Before you read any further, I feel it only fair to warn the squeamish amongst you that todays post is going to deal with the topic of urine. I myself have a little ‘three-nager’ who is potty training at the moment, so I am rather intimately familiar with the stuff. But consider yourselves fair warned.
Warning out of the way, let’s begin:
What is urine?
Urine is a liquid excression from our body, consisting of water, salts and a substance called urea. It is made in the kidneys, temporarily stored in the bladder, and eventually released through the urethra. Pretty simple right.
On a good day approximately 90-95% of your urine will be water. Within the remaining 5%, however, there is a lot of solids that have been removed from the blood system by the kidneys. Those solids may be considered waste by our bodies, but they can tell us a lot about what is happening inside us.
Last week some researchers from the University of Alabama and Columbia University (NY) published a study that analysed some of those solids – looking at one enzyme in particular – being excreted in urine. They wanted to determine whether there were any differences between normal healthy individuals and people with Parkinson’s disease.
Their results are really interesting:

Title: Urinary LRRK2 phosphorylation predicts parkinsonian phenotypes in G2019S LRRK2 carriers.
Authors: Fraser KB, Moehle MS, Alcalay RN, West AB; LRRK2 Cohort Consortium.
Journal: Neurology. 2016 Feb 10.
PMID: 26865512
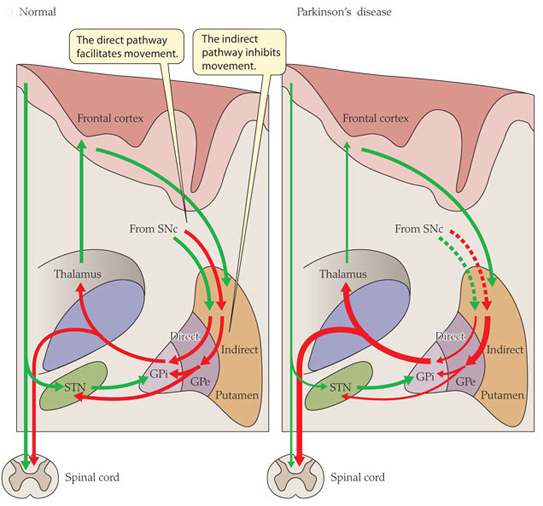
We have previously discussed Lrrk2 (and you can find that post here). It is a gene that is particularly interesting with regards to Parkinson’s disease because mutations in that gene are associated with susceptibility to the condition.
The Lrrk2 gene gives rise to an enzyme that has different functions in our cells. The researchers in the current study extracted the lrrk2 enzyme from the solid waste of urine and started analysing the “phosphorylation status of the enzyme”.
Ok, um,…what is Phosphorylation?
Phosphorylation is the process by which a phosphoryl group is added to a molecule.
And what is a phosphoryl group?!?
Oh, never you mind. Just remember that phosphorylation is basically the way in which many enzymes – like Lrrk2 – are turned on (and off when they are dephosphorylated). Through phosphorylation the function/activity of an enzyme is changed. They can go from dormant to active through this process. And this addition of the phosphoryl group to the molecule can occur at different places on that molecule, affecting the resulting activity in different ways.
So what did the researchers find?
The scientists found that people with Parkinson’s disease who also have a particular mutation in the Lrrk2 gene (that mutation is called p.G2019S) had almost 5 times more phosphorylation at a particular part of the Lrrk2 enzyme than normal healthy control subjects. Interestingly, those levels were also 4.5 times higher than those of people with PD, but who did not have the Lrrk2 mutation.
This means that the researchers have found a potential biomarker of the Lrrk2 mutation (independent of Parkinson’s disease itself). This finding could offers us a means of determining people with the Lrrk2 mutation – who may be susceptible to Parkinson’s disease – with a simple urine test.
But the researchers also noticed that among all of the study participants who have the Lrrk2 mutation,those who also had Parkinson’s disease had levels of phosphorylation twice as high as those who did not have Parkinson’s disease. Thus the overall results suggest that regardless of mutation status, higher levels of Lrrk2 phosphorylation are associated with a greater risk or the presence of Parkinson’s disease.
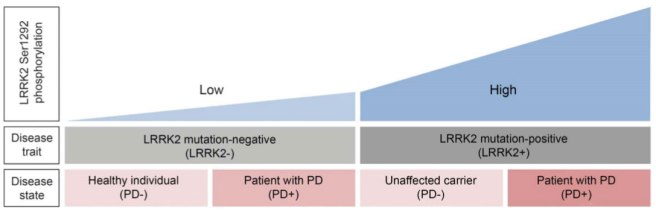
A diagram graph illustrating the findings of the Lrrk2 study. Source: Neurology
What does this mean?
Firstly, we need to point out that the study was conducted on a small population of men (two studies actually – the first had 14 subjects, and the second had 62 subjects). The results need to be independently replicated in larger groups (ideally also containing some female participants).
The results are very exciting, however, as they may point towards potential therapeutic pathways. It could also provide a means of monitoring clinical trials – a feature that the University of Alabama researchers are currently testing in another clinical trial. They are investigating if a LRRK2 inhibitor drug, called Sunitinib, results in lower leaves of phosphorylated Lrrk2 in the urine.
The research is also encouraging with regards to the search for biomarkers in Parkinson’s disease – a quest that has struggled somewhat until recently. Novel biomarkers provide useful tools in our fight against this terrible disease.




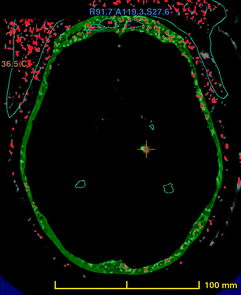 A brain scan image of the area being targeted (red cross). The skull is in green, and the cooled water unit is is red. Source:
A brain scan image of the area being targeted (red cross). The skull is in green, and the cooled water unit is is red. Source: