Losing the sense of smell is a common feature associated with Parkinson’s disease. But this feature of the condition may help us to better understand the condition. Some autopsy studies have suggested that the olfactory system is one of the first structures in the brain to be affected by the disease.

Source: Guardian
How do we smell?
Bad.
That’s both a pathetic attempt at humour and a serious answer. Compared with fellow members of the mammalian family, human beings have a pretty poor sense of smell.
The process of smelling stuff is conducted through structures called the olfactory bulbs. The human olfactory bulbs lie on the base of our brains, protruding forward towards our nose (and nasal cavity).

A view of the human brain from below (olfactory bulbs are in yellow). Source: StudyBlue

A view of the human brain from in front (olfactory bulbs are in yellow). Source: StudyBlue
Inside your nose there is an area of smell sensitive cells that lies on the roof of the nasal cavity (about 7 cm behind your nostrils). That area is called the olfactory epithelium, and it plays a critical role in our sense of smell.
The size of the human olfactory epithelium is rather small and reflects our poor sense of smell, especially when compared, for example, to a dog (humans have about 10 cm2 (1.6 sq in) of olfactory epithelium, while some dogs have 170 cm2 (26 sq in)).

The human olfactory system. Source: Biology junction.com
When you inhale an odor (or odorant molecules) through your nose, there are tiny receptors (called olfactory receptors) on the olfactory epithelium that are the first step in detecting the smell. Every single olfactory receptor cell presents just one (and only one) type type of odorant receptor. When they detect that odor, the olfactory receptor cell reacts by sending an electrical signal along its branch (called an axon) to the olfactory bulbs in the brain.
As the axon of olfactory receptor cell enters the olfactory bulb it forms clusters with other olfactory receptor cell axons, and these clusters are called glomeruli. Inside the glomerulus (singular), the axons make contact the branches of a type of brain cell called a mitral cell. Mitral cells send their axons to many different areas of the brain, including the anterior olfactory nucleus, piriform cortex, the amygdaloid complex, the entorhinal cortex, and the olfactory tubercle.
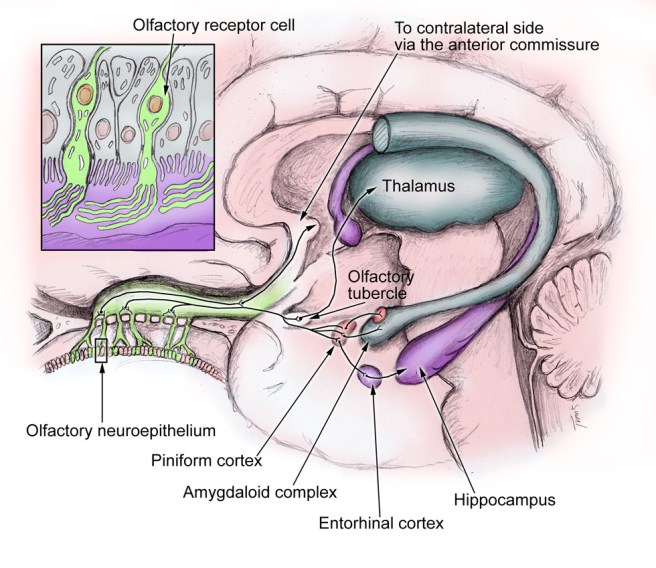
Source: YaleScientific
From here our understanding of olfactory processing is less well understood. The piriform cortex is considered the area most likely associated with identifying particular odor. The amygdala is involved in emotional and social functions (eg. mating and recognition), while the entorhinal cortex (and connected hippocampus) is associated with memory – this area is probably activated when a particular smell reminds us of something in our childhood.
What is known about our sense of smell in Parkinson’s disease?
In 1975, two researchers in Minnesota noticed that many of their people with Parkinson’s disease that they were assessing had reduced olfactory abilities. They decided to test this observation:

Title: Olfactory function in patients with Parkinson’s disease.
Authors: Ansari KA, Johnson A.
Journal: J Chronic Dis. 1975 Oct;28(9):493-7.
PMID: 1176578
The researchers took 22 people with Parkinson’s disease and 37 age/sex-matched controls and repeatedly tested them in a double blind study to determine their olfactory acuity. In each test, the subjects were given five test tubes. Two of the tubes in each set contained 0.5 ml of diluted amyl acetate (which has a distinct smell). The other three tubes contained just water. The subjects were asked to inhale through their nose and then identify which two tubes in each set contained the amyl acetate. The highest dilution (the weakest smelling solution) at which the subject could correctly identify the two amyl acetate containing tubes was designated as their olfactory threshold.
The researchers found that people with Parkinson’s disease had a significantly reduced olfactory acuity (a lower olfactory threshold than compared to control subjects). They also noted that subjects with more progressive forms of the disease exhibited a worse performance on the test. Numerous studies have now replicated this overall result, including a recent study that indicated that smoking may have a protective role on the olfactory ability (Click here and here for more on this).
EDITORIAL NOTE: Please understand that the loss of smell in Parkinson’s disease does not immediately mean that you will have a more progressive form of the condition. There is simply a trend in the data that suggests the loss of smell is a risk factor for having a more progressive version of the condition.
We would also like to discourage any thoughts of taking up smoking in order to protect your sense of smell.
So what is actually happening in the Parkinson’s disease brain?
This is Prof Heiko Braak:

He’s a dude. We’ve mentioned him before in a previous post.
Many years ago, he and his colleagues were intrigued with the hyposmia (reduction in olfactory ability) in Parkinson’s disease. They conducted a series of autopsy studies, looking at 413 brains! Specifically, they were looking for deposits of the Parkinson’s disease-related protein, alpha synuclein, in the brains and where the protein was accumulating. The accumulation of alpha synuclein is believed to be associated with the loss of cells in the brain.
In total they found 30 brains that exhibited accumulation of alpha synuclein. Of interest, they found that 16 of those brains had accumulation of alpha synuclein in the olfactory bulb. And in one particular case, the olfactory bulb was the only affected part of the brain, except for a tiny region of the brain stem.
The researchers were curious about the possibility that the olfactory system could be a potential starting point for Parkinson’s disease, but they were quick to point out that only half the cases they analysed (16/30) had accumulation of alpha synuclein in the olfactory bulb. Thus, while the olfactory system may be involved, it seems unlikely that the nose is the sole induction site of Parkinson’s disease.
After this study was published, however, Braak and his colleagues went on to analyse the accumulation of alpha synuclein in the lining of the gut and their results suggested this as another possible site of induction (we have written about this in a previous post). They have subsequently proposed a model of disease spread based on entry to the brain via the nose and gut:

The Braak stages of Parkinson’s disease. Source: Nature Reviews Neurology.
It is interesting to observe that studies by other scientists have indicated that the nasal epithelium of people with Parkinson’s disease (both with and without the loss of olfactory abilities) is not damaged or presenting an accumulation of alpha synuclein (Click here for more on this).
So what happens to the olfactory bulbs in Parkinson’s disease?
A recent review of the previous studies investigating olfactory bulb volume in people with Parkinson’s disease was published in the Open Access journal PlosOne:

Title: Changes in Olfactory Bulb Volume in Parkinson’s Disease: A Systematic Review and Meta-Analysis.
Authors: Li J, Gu CZ, Su JB, Zhu LH, Zhou Y, Huang HY, Liu CF.
Journal: PLoS One. 2016 Feb 22;11(2):e0149286.
PMID: 26900958 (this report is OPEN ACCESS if you would like to read it)
The authors of the study conducted a systematic review (or meta-analysis) of all of the previous studies (six in total) that have measured the size of the olfactory bulb in the brains of people with Parkinson’s disease (using brain imaging techniques). They found that in all of the 6 studies (collectively 216 PD patients and 175 healthy controls) there was a significant reduction in the size of the olfactory bulbs of people with Parkinson’s disease. Strangely, they authors also found the right olfactory bulb was larger than the left in subjects with Parkinson’s disease across all of the studies, and this effect was not found in the healthy controls.
The motor features of Parkinson’s disease usually begin asymmetrically – by this we mean that the left arm is affected before the right, or the right leg has tremor before the left. This is different for each person, as the disease has no particular preference for either side of the body. So why on earth is the right olfactory bulb more affected than the left?
There is your homework question for tonight!
I’ll expect your answers tomorrow.
 Dr Les Milne. Source: BBC
Dr Les Milne. Source: BBC Joy Milne. Source: Telegraph
Joy Milne. Source: Telegraph













