
Addex Therapeutics and the Michael J Fox Foundation are preparing to initiate a new clinical trial testing a new drug called Dipraglurant on levodopa-induced dyskinesia (Source).
Dipraglurant is a mGluR5 negative allosteric modulator (don’t panic, it’s not as complicated as it sounds).
In today’s post, we’ll explain what all of that means and look at the science behind this new treatment.
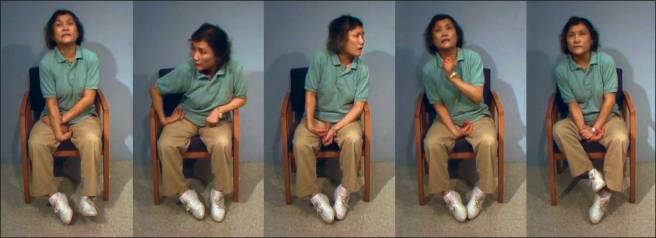
An example of a person with dyskinesia. Source: JAMA Neurology
For anyone familiar with Parkinson’s disease, they will know that long term use of the treatment L-dopa can lead to two possible outcomes:
- The treatment loses it’s impact, requiring ever higher doses to be administered
- The appearance of dykinesias
Now, not everyone taking L-dopa will be affected by both of these outcomes, but people with young, onset Parkinson’s disease do seem to be at risk of developing L-dopa induced dykinesias.
What are Dyskinesias?
Dyskinesias (from Greek: dys – abnormal; and kinēsis – motion, movement) are simply a category of movement disorders that are characterised by involuntary muscle movements. And they are certainly not specific to Parkinson’s disease.
As we have suggested above, they are associated in Parkinson’s disease with long-term use of L-dopa.
Below is a video of two legends: the late Tom Isaacs (who co-founded the Cure Parkinson’s Trust) and David Sangster (he founded www.1in20Parkinsons.org.uk). They were both diagnosed with Parkinson’s disease in their late 20’s. Tom, having lived with Parkinson’s for 20 years at the time of this video provides a good example of what dyskinesias look like:
As you can see, dyskinesias are a debilitating issue for anyone who suffers them.
How do dyskinesias develop in Parkinson’s disease?
Before being diagnosed and beginning a course of L-dopa, the locomotion parts of the brain in a person with Parkinson’s disease gradually becomes more and more inhibited. This increasing inhibition results in the slowness and difficulty in initiating movement that characterises this condition. A person with Parkinson’s may want to move, but they can’t.
They are akinetic (from Greek: a-, not, without; and kinēsis – motion).

Drawing of an akinetic individual with Parkinson’s disease, by Sir William Richard Gowers
Source: Wikipedia
L-dopa tablets provide the brain with the precursor to the chemical dopamine. Dopamine producing cells are lost in Parkinson’s disease, so replacing the missing dopamine is one way to treat the motor features of the condition. Simply giving people pills of dopamine is a non-starter: dopamine is unstable, breaks down too quickly, and (strangely) has a very hard time getting into the brain. L-dopa, on the other hand, is very robust and has no problem getting into the brain.

Sinemet is L-dopa. Source: Drugs
Once inside the brain, L-dopa is quickly converted into dopamine. It is changed into dopamine by an enzyme called DOPA decarboxylase, and this change rapidly increases the levels of dopamine in the brain, allowing the locomotion parts of the brain to function more normally.
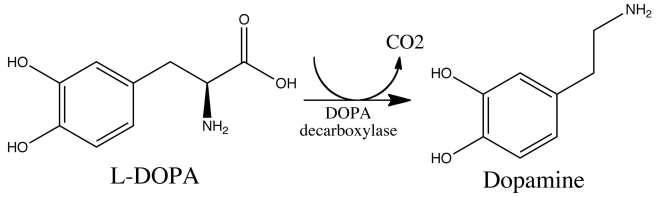
The chemical conversion of L-dopa to dopamine. Source: Nootrobox
In understanding this process, it is important to appreciate that when an L-dopa tablet is consumed and L-dopa enters the brain, there is a rapid increase in the levels of dopamine. A ‘spike’ in the supply of dopamine, if you will, and this will last for the next few hours, before the dopamine is used up.
As the effects of the L-dopa tablet wear off, another tablet will be required. This use of multiple L-dopa pills across the day gives rise to a wave-like shape to the dopamine levels in the brain over the course of the day (see the figure below). The first pill in the morning will quickly lift the levels of dopamine enough that the individual will no longer feel akinetic. This will allow them to be able to function with normal controlled movement for several hours before the L-dopa begins to wear off. As the L-dopa wears off, the dopamine levels in the brain drop back towards levels that will leave the person feeling akinetic and at this point another L-dopa tablet is required.

After several years of L-dopa use, many people with Parkinson’s disease will experience a weaker response to each tablet. They will also find that they have more time during which they will be unable to move (exhibiting akinesia). This is simply the result of the progression of Parkinson’s disease – L-dopa treats the motor features of the disease but only hides/masks the fact that the disease is still progressing.
To combat this shorter response time, the dose of L-dopa is increased. This will result in increasing levels of dopamine in the brain (as illustrated by the higher wave form over time in the image below). It will take more L-dopa medication induced dopamine to lift the individual out of the akinetic state.
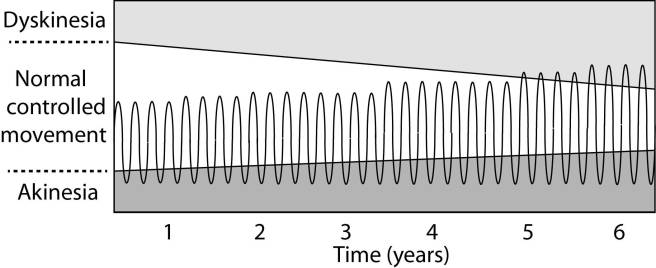
This increasing of L-dopa dosage, however, is often associated with the gradual development of abnormal involuntary movements that appear when the levels of L-dopa induced dopamine are the highest.
These are the dyskinesias.
Are there different types of dyskinesias?
Yes there are.
Dyskinesias have been broken down into many different subtypes, but the two main types of dyskinesia are:
Chorea – these are involuntary, irregular, purposeless, and unsustained movements. To an observer, Chorea will look like a very disorganised/uncoordinated attempt at dancing (hence the name, from the Greek word ‘χορεία’ which means ‘dance’). While the overall activity of the body can appear continuous, the individual movements are brief, infrequent and isolated. Chorea can cause problems with maintaining a sustained muscle contraction, which may result in affected people dropping things or even falling over.
Dystonia – these are sustained muscle contractions. They often occur at rest and can be either focal or generalized. Focal dystonias are involuntary contractions in a single body part, for example the upper facial area. Generalized dystonia, as the name suggests, are contraction affecting multiple body regions at the same time, typically the trunk, one or both legs, and another body part. The intensity of muscular movements in sufferers can fluctuate, and symptoms usually worsen during periods of fatigue or stress.
We have previously discussed the current treatment options for dyskinesias (click here to see that post).
Ok, so what clinical trials are Addex Therapeutics and the Michael J Fox Foundation preparing and why?
They are preparing to take a drug called Dipraglurant through phase III testing for L-dopa inducing dyskinesias in Parkinson’s disease. Dipraglurant is a mGluR5 negative allosteric modulator.
And yes, I know what you are going to ask next: what does any of that mean?
Ok, so mGluR5 (or Metabotropic glutamate receptor 5) is a G protein-coupled receptor. This is a structure that sits in the skin of a cell (the cell membrane), with one part exposed to the outside world – waiting for a chemical to bind to it – while another part is inside the cell, ready to act when the outside part is activated. The outside part of the structure is called the receptor.
Metabotropic receptors are a type of receptor that is indirectly linked with channels in cell membrane. These channels open and close, allowing specific elements to enter the cell. When a chemical (or agonist) binds to the receptor and it becomes activated, the part of the structure inside the cell will send a signal to the channel via a messenger (called a G-protein).
The chemical that binds to mGluR5 is the neurotransmitter glutamate.

Metabotropic glutamate receptor 5 activation. Source: Nature
But what about the “negative allosteric modulator” part of ‘mGluR5 negative allosteric modulator’
Good question.
This is the key part of this new approach. Allosteric modulators are a new class of orally available small molecule therapeutic agents. Traditionally, most marketed drugs bind directly to the same part of receptors that the body’s own natural occurring proteins attach to. But this means that those drugs are competing with those endogenous proteins, and this can limit the potential effect of the drug.
Allosteric modulators get around this problem by binding to a different parts of the receptor. And instead of simply turning on or off the receptor, allosteric modulators can either turn up the volume of the signal being sent by the receptor or decrease the signals. This means that when the body’s naturally occurring protein binds in the receptor, allosteric modulators can either amplify the effect or reduce it depending on which type of allosteric modulators is being administered.

