
|
In my previous post, we briefly reviewed the results of the phase II double-blind, randomised clinical trial of Exenatide in Parkinson’s disease. The study indicates a statistically significant effect on motor symptom scores after being treated with the drug. Over the last few days, there have been many discussions about the results, what they mean for the Parkinson’s community, and where things go from here, which have led to further questions. In this post I would like to address several matters that have arisen which I did not discuss in the previous post, but that I believe are important. |

I found out about the Exenatide announcement – via whispers online – on the afternoon of the release. And it was in a mad rush when I got home that night that I wrote up the post explaining what Exenatide is. I published the post the following evening however because I could not access the research report from home (seriously guys, biggest finding in a long time and it’s not OPEN ACCESS?!?!?) and I had to wait until I got to work the next day to actually view the publication.
I was not really happy with the rushed effort though and decided to follow up that post. In addition, there has been A LOT of discussion about the results over the weekend and I thought it might be good to bring aspects of those different discussion together here. The individual topics are listed below, in no particular order of importance:
1. Size of the effect
There are two considerations here.
Firstly, there have been many comments about the actual size of the effect in the results of the study itself. When people have taken a deeper look at the findings, they have come back with questions regarding those findings.
And second, there have also been some comments about the size of the effect that this result has already had on the Parkinson’s community, which has been considerable (and possibly disproportionate to the actual result).
The size of the effect in the results
The results of the study suggested that Exenatide had a positive effect on the motor-related symptoms of Parkinson’s over the course of the 60 week trial. This is what the published report says, it is also what all of the media headlines have said, and it sounds really great right?
The main point folks keep raising, however, is that the actual size of the positive effect is limited to just the motor features of Parkinson’s disease. If one ignores the Unified Parkinson’s Disease Rating Scale (UPDRS) motor scores and focuses on the secondary measures, there isn’t much to talk about. In fact, there were no statistically significant differences in any of the secondary outcome measures. These included:
- the experiences of daily living subparts of the MDS-UPDRS
- on–off patient diaries
- timed motor tests, non-motor symptom scale
- Mattis dementia rating scales
- quality-of-life scales

The investigators openly acknowledge this in the report. But in defence of this comment, one should point out that A.) the motor section of the UPDRS is not a singular component – it is made up 18 individual elements (and then some), dealing with features such as rigidity and tremor. So the result is not limited to just an individual result. And B.) it should be noted that the direction of effect in the many of those secondary measures was favouring the use of Exenatide. Perhaps in a larger study with more participants and conducted over a longer period of time, some of those measures might have become significant.
Another comment that has repeatedly popped up focuses on the differences between the two groups of participants. The participants in the Exenatide group were older than the placebo group (average 61.6 yrs verses 57.8 yrs), and they had higher UPDRS scores (meaning that their Parkinson’s disease was more severe). The age issue is arguably an intrinsic flaw of the randomised system used in phase II clinical trials which can not be helped. By shear chance subjects are distributed to one group or the other, and we have to live with the fact that more older participants were allocated to the Exenatide group. Whether this would have any actual impact on the results is debatable, particularly as the disease duration (the time since diagnosis) was exactly the same for both groups (6.4 years vs 6.4 years).
The higher starting UPDRS score may be partly due to the fact that 2 of the 29 participants being more advanced (H&Y stage 2-5) compared to all 29 of the placebo group having mild Parkinson’s disease (H&Y stage 1-2). But on average, yes the Exenatide group were more advanced in the disease course than the placebo group. In panel A of the image below, you can see that the Exenatide group (blue line) begins at 0 weeks with a UPDRS score of 32.8, and this is much higher than the 27.1 score of the placebo group (red line).
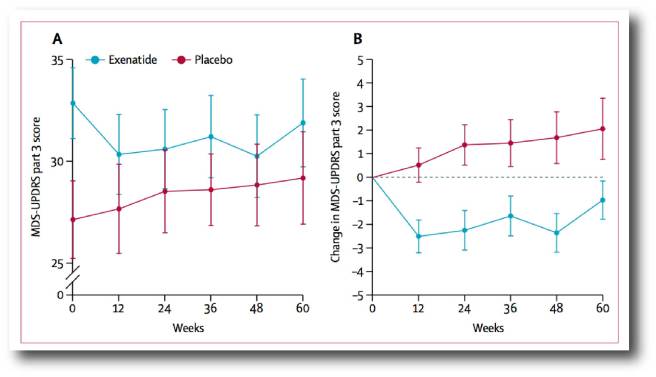
Motor scores in Exenatide study. Source: Lancet
Under normal circumstances the blue line in panel A. should have run in a parallel fashion to that of the red line, gradually climbing higher. But it didn’t. And this is particularly apparent when you look at the actual change in the UPDRS scores in panel B, there is a noticeable drop in the Exenatide group which remains relatively stable across the 60 week period of the study, while the placebo group continue to worsen.
Then there is also the comments about what Exenatide is actually doing.
A plateauing of the UPDRS motor features in the Exenatide group could indicate a disease-modifying effect….. OR it could simply indicate that the dopamine system is being affected by the drug (remember, only the motor component is showing a significant effect – similar to what one may see with Levodopa treatment).
Exenatide is known to influence various aspects of dopamine processing. In particular, it may be having an effect on a protein called dopamine transporter (or DAT), which helps to recycle dopamine back into the neuron after it has been released. GLP-1 receptor stimulation is known to affect DAT activity in rodents:

Title: Glucagon-like peptide 1 receptor activation regulates cocaine actions and dopamine homeostasis in the lateral septum by decreasing arachidonic acid levels.
Authors: Reddy IA, Pino JA, Weikop P, Osses N, Sørensen G, Bering T, Valle C, Bluett RJ, Erreger K, Wortwein G, Reyes JG, Graham D, Stanwood GD, Hackett TA, Patel S, Fink-Jensen A, Torres GE, Galli A.
Journal: Transl Psychiatry. 2016 May 17;6:e809.
PMID: 27187231 (This article is OPEN ACCESS if you would like to read it)
These researchers found that Exenatide (a GLP-1 agonist) inhibits the activity of the drug cocaine. Cocaine acts by targeting the DAT – it blocks the activity of DAT, which results in increased levels of dopamine floating around outside the neuron. And this result is supported by similar results from other researchers – Click here to read more.
Thus, it is possible that Exenatide could be causing changes in the activity of DAT which results in more dopamine floating around outside the neurons in the treated group. Such an increase in dopamine could explain some of the motor results in the study (and the reduced rates of decline in the brain imaging).
It is difficult to see though, how this could explain the results of the 12 week washout period. At the end of the study, the participants had their Exenatide and placebo treatments removed and 12 weeks later they were assessed again, and the Exenatide treated group were still found to have better performance on the UPDRS motor scores.
The investigators tried to manage the effect of the dopamine-based medication. But another difference between the two groups in the study was that the total dose of Levodopa was lower in the Exenatide group at the start of the study compared to the placebo group (774mg vs 825mg, respectively). In addition, subjects in the Exenatide group had greater increases in their dopaminergic therapy over the course of the trial, than those in the placebo group (changes in current medication were allowed throughout the trial to minimise drop out from either group). So if the Exenatide group were increasing their Levodopa treatment over the course of the study and Exenatide was affecting the dopamine system, then perhaps the findings of the study could be explained by dopamine-based mechanisms.
It is important to remember here, however, that the clinical assessments were all made during the off-medication state. In the report, off-medication was defined in the methods section of the report as “a period of withdrawal of levodopa for at least 8 h (ie, overnight) or 36 h in the case of long-acting drugs such as ropinirole, pramipexole, rasagiline, and rotigotine”. So it is difficult to see how Exenatide could be having an affect on the dopamine system.
But then again, how can we tell? This study did not give us an answer to this issue.
I hope that what is clear from all of these discussions is that while we have a positive effect, we do not understand how that positive effect has arisen.
We are not clear on whether Exenatide is having a neuroprotective effect, or simply stimulating a dopamine-based positive motor outcome. And this is why the scientific research community are saying that it is too early to be getting too excited by the result and making judgements about the use of Exenatide in Parkinson’s disease.
To be fair, determining how a drug works is not really the role of the phase II trial. The second round of clinical trials is supposed to be a randomised, blind assessment of whether a drug has having any effect in a small sample of the population of interest. Larger, more long-term phase III investigation of a drug candidate are supposed to explore what the effect actually is.
Some of the investigators involved in the study have already published an OPEN ACCESS commentary in the Journal of Parkinson’s disease, which is well worth reading if you are interested in these issues.

But one of the take home messages of this post is: there is a lot about this result that we simply do not understand and that requires further investigation.
And given this position, we must now discuss:
The size of the effect on the community
Until 23:30 on the 3rd August 2017, there was always hope but probably a great deal of scepticism that anything truly novel treatment-wise was just around the corner for the Parkinson’s community. After a long string of failed clinical trials, the community were not exactly holding their breath.
But to quote Dinah Washington, “What a difference a day makes”.
With the headlines in the morning and over subsequent days, there has come a stunned disbelief that something could be just around the corner. And that was followed over the weekend by a sudden change in mind-set that progress is possible and is being made, rising to a sense of maybe there is more around the next corner as well! On some of the online forums, there is a cautious but genuinely excited feeling that a disease modifying treatment could be near.
While I sincerely do not want to dampen any of that positive vibe, I am concerned that the reaction may now have gone beyond the actual findings of the study. In effect, making the news more than it is.
I think that the response has been a perfectly natural and human reaction for a community that has been so long starved of positive news. And it has been further encouraged by mass media headlines, such as this one. But one does worry that the expectations are currently going too far. Remember, the drug has only indicated a significant effect on the motor features of Parkinson’s disease.
2. The inevitable rush
In response to all the requests for advice and information regarding Exenatide over the weekend, I have given the same answer:
“I am not a clinician (just a research scientist) so I can not advise you on changes to your treatment regime – you should discuss any such changes with your doctor. In addition, we currently don’t know enough about this drug in Parkinson’s disease so I would suggest a ‘wait till we know more’ policy”.
There is simply too much that we do not know, so much so that official health regulators would have a very difficult time providing favourable advice or recommendations.
Having said all of that, I fully appreciate that my words are not going to stop folks who are desperate for anything to treat their condition from rushing out to get their hands on this drug. There is terrible a moral dilemma in asking people with a progressive condition to wait before taking something that could potentially slow down their condition.
But there is a slight issue with getting ones hands on this drug:
A 1 kit (containing 4 pens; with 2mg per pen) of Bydureon currently costs about US$500-700. And based on the results of the study, one would require 1 kit per month.

Bydureon pen containing Exenatide. Source: Businesswire
One of the many things we do not know, is exactly how much Exenatide is needed to have a positive effect on motor features. The clinical trials have been limited to testing at doses used in the treatment of diabetes (where we know the drug is relatively safe). Thus, it could be possible that only a small amount of the drug is actually necessary, but it will still be a costly treatment to use.
Generic brands are one solution to expensive drugs, but this is only possible when ‘brand-name’ drugs come ‘off-patent’. Drug companies currently have 20 years to develop and sell a brand-name drug after they have patented it for use in a particular disease. Once that period of time is up, the drug is considered off-patent and other companies can begin making generic copies of that drug. This usually brings the price down as market forces take hold.
Currently there are no generic versions of Exenatide, but a generic version of Byetta will be available from October 2017. Please note that Byetta is a different formulation of Exenatide to that used in the current study. As opposed to Bydureon (which is a slow-release, once-per-week version of Exenatide), Byetta treatment involves taking the drug twice a day. Byetta was used in the phase I clinical trial for Parkinson’s disease (Click here to read more about that trial).

Byetta pen containing Exenatide. Source: Drugs
But there is a serious word of caution with regards to using generic drugs:
About 90% of the prescriptions given out in the United States are for generic drugs. They are very widely used. But these drugs do come with an unsettling hidden risk. According to the Food and Drug Administration (FDA), only makers of brand-name drugs have a legal duty to update their warning labels if they suddenly learn of a new risk or side effect. FDA rules actually forbid generic makers from acting on their own in updating their warning labels (this strange measure is to limit the number of lawsuits). And a rethink of this policy has repeatedly been postponed – most recently last week (Click here to read more). The point is: when generics come to market, the brand-name drugs often disappear completely. This departure leaves consumers extremely vulnerable as there will be no warning labels if a new side effect develops. And this could be particularly dangerous in the case of repurposed drugs.
(I am not clear on UK or EU standards regarding this aspect of generic drugs)
So again, I must emphasise caution: we don’t know the long term impact of using this medication on Parkinson’s disease. Of particular concern to me is the weight loss issue that was observed in the Exenatide groups in both clinical trials. Weight loss is already an issue for many people with Parkinson’s disease, thus taking a drug that encourages further weight loss should not be approached lightly.
For those who are interested in exploring the use of Exenatide-like drugs for Parkinson’s disease, the better approach is take the drugs under controlled conditions, such as clinical trials. And there are new clinical trials for other GLP-1 agonists that are currently recruiting. The Cure Parkinson’s Trust is starting a phase II safety and efficacy trial of Liraglutide in people with Parkinson’s disease who are also insulin-resistant at the Cedars-Sinai Medical Center in California (Click here for more information), and the Cure Parkinson’s Trust is also initiating a 21 centres clinical study across France investigating Lixisenatide in people with early stage Parkinson’s disease.
Like Exenatide, both Liraglutide and Lixisenatide are glucagon-like-peptide-1 (GLP-1) agonists which are approved for treatment of Type 2 diabetes.
But understand that the inevitable rush for this new “potentially disease-modifying” drug will have consequences on future drug development efforts for the Parkinson’s disease community. This might sound weird, but the identification of drugs that truly impact the disease course actually creates a new basket of problems for the Parkinson’s research community.
3. The dilemma of success
Once you have a drug that truly demonstrates beneficial neuroprotective effects several events will take place in rapid succession. First, it will be acknowledged as an amazing step forward, then there is the back-slapping of the researchers, and finally everyone in the affected community will naturally want to get their hands on it ASAP.
A problem arises, however, when the researchers get back to work and want to test the next generation of potentially neuroprotective drug. The first neuroprotective drug will need to be considered an ‘exclusionary’ (or a factor that would exclude someone) in any future trials because it could potentially affect the outcome of the final results. And this is obviously going to hinder future future clinical trials. The situation starts to become even more complicated as we move ahead and additional subsequent potentially neuroprotective drug candidates become available.

Dilemma. Source: Huffpost
One suggested solution was to simply ‘move the goal posts‘ or change the aim of the clinical trial. Simply accept that everyone will be using the drug and continue on with the next trials regardless. The problem is that a truly neuroprotective drug will hide the effect of the new drug if it is also neuroprotective. Thus this ‘solution’ is a non-starter.
Another suggested solution is to test all new drugs in newly diagnosed, drug naive people. But this too creates a lot of issues. For example, many of those people are not necessarily going to be psychologically ready to partake in an experimental drug trial, especially if there is a neuroprotective option already available. And this ‘solution’ would severely limit the number of potential participants for all the clinical trials that are currently being planned.
Are you starting to see the dilemma?
Isn’t it curious though, how we weren’t even thinking about all of this way back before the 3rd August.
My goodness, what a difference a day makes.
4. Tom Isaacs
One person who was not here for that day, however, was Tom Isaacs.
A heart warming aspect of the Exenatide study, however, is that Tom found out about the result shortly before he passed away earlier this year. Tom was a co-founder of the Cure Parkinson’s Trust here in the UK, and there have been few individuals within the global Parkinson’s community who could match him for energy, enthusiasm and dedication to finding a cure for this condition. The Cure Parkinson’s Trust were one of the main driving forces behind the first Exenatide study and Tom attended a closed-door conference in late May of this year when the results of the phase II trial were first presented and discussed.

Tom Isaacs. Source: MJFF
I would also take this opportunity to mention that there will be a memorial in honour of Tom on the 12th September at the Merchant Taylors’ Hall in London (Click here for more information). I will be there.
5. Phase III?
Finally, another comment on what happens next.
It is my understanding that there are discussions with regulators for a phase III trial of Exenatide in Parkinson’s disease. It is the natural next step, but it is not absolutely required.
The current situation for FDA approval of new medications is changing with the adoption of the Obama Administration’s “21st Century Cures Act“. While the requirement for a phase III clinical trial has not always been strictly held (Click here for an example of ‘phase III skipping’), the rules appear to be loosening further, with suggestions of compressing the phase II and phase III clinical trials into “one big, adaptive design” (Source).
There is hope in some corners that Exenatide could skip a large (1-300 participants), costly (think of a really big number) phase III trial, by conducting several carefully designed smaller phase II trials. This is a possibility given that the drug is considered safe in another indication (diabetes), and is being re-purposed for Parkinson’s disease. My understanding is that this option is currently being looked at.
Summing up
There have been a lot of interesting discussions over the weekend following the release of the results of the phase II clinical trial of Exenatide in Parkinson’s disease last week. New mindsets and novel dilemmas. Fascinating stuff.
The Exenatide result has put me in a tricky position. I am associated and friendly with quite a few folks with Parkinson’s disease, and while I am desperate to help them, my head tells me that we must always be cautious when wandering into uncharted waters. And that is precisely where we are now. While there is suddenly the possibility of a disease modifying drug, we must proceed with caution.
I dare say that things are going to happen pretty quickly now. There is going to be a great deal of basic research results coming soon that will try to explain what Exenatide and the other GLP-1 agonists are doing in Parkinson’s disease. And there certainly will be a hive of activity on the clinical side of things.
Watch this space.
Oh and one more thing: I would like to share one amusing anecdote from the day of the release of the Exenatide results: I belong to a Facebook group that discusses Parkinson’s disease research and by sheer co-incidence on the afternoon before the release, a member asked when the results would be released. At the time I only knew that the manuscript of the results was going through the peer-review process, but I added that it “should be published soon”.
Later that evening, as I was eagerly awaiting the release, another member made the statement “When you say soon about a publication going through peer review, don’t you mean several months”, and I could not help but reply with a cheeky answer:
And it got two ‘likes’!
With all the excitement of the moment, I hope I can be forgiven by the person in question.
EDITORIAL NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. Please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Startupregister
Dear Simon
Thank you so much for your commitment to helping us understand the issues.
There appears to be a clear benefit of being in the Exenatide group in the first 12 weeks. But is this followed by (A) a plateau and a rapid decline or (B) a progressive decline that roughly parallels the control group? It makes more sense that benefits are maintained whilst taking the drug (option A). But, the rate of decline over the last 12 weeks wash out period is much greater than for the control group. So, could this suggest the start of a nose dive back towards the curve they would have followed if they hadn’t been treated? If it is, would this mean it is not neuroprotective? It’s a pity there aren’t more data points, particularly to extend the wash out period.
I have to say that I will require more information and/or the reassurance of close supervision before I consider taking Exenatide. I’ve had enough trouble with established Parkinson’s medication and I’m particularly concerned about the weight loss seen on the Exenatide trial (is this an early effect that stabilises?). However, I can appreciate the desperation of many people with Parkinson’s. I just hope they consider doing some self-monitoring that they document – it will produce data that they can show to their neurologist and may benefit the community (I have found this invaluable for quantifying my own problems with established meds and would suggest it is essential if risking going it alone on a new drug). Perhaps some academically coordinated analysis may become inevitable at some point. We are moving into uncharted territory – lets think about how we chart it.
LikeLike
Hi Jim,
Thanks for your comments – much appreciated and adding to an interesting discussion regarding the results of the Exenatide study. It is impossible to answer many of your questions based on the current results, but I will ask this: What were we expecting? A return to full health? No, too many dopamine cells have been lost by 6 years post diagnosis, and one drug is unlikely to have such an amazing impact as to correct this. A plateauing of motor features, however, suggests that Exenatide is doing something. Whether it is dopamine-based or neuroprotective is to be determined, but it does look encouraging. The weight-loss issue is a concern for me as well and I hope that any folks jumping on the band wagon will keep a watchful eye on that.
