|
In my previous post, we briefly reviewed the results of the phase II double-blind, randomised clinical trial of Exenatide in Parkinson’s disease. The study indicates a statistically significant effect on motor symptom scores after being treated with the drug. Over the last few days, there have been many discussions about the results, what they mean for the Parkinson’s community, and where things go from here, which have led to further questions. In this post I would like to address several matters that have arisen which I did not discuss in the previous post, but that I believe are important. |

I found out about the Exenatide announcement – via whispers online – on the afternoon of the release. And it was in a mad rush when I got home that night that I wrote up the post explaining what Exenatide is. I published the post the following evening however because I could not access the research report from home (seriously guys, biggest finding in a long time and it’s not OPEN ACCESS?!?!?) and I had to wait until I got to work the next day to actually view the publication.
I was not really happy with the rushed effort though and decided to follow up that post. In addition, there has been A LOT of discussion about the results over the weekend and I thought it might be good to bring aspects of those different discussion together here. The individual topics are listed below, in no particular order of importance:
1. Size of the effect
There are two considerations here.
Firstly, there have been many comments about the actual size of the effect in the results of the study itself. When people have taken a deeper look at the findings, they have come back with questions regarding those findings.
And second, there have also been some comments about the size of the effect that this result has already had on the Parkinson’s community, which has been considerable (and possibly disproportionate to the actual result).
The size of the effect in the results
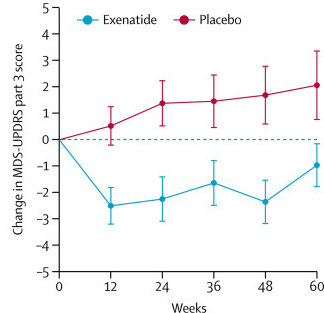
The results of the study suggested that Exenatide had a positive effect on the motor-related symptoms of Parkinson’s over the course of the 60 week trial. This is what the published report says, it is also what all of the media headlines have said, and it sounds really great right?
The main point folks keep raising, however, is that the actual size of the positive effect is limited to just the motor features of Parkinson’s disease. If one ignores the Unified Parkinson’s Disease Rating Scale (UPDRS) motor scores and focuses on the secondary measures, there isn’t much to talk about. In fact, there were no statistically significant differences in any of the secondary outcome measures. These included: