- Bacterial toxins (such as nigericin and maitotoxin)
- Inorganic particles (think titanium dioxide, silicon dioxide, and asbestos)
- Crystallized molecules (like cholesterol crystals and urate crystals in atherosclerosis and gout, respectively)
Once activated, the NLRP3 protein will start binding to other activated NLRP3 proteins and this is the beginning of the formation of a NLRP3 inflammasome.
And this is where the second component of the inflammasome comes into the picture: the adaptor protein
This is getting complicated. What is the adaptor protein?
Don’t panic.
If you are not interested in the biology lesson here, skip down to the first recap and read on from there.
In the NLRP3 inflammasome, there is a protein called PYCARD, which is sometimes referred to as ASC (or “Apoptosis-associated speck-like protein containing a CARD”). This is the adaptor protein. For simplicity sake, for the rest of this post, we will refer to the adaptor protein in the NLRP3 inflammasome as ASC.
An interesting feature of the adaptor protein ASC has recently been shown to act in a prion-like fashion (Click here to read more about this). Emerging evidence suggests that ASC is also released from inflammasome‐activated cells as ‘ASC specks’. These ASC specks accumulate in inflamed tissues, where they can continue to encourage the production of mature cytokines. There is also evidence of interactions with other proteins.
When the NLRP3 protein becomes activated and starts binding to other activated NLRP3 proteins, ASC will bind to it and this process attracts the zymogen procaspase-1.
Ok, now can I panic? That sounds REALLY complicated. What is the zy…mo…gen pro…cas…pase-1?
A zymogen is simply an inactive precursor of an enzyme, and procaspase-1 is the precursor to the enzyme caspase-1.
And what is caspase-1?
Caspase-1 is the enzyme that causes the release of the proinflammatory messenger protein (the cytokines that we mentioned above), IL-1b and IL-18.
So the take home message here is: By forming an inflammasome, NLRP3, ASC, and procaspase-1 cause the activation of caspase-1 which in turn results in the release of proinflammatory cytokines.
All of it acting as an amplifier for the immune system.
|
# RECAP #1: Inflammation is a natural process that our bodies use to let the immune system know that something is wrong. By sending out tiny messenger proteins, damaged cells can activate the immune system to respond. Sometimes a stronger than normal immune response is required and this is where ‘inflammasomes’ can help to amplify the message being sent to the immune system. The NLRP3 inflammasome is one type of immune system amplifier. # |
So what does this have to do with Parkinson’s?
In late 2018, this report was published:
 Title: Inflammasome inhibition prevents α-synuclein pathology and dopaminergic neurodegeneration in mice
Title: Inflammasome inhibition prevents α-synuclein pathology and dopaminergic neurodegeneration in mice
Authors: Gordon R, Albornoz EA, Christie DC, Langley MR, Kumar V, Mantovani S, Robertson AAB, Butler MS, Rowe DB, O’Neill LA, Kanthasamy AG, Schroder K, Cooper MA, Woodruff TM.
Journal: Sci Transl Med. 2018 Oct 31;10(465)
PMID: 30381407 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers began by assessing postmortem human brains for signs of inflammasome activation. They used samples from 5 late-stage Parkinson’s brains and 5 control brains, and they found evidence of increased cleaved caspase 1 and ASC (hallmarks of inflammasome activation) in the Parkinsonian brains. When they looked at which cell types were producing the markers of inflammasomes in the PD brains, they found that the resident immune cells – microglia – were activated and had increased levels of NLRP3 and ASC.
Next the investigators looked at blood samples collected from 21 people with Parkinson’s and they found further evidence of inflammasome activation, suggesting that a ‘systemic’ (body-wide) increase in inflammasome activity may be occurring in Parkinson’s.
 Source: PMC
Source: PMC
The researchers then shifted their attention to animal models of PD, to determine if inflammasome activation is also occurring. Across multiple models (neurotoxin-based, genetic, and alpha-synuclein preformed fibrils), they found evidence of NLRP3 inflammasome activation.
Given that fibrils of alpha synuclein can cause NLRP3 activation (see above), the researchers in this current study wanted to evaluate the response of microglial cells to preformed fibrils of alpha synuclein protein. They collected microglia cells from normal mice and mice that were genetically engineered to have no NLRP3. They grew these cells as separate cultures and then exposed the cells to preformed fibrils of alpha synuclein protein.
They found that even when normal microglia were exposed to preformed fibrils of alpha synuclein protein, they would start to form characteristic ASC specks and release IL-1b 24 hours later. When microglia with no NLRP3 were exposed to preformed fibrils of alpha synuclein protein there was no release of IL-1b or formation of ASC specks (even at 24 hours after treatment). Interestingly, the alpha synuclein exposure did not cause pyroptosis in the microglia from normal mice.
Given the lack of response in microglia with no NLRP3 protein, the investigators next exposed the normal microglia to a drug called MCC950.
What is MCC950?
MCC950 is a potent small-molecule inhibitor of NLRP3.
When the investigators exposed normal microglia to preformed fibrils of alpha synuclein protein and treated those cells with MCC950, they witnessed a significant reduction in NLRP3 inflammasome activation. Even at very low doses, MCC950 was able to block the release of IL-1b.
As a result of this result, the researchers next turned their attention to models of Parkinson’s. Three models in fact. They tested MCC950 in a neurotoxin model (6-OHDA), a genetic model (MitoPark), and an alpha synuclein model of PD (pre-formed fibrils).
 A lab mouse. Source: USNews
A lab mouse. Source: USNews
Not only did the researchers find that MCC950 entered the brain (crossing the blood brain barrier is one of the great challenges of any PD-oriented treatment), but it also improved motor features and reduced the level of dopamine cell loss in all three of these models of Parkinson’s. In addition, MCC950 was found to be a potent inhibitor of inflammation in the brain, reducing IL-1b, Caspase-1, and ASC levels dramatically.
This is really interesting. What did they do next?
The researchers were interested to have a look at what effect MCC950 treatment may have on the spread of alpha synuclein pathology in the pre-formed fibrils model of Parkinson’s. They analysed the brains of mice 8 months after the fibrils had been injected and they found signs of alpha synuclein in regions beyond the dopamine system in mice that were NOT treated with MCC950 (these regions included the cerebral cortex).
In mice that were treated with MCC950 (following the delivery of pre-formed alpha synuclein fibrils), however, there was a very different picture: While there was no difference in the total amount of alpha synuclein being produced in the brain, there was a marked reduction in the number of alpha synuclein aggregates.
These results led the researchers to conclude that that chronic NLRP3 activation contributes to the propagation of pathology seen in the pre-formed fibrils model, and pharmacological inhibition of NLRP3 using MCC950 can effectively reduce this pathological process.
Other (independent) research groups have also reported increased levels of NLRP3 in the dopamine neurons of people with Parkinson’s. For example:
 Title: NLRP3 expression in mesencephalic neurons and characterization of a rare NLRP3 polymorphism associated with decreased risk of Parkinson’s disease.
Title: NLRP3 expression in mesencephalic neurons and characterization of a rare NLRP3 polymorphism associated with decreased risk of Parkinson’s disease.
Authors: von Herrmann KM, Salas LA, Martinez EM, Young AL, Howard JM, Feldman MS, Christensen BC, Wilkins OM, Lee SL, Hickey WF, Havrda MC.
Journal: NPJ Parkinsons Dis. 2018 Aug 15;4:24.
PMID: 30131971 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers analysed postmortem brain tissue from 17 cases of PD and 11 controls, and found that the PD samples had significantly higher levels of NLRP3:
 Source: PMC
Source: PMC
And elevated NLRP3 levels are not only observed in the brain of people with Parkinson’s, but also in the periphery as well:
 Title: Systemic activation of NLRP3 inflammasome and plasma α-synuclein levels are correlated with motor severity and progression in Parkinson’s disease.
Title: Systemic activation of NLRP3 inflammasome and plasma α-synuclein levels are correlated with motor severity and progression in Parkinson’s disease.
Authors: Fan Z, Pan YT, Zhang ZY, Yang H, Yu SY, Zheng Y, Ma JH, Wang XM.
Journal: J Neuroinflammation. 2020 Jan 8;17(1):11.
PMID: 31915018 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers collected blood samples from 43 people with Parkinson’s and 24 control participants, and they assessed each for levels of NLRP3, ASC, and caspase-1. They found that the NLRP3 inflammasome is activated in the blood cells from Parkinson’s patients (see panel B in the image below, where NLRP3 protein levels are significantly elevated compared to controls):
 Source: PMC
Source: PMC
So there is a growing body of evidence to support the idea that NLRP3 inflammasome activation may be playing a role in Parkinson’s (for those interested in reading more on this topic, click here for a very thorough recent review), and this has led to numerous researchers and biotech companies developing inflammasome inhibitors as potential therapies for PD.
|
# # RECAP #2: Researchers have demonstrated that elements of the NLRP3 inflammasome are elevated in people with Parkinson’s (both in the brain and blood). Inhibitors of NLRP3 have been developed and tested in preclinical models of PD, exhibiting neuroprotective benefits. # # |
So what do we know about the Roche clinical trial?