Your comments on more data points and more tracking are wonderful to hear. I really do hope there will be an effort in future clinical trials to start to incorporate more objection and continuous tracking technologies (eg. FitBit or Apple Watch sort of data). Devices that can look at things like sleep quality as well as the usual motor symptoms stuff. UPDRS once every few months is starting to feel a bit primitive. I might write a post about this (thanks for the idea!).
Kind regards,
Simon
LikeLike
Simon,
Thank you so much for another very informative and even handed post. There are very few people trying to paint the full picture for us.
A few comments.
Exenatide has loads of cell model/rat data showing its protective. Agree that the models don’t necessarily translate to humans, but do we need to remind the community of this? This is not the nilotonib trial… handing a few pills out, taking videos and throwing high fives to each other in front of the news cameras… the UCL team and the rest of the GLP 1 gang seem to be taking their time. This data seems to be another stone in the foundation building steadily. Not a boulder and certainly not a pebble.
There are millions of people taking GLP-1’s for diabetes. In this population PD should actually be over represented (assuming the GLP-1s aren’t forestalling onset) given the diabetes PD link. So there should be thousands of people with PD taking GLP-1s. Where is the data on these people? Where are negative PD related effects? One would think we would already know the negative impacts given the large overlapping populations. I don’t fully understand the focus on the potential negatives of these treatments. Wouldn’t we already know?
Re cost. Here in the US costs very much depend on your insurance plan. In my case Generic Rasagaline is $350/month which potentially maybe might be protective if I take 1 mg (but not if I take 2 mg). On the other hand, Bydureon would be $30 per month with my plan. (Insert fat diabetic American discount joke here) I need to ask myself what gives me more potential protection per dollar… Even at $700 per month we should also consider the back end costs. How much care could be saved if exenatide allows someone to work for another year or keep them from needing in home care for a year? It is a lot to spend if it’s useless.
Symptomatic relief. It is interesting that the UDPRS numbers in the exenatide group are relatively flat over time while on the drug. I would think that if we were just seeing symptom relief that you’d need to increase dose at week 48 as the PD progressed to get the same score as week 12.
Thanks for the great posts. Please keep them coming.
LikeLike
Hi Don,
Thanks for your well considered comments.
Regarding the UCL team’s approach, I agree. And I think their communication since the publication of the results has been prudent and well handled (particularly the Journal of Parkinson’s disease commentary – http://content.iospress.com/articles/journal-of-parkinsons-disease/jpd171192). For all the faults of the study, they are being very open about things. I think the next step for them will be to follow this up effort by keeping the community aware of what is going to happen next. And the ‘foundations’ will hopefully grow with the new GLP-1 agonist trials in California and France.
Regarding the millions of people taking GLP-1 agonists, there is evidence of these diabetic drugs are impacting PD already available, but I will leave that for a post that is coming very shortly 🙂
And on the comment about not needing to increase the dose of Exenatide at 48 weeks of the study, that is a really interesting thought! But remember that the participants were allowed to increase their normal dopamine-based medication and that the Exenatide group showed the higher increase in this domain. You are right though, that if the disease was simply slowed by Exenatide then one would expect to see a gradual worsening in the treatment group towards the end of the 48 week part of the study. Intriguing.
Thanks again for your thoughts.
Kind regards,
Simon
LikeLike
Thanks Simon, I look forward to your upcoming posts!
Thanks also for posting the team’s commentary. That was well handled indeed and I first saw it here. 👍 It’s nice to see when researchers try view the issue it from the patient perspective. I think they do in their commentary.
LikeLike
Just signing up for comments
LikeLike
Hi Simon !!
And now ?? Some new news about Exenatide ??
Greetings from Switzerland !
LikeLike
Hi Jan,
Thanks for your comment. I haven’t heard anything official, but I know that there are ongoing attempts to get a large Phase III trial of Exenatide funded. In addition, there are the other GLP-1 agonists clinical trials for Parkinson’s ongoing (Liraglutide in the US – https://clinicaltrials.gov/ct2/show/NCT02953665 – and Lixisenatide in France – https://clinicaltrials.gov/ct2/show/NCT03439943). I put up a post as soon as we hear anything more about the Exenatide plans.
Kind regards,
Simon
LikeLike
Hi Simon,
Has there been any further updates on the phase 3 trial?
LikeLike