How Allosteric modulators work. Source: Addrex Thereapeutics
There are two different types of allosteric modulators: positive and negative. And as the label suggests, positive allosteric modulators (or PAMs) increase the signal from the receptor while negative allosteric modulators (or NAMs) reduce the signal.
So Dipraglurant turns down the volume of the signal from the mGluR5 receptor?
Exactly.
By turning down the volume of the glutamate receptor mGluR5, researchers believe that we can reduce the severity of dyskinesias.
But hang on a second. Why are we looking at glutamate in dyskinesias? Isn’t dopamine the chemical of interest in Parkinson’s disease?
So almost 10 years ago, some researchers noticed something interesting in the brains of Parkinsonian monkeys that had developed dyskinesias:

Title: mGluR5 metabotropic glutamate receptors and dyskinesias in MPTP monkeys.
Authors: Samadi P, Grégoire L, Morissette M, Calon F, Hadj Tahar A, Dridi M, Belanger N, Meltzer LT, Bédard PJ, Di Paolo T.
Journal: Neurobiol Aging. 2008 Jul;29(7):1040-51.
PMID: 17353071
The researchers conducting this study induced Parkinson’s disease in monkeys using a neurotoxin called MPTP, and they then treated the monkeys with L-dopa until they began to develop dyskinesias. At this point when they looked in the brains of these monkeys, the researchers noticed a significant increase in the levels of mGluR5, which was associated with the dyskinesias. This finding led the researchers to speculate that reducing mGluR5 levels might reduce dyskinesias.
And it did!
Subsequent preclinical research indicated that targeting mGluR5 might be useful in treating dyskinesias, especially with negative allosteric modulators:

Title: The mGluR5 negative allosteric modulator dipraglurant reduces dyskinesia in the MPTP macaque model
Authors: Bezard E, Pioli EY, Li Q, Girard F, Mutel V, Keywood C, Tison F, Rascol O, Poli SM.
Journal: Mov Disord. 2014 Jul;29(8):1074-9.
PMID: 24865335
In this study, the researchers tested the efficacy of dipraglurant in Parkinsonian primates that had developed L-dopa induced dyskinesias. They tested three different doses of the drug (3, 10, and 30 mg/kg).
Dipraglurant significantly reduced dyskinesias in the monkeys, with best effect being reached using the 30 mg/kg dose. Importantly, the dipraglurant treatment had no impact on the efficacy of L-dopa which was still being used to treat the monkeys Parkinson’s features.
This research lead to a clinical trials in man, and last year Addex Therapeutics published the results of their phase IIa clinical trial of Dipraglurant (also called ADX-48621):

Title: A Phase 2A Trial of the Novel mGluR5-Negative Allosteric Modulator Dipraglurant for Levodopa-Induced Dyskinesia in Parkinson’s Disease.
Authors: Tison F, Keywood C, Wakefield M, Durif F, Corvol JC, Eggert K, Lew M, Isaacson S, Bezard E, Poli SM, Goetz CG, Trenkwalder C, Rascol O.
Journal: Mov Disord. 2016 Sep;31(9):1373-80.
PMID: 27214664
The Phase IIa double-blind, placebo-controlled, randomised trial was a dose escalation study, conducted in 76 patients with Parkinson’s disease L-dopa-induced dyskinesia – 52 subjects were given dipraglurant and 24 received a placebo treatment. The dose escalation assessment of dipraglurant started at 50 mg once daily to 100 mg 3 times daily. The study was conducted over 4 weeks.
The investigators found that dipraglurant significantly reduced the dyskinesias on both day 1 of the study and on day 14, and this treatment did not result in any worsening of the Parkinsonian features. And remember that this was a double blind study, so both the investigators and the participants had no idea which treatment was being given to each subject. Thus little bias can influence the outcome, indicating that dipraglurant really is having a beneficial effect on dyskinesias.
The company suggested that dipraglurant’s efficacy in reducing L-dopa-induced dyskinesia warrants further investigations in a larger number of patients. And this is what the company is now doing with the help of the Michael J. Fox Foundation (MJFF). In addition, dipraglurant’s potential benefits on dystonia are also going to be investigated with support from the Dystonia Medical Research Foundation (DMRF).
And the really encouraging aspect of this research is that Addex Therapeutics are not the only research group achieving significant beneficial results for dykinesias using this treatment approach (click here to read about other NAM-based clinical studies for dyskinesias).
Fingers crossed for more positive results here.
What happens next?
L-dopa induced dyskinesias can be one of the most debilitating aspects of living with Parkinson’s disease, particularly for the early-onset forms of the condition. A great deal of research is being conducted in order to alleviate these complications, and we are now starting to see positive clinical results starting to flow from that research.
These results are using new type of therapeutic drug that are designed to increase or decrease the level of a signal occurring in a cell without interfering with the normal functioning of the chemicals controlling the activation of that signal.
This is really impressive biology.
The banner for today’s post was sourced from Steam