In April of 2022, Roche registered a Phase 1b multi-center, randomized, double blind, placebo-controlled, parallel design clinical study to investigate the safety, tolerability, pharmacokinetics, and pharmacodynamics of their NLRP3 inhibitor Selnoflast (also known as RO7486967) in participants with recently diagnosed idiopathic Parkinson’s (Click here to read more about the trial).
Selnoflast is an orally active, selective, and reversible small molecule NLRP3 inhibitor that has already been clinically tested in ulcerative colitis and found to be safe and well tolerated (Click here to read more about this).
The Phase 1b study in Parkinson’s involves 48 participants being recruited in the USA, UK and the Netherlands. It will require 28 days of treatment (taking either 200mg Selnoflast twice per day or a placebo, ratio of 2:1 Selnoflast to placebo) and then a further 24 days of follow up assessments:
 Source: Medically
Source: Medically
The assessments includes several different types of brain imaging, such as a DAT-Scan (to measure dopamine activity in the brain), a MRI scan (to make sure everything is structurally ok) and a TSPO-PET scan (which assesses levels of inflammation in the brain). Clinical measure and biological samples (such as blood and cerebrospinal fluid) are also being taken (Click here to read more about this study). The Michael J Fox Foundation Fox Trial Finder website also has information on this study (Click here to read that page).
This is the first NLRP3 inhibitor clinical trial in Parkinson’s so it will be interesting to see what Roche finds in their data.
Are there other trials focused on the inflammasome coming?
Probably.
The inflammasome space is white hot at the moment. Before buying Inflazome, Roche actually purchased another biotech company called Jecure Therapeutics in late 2018 to get access to their portfolio of preclinical NLRP3 inhibitors aimed at various inflammatory conditions (Click here to read more about this).
 And then on the 1st April (no joke) 2019, the large pharmaceutical company Novartis acquired one clinical and two preclinical programs targeting the NLRP3 inflammasome from the biotech firm IFM Therapeutics.
And then on the 1st April (no joke) 2019, the large pharmaceutical company Novartis acquired one clinical and two preclinical programs targeting the NLRP3 inflammasome from the biotech firm IFM Therapeutics.
 The clinical program is for a drug called IFM-2427 which is a systemic antagonist of NLRP3 being targeted at an array of chronic inflammatory disorders (including gout, atherosclerosis and nonalcoholic steatohepatitis (NASH)). The two preclinical programs, however, include a gut-directed molecule for the treatment of inflammatory bowel disease but (more importantly) a brain-penetrant molecule which is being developed for targeting neurodegenerative conditions like Alzheimer’s and Parkinson’s (Source).
The clinical program is for a drug called IFM-2427 which is a systemic antagonist of NLRP3 being targeted at an array of chronic inflammatory disorders (including gout, atherosclerosis and nonalcoholic steatohepatitis (NASH)). The two preclinical programs, however, include a gut-directed molecule for the treatment of inflammatory bowel disease but (more importantly) a brain-penetrant molecule which is being developed for targeting neurodegenerative conditions like Alzheimer’s and Parkinson’s (Source).
Then at the end of September 2022, the diabetes focused pharma Novo Nordisk entered an exclusive development and licensing agreement with the Canadian biotech company Ventus Therapeutics potentially worth $700 million (Click here to read more about this).
In addition, another inflammation focused biotech company called NodThera recently announced completion of their Phase 1 clinical trialing of their two clinical stage NLRP3 inflammasome inhibitors, NT-0796 and NT-0249. Of interest here is that while NT-0249 is targeted towards peripheral inflammatory diseases, NT-0796 is brain-penetrant and “reduced inflammatory biomarkers supporting advancement in neuroinflammatory and peripheral inflammatory diseases” (Click here to read more about this).
For those interested, click here to read a recent update about the biotech efforts to develop NLRP3 inhibitors.
So what does it all mean?
The long wait for the new class of NLRP3 inhibitors to start being clinically tested is finally over, with the pharmaceutical company Roche initiating a Phase 1b trial in Parkinson’s this year. With support from the Parkinson’s community they will hopefully be able to complete this study quickly and move on to Phase 2 to establish if their agent (Selnoflast) is possibly impacting progression.
