|
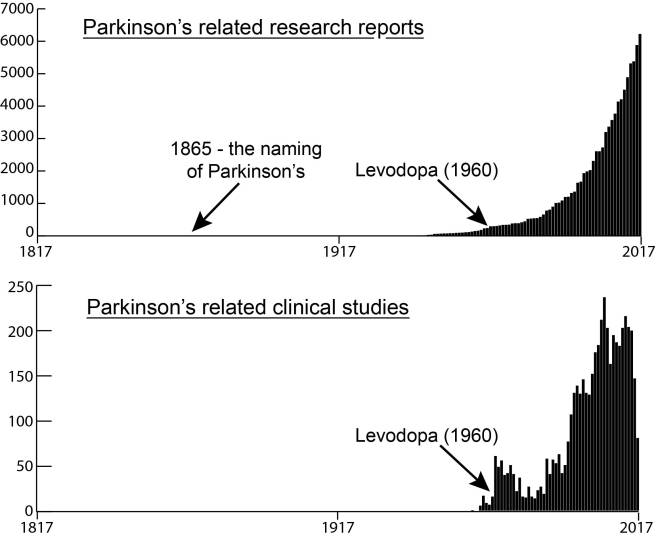
In 2019, researcher from around the world gathered for a special meeting in Toronto (Canada) to discuss/debate some of the most important issues slowing Parkinson’s research. Specifically, they asked if Parkinson’s is not a single condition, but rather a collection of diseases that look very similar, can current research efforts be revised to cater for this shift in definitions, or does Parkinson’s research need to be reconstructed? Recently a summary of the discussions at the meeting has been published. In today’s post, we will review that summary document, explore some of the topics discussed, and consider some of their proposed solutions. |
 Source: Medium
Source: Medium
Let’s start this post with a simple question: What are the first principles of Parkinson’s?
What do you mean “first principles”?
First principles are what philosophy and engineering types considered the most basic propositions (or assumptions) that cannot be deduced from any other propositions or assumptions.
Simply, they are the fundamental facts from which everything else stems.
Elon Musk likes to talk about first principles in all of his projects, and on all matters he suggests we should : “boil things down to their fundamental truths and say, ‘What are we sure is true?’… and then reason up from there, as opposed to reasoning by analogy”.
What is meant by ‘analogy’ here?
Analogy refers building knowledge and solving problems based on prior assumptions; using beliefs widely held and approved by a majority of people. It allows you to take your understanding of one domain and compare (or apply) it with another.
The example of analogy is that it is easier to teach students that electrons whizz around an atom’s nucleus the same way planets orbit the sun because they will have been exposed to this idea, than actually explaining the intricacies of nuclear physics…even though there are more than a few differences.
 Source: Blog44
Source: Blog44
Let’s return to the initial question though: What are the first principles of Parkinson’s?
I don’t know. Are you going to tell me?
Nope. I don’t know either.
Ok. Um. So is this going to be a really short SoPD post then?
No.
Recently Parkinson’s researchers around the world have been calling for efforts to establish the “first principles” of Parkinson’s (and Parkinson’s is not alone here – neurodegenerative research in general is going through a similar period of self reflection – click here to read more about this in Alzheimer’s).
A good example of this process is what happened in Toronto last April.
What happened in Toronto?