
Interest press release from the biotech company AFFiRiS last week (Click here for the press release) regarding their clinical trial of a vaccine for Parkinson’s disease. We have previously outlined the idea behind the trial (Click here for that post) and the team at Michael J Fox foundation also provide a great overview (Click here for that – MJF are partly funding the trial). In today’s post we will briefly review what results AFFiRiS has shared.

Vaccination. Source: WebMD
Vaccination represents an efficient way of boosting the immune system in the targeting of foreign or problematic agents in the body. For a long time it has been believed that the protein Alpha Synuclein is the ‘problematic agent’ involved in the spread of Parkinson’s disease inside the brain. Alpha synuclein is required inside brain cells for various normal functions. In Parkinson’s disease, however, this protein aggregates for some reason and forms circular clusters inside cells called Lewy bodies.
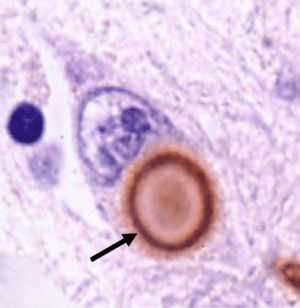
A lewy body (brown with a black arrow) inside a cell. Source: Cure Dementia
It has been hypothesized (and there is a lot of experimental evidence available to support the idea) that released alpha synuclein – freely floating between brain cells – may be one method by which Parkinson’s disease spread through the brain. With this in mind, groups of scientists (like those at AFFiRiS) are attempting to halt the spread of the condition, by training the immune system to target free-floating alpha synuclein. Vaccination is one method by which this is being attempted.
AFFiRiS is a small biotech company in Vienna (Austria) that has an ongoing clinical trial program for a vaccine (called ‘AFFITOPE® PD01A’) against alpha synuclein. The subjects in the study (22 people with Parkinson’s disease) received four vaccinations – each injection given four-weeks apart – and then the subjects were observed for 2-3 years (6 additional subjects were included in the study for comparative sake, but they did not receive the vaccine.
Last week the company issued a press release regarding a phase 1 trial (AFF008), which indicated that PD01A is safe and well tolerated, and causing an immune response (which is a good thing) in 19 of 22 (86%) of vaccinated subjects. In 12 of those 19 (63%) participants with and immune response, the researchers found alpha-synuclein antibodies in the blood, suggesting that the body was reacting to the injected vaccine and producing antibodies against alpha synuclein (for more on what antibodies are, click here).
The scientists also conducted some exploratory efficacy assessments – to determine if they could see if the vaccine was working clinically and slowing down the disease. Eight of the 19 (42%) subjects with an immune response, had no increase of their dopaminergic medication (eg. L-Dopa) over the course of the observational period (average three years per subject). And five of those eight subjects had stable clinical motor scores at the end of the study.
The company also conducted parallel laboratory-based experiments which indicate that AFFITOPE® PD01A-induced antibodies are binding to alpha-synuclein in various models of Parkinson’s disease.
The company will be presenting the results on a poster at the 4th World Parkinson Congress in Portland, Oregon, USA on September 21.
So this is a good result right?
It is easy to get excited by the results announced in the press release, but they must be taken with a grain of salt. This is a Phase I trial which is only designed to test the safety of a new therapeutic agent in humans. From this point of view: Yes, the study produced a good result – the vaccine was well tolerated by the trial subjects.
Drawing any other conclusions, however, is not really possible – the study was not double-blind and the assignment of subjects to the treatment groups was not randomize. In addition, the small sample size makes it very difficult to make any definitive conclusions. It must be noted that of the 22 people with Parkinson’s disease that started the study, only five exhibited stabilized clinical motor scores at the end of the study. It may be too soon to tell if the vaccine is having an effect in most of the people involved in the study. Thus longer observation periods are required – which the company is currently undertaking with their follow-up study, AFF008AA. The results of that study are expected in middle-late 2017.
We shall keep you posted.
The banner for today’s post was sourced from AFFiRiS