In addition, there are other biotech companies that have been developing brain-penetrant NLRP3 inhibitors to test whether reducing neuroinflammation can slow the progression of neurodegenerative conditions like Parkinson’s and Alzheimer’s. It is encouraging to see novel therapeutics being tested as it increases the number of shots on goal, raising the possibility that one of them will be successful.
We’ll be waiting this space closely.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, many of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced. This post has been produced for educational purposes only.
The banner for today’s post was sourced from Roche.





Very encouraging, because vitamin D and the VDR has been show to downregulate NLRP3 in several tissues and conditions. And I use 4000IU daily D3 to reduce inflammation. Fingers crossed !
LikeLiked by 1 person
Hi,
seems like riboflavin has also similar effect (https://www.nature.com/articles/s41598-020-76251-7#Sec2).
mh
LikeLiked by 1 person
The study showing that inhibiting the NLRP3 inflammasome also inhibits the spread of alpha-synuclein aggregates to other parts of the brain seems very significant. To me it suggests that the apoptosis of neurons that are afflicted with aggregated alpha-synuclein is releasing and spreading that misfolded protein.
Regarding NLRP3 inhibitors, it seems worth mentioning that while MCC950 is perhaps the most effective inhibitor of NLRP3, there are quite a number of others that have significant ability to inhibit NLRP3, and which are much more easily and inexpensively obtained. These include the traditional Chinese medicine oridonin, the feverfew derivative parthenolide, resveratrol, curcumin, omega 3 fatty acids, and quercetin.
I would also point out that NLRP3 is not the only receptor involved in activating innate immunity in Parkinson’s. The toll-like receptors TLR4 and TLR2 are also involved, and in fact they may even be involved in priming NLRP3 for its own activation. Several substances that are inhibitory for NLRP3 are also inhibitory for toll-like receptors, including curcumin, resveratrol, quercetin, and omega-3 fatty acids. It seems possible that by acting on both kinds of receptors they might be even more effective that a substance that acts upon only NLRP3.
Finally, I would note that the NLRP3 inflammasome is just one part of an auto-inflammatory feedback loop, and interventions to damp down that feedback process can occur at multiple points in that cycle besides just inhibition of receptors like NLRP3. For example, fortifying mitochondria can give neurons the energy they require to defend themselves against the effects of cytokines, and antioxidants can interfere with the oxidative aspects of apoptosis, thereby protecting cells from destruction. If fewer neurons die, then there will be fewer DAMPs triggering receptors like NLRP3 on microglia.
The tendency to look for silver bullets that work as monotherapies, and for those bullets to be proprietary to private companies, is clearly driven by profit, and that motive in this case may be leading to the overlooking of multimodal therapies–especially those involving inexpensive substances that nobody owns or controls.
LikeLike
I should add that not all of the substances I have mentioned have been proven to cross the blood-brain barrier, but curcumin, resveratrol and omega-3 fatty acids all have been shown to do so. Oh, and I forgot to mention melatonin, which inhibits NLRP3 and also can cross into the brain.
LikeLike
Another reason – amongst many- I supplement with ‘vitamin’D3 is to ensure the integrity of my aged BBB. My concern is carbidopa penetrating the bbb. It is supposed not to pass but I cannot find any papers showing that. But there is plenty of evidence for bbb deterioration with age. If carbidopa penetrates that raises the spectre of iatrogenic suppression of dopaminergic signalling. I am not holding my breath that the PD establishment will embrace that possibility with any enthusiasm, so I forge my own regime.
LikeLiked by 1 person
It had not occurred to me that BBB compromise could allow carbidopa (or benserazide) to cross into the brain, inhibiting dopamine formation. Mousing around, I found that the researcher Paul Harvey looked at this and suggests that the BBB could be breaking down selectively in areas most affected by neuroinflammation:
LikeLike
And one of the most important actions of D3 is anti-inflammatory, especially on microglia. recent review: https://www.mdpi.com/2072-6643/15/3/767
LikeLike
It had not occurred to me that breakdown of the BBB might allow carbidopa to enter the brain, sabotaging dopamine production. Mousing around a bit I found this article suggesting that the BBB may be selectively compromised in areas experiencing neuroinflammation, which for PD patients would typically include the nigra-striatal area where you would least want exogenous carbidopa floating in.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2761151/
This would seem to emphasize the importance of anti-inflammatories that cross the BBB, like Longvida curcumin, baicalin and EGCg.
LikeLike
Here is a little more information from Roche on Selnoflast
LikeLike
Hi how can why try-out this New medication?
LikeLike