|
The clinical testing new compounds is horrifically slow. There is simply no easy way to word it. From lab bench to regulator approval, we are currently looking at a process which will take at least a decade. The repurposing of clinically available treatments has shortened this process, but there are a limited number of drugs that can be repurposed, and the periods of time between clinical trials is still too long. Acknowledging this situation, we can do one of two things: Accept the circumstances and carry on doing things the way we have always done it (hoping that it will be different next time – a la Einstein’s definition of insanity),… OR we can try to change it. In today’s post, we will discuss an interesting project that is seeking to do the latter.
|
The guy at the podium (and in the upper left inset) is Barry Chandler.
A few months ago, Barry came to me and asked “What can I do to help?”
And I replied by asking “What do you do?”
Two things you need to know about Barry:
- He was diagnosed with young onset Parkinson’s 6 years ago, and
- He is a very well connected guy.
VERY well connected!
 I am the green string. Barry is everything else. Source: Philiphemme
I am the green string. Barry is everything else. Source: Philiphemme
By day, Barry works in the city of London as a DevOps practitioner (that was a new one for me too – “a combination of cultural philosophies, practices, and tools that increases an organization’s ability to deliver applications and services at high velocity“). But in the evenings and on weekends, Barry is an events co-ordinator.
And these two worlds merge nicely in the form of a meetup group that Barry runs, called “SEAM”.
What is SEAM?
The Study of Enterprise Agility Meetup is a group of 1000+ members who get together one evening per month to listen to speakers and then brainstorm/network ideas as to how systems and processes can be improved.
Sounds like a hoot right?
But seriously, think about it: A bunch of really intelligent people from a variety of backgrounds/occupation gathered in one spot seeking to learn something new, solve problems, and make things better.
No vested interests – beyond wanting to eat pizza, drink some beer, and propose some potential solutions.
I couldn’t find a networking photo with pizza, so…. Source: IA
And this is the point in the story where Barry and I started talking about one of my pet peeves: the clinical trial process.
This is a topic I have previously ranted and raved about at length (Click here for one example), and I will not repeat myself here – today’s post is all about solutions.
Our discussion quickly migrated from a friendly chat over a snack to a meeting room with a white board (and post-it notes… lots of post-it notes). Barry wanted to take me through a process of identifying areas where improvements could be made.
We started by mapping out the current situation regarding the clinical trial process.
What is the clinical trial process?
Good question.
There are 5 Phases involved with getting a new treatment approved for clinical use and they apply to almost all medical conditions:
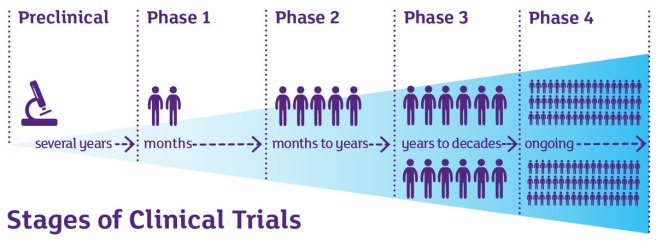 Source: Cancer
Source: Cancer
To begin with, there is the long, labourious process of discovering and testing a new treatment/compound. This is the preclinical phase. Technology (like CRISPR – click here to read more about that) is helping to speed up that part of this journey, but it still takes a long time and there is a lot of sweat and luck involved with it.
Once a compound/treatment has been identified, validated in various preclinical models of a medical condition, and a basic phamacological profile has been established in animals (this involves determining if the treatment is toxic and investigating how long the treatment lasts in the body of a lab animal), then an application to health regulators will be made to start testing the treatment in humans.
This first test is called a Phase I clinical trial.
 Source: Closerlookatstemcells
Source: Closerlookatstemcells
The goal of Phase I trials is to determine if the drug is safe. Efforts will also be made to assess the ideal dose for further clinical testing. Phase I trials will often be in healthy individuals (perhaps 20-50 people), involve a single or multiple doses of the treatment, and they are usually very quick (think weeks or a few months).
If the treatment is found to be safe in Phase I, the investigative team will shift their efforts to a Phase II trial.
 Source: Closerlookatstemcells
Source: Closerlookatstemcells
The goal of a Phase II clinical trial is to determine if the drug is safe in your patient population of interest and attempt to provide proof of efficacy (that is, try to provide evidence that the treatment is doing what it is supposed to). Phase II trials usually involve 50 – 100 individuals being on the treatment for a long period of time (they can last up to 12 months).
If the treatment is found to be safe and have an effect in Phase II clinical trials (an example in Parkinson’s of this is the Exenatide study – Click here and here to read more about this), the investigative team will then set up for a large Phase III clinical trial.
 Source: Closerlookatstemcells
Source: Closerlookatstemcells
The goal of a Phase III trial is to determine efficacy in a large group of the patient population of interest (200-300+ participants). These are huge, expensive trials that will last 1-2 years on average in the case of a slowly progressing condition like Parkinson’s.
And this is one of the main reasons why the clinical trial process is so slow for Parkinson’s: it is a slow progressive condition.
(There is also a Phase IV clinical trial which comes after a drug is approved by regulators. This involves companies/clinicians/researchers continually following the use of the treatment and collecting information in the clinical setting. This is outside of the scope of our interest here as it has nothing to do with getting treatments to the clinic).
That all sounds straightforward though. So why does the clinical trial process take so long?
Because around the active parts of the clinical testing process (that is the Phase I, II & III testing parts), there are long periods of setting up and organising clinical trials. And while the actual clinical testing of a treatment may be slow, one area where improvements could definitely be made is in the inbetween periods.
And this is where Barry wanted to initally focus our attention.
So we mapped it all out.
On a very basic level, the process between the end of a Phase II clinical trial, conducting a Phase III clinical trial and then getting a regulator’s approval a treatment for clinical use looks something like this:
This is the flow chart of process that Barry (and his partner in crime James Enoch) mapped out.
If a treatment is found to have an effect in Phase II, trial organisers will write up a plan (or protocol) for a Phase III clinical trial. They will then apply for funding to conduct the clinical trial, and this process can take between 6-12 months. That period of time takes into account matters like peer-review process, committee meeting dates, etc. Once the funding is secured, the organisers can apply for regulator approval to conduct the actual trial (approximately another 6 months, again with peer-review process, etc).
When the green light is finally given from the regulator, the study organisers can start recruiting participants for their study. For a large Phase III study, it can take 12 – 18 months to recruit 200-300 people in the study. These trials are so large that it is impossible to start everyone at the same time (there simply isn’t the infrastructure or staff to assess and start dosing everyone at the same time). Thus, the participants will begin the study 4-5 at a time.
And even with multiple research centres, it is still a slow process as most centres have clinic days only once or twice per week, with 2-4 people being assessed each of those days. As a result, recruiting and clinical testing will initially be conducted in parallel.
The actual testing period of the clinical trial will require 1-2 years with our current methods of assessment for the slow nature of Parkinson’s. There is simply no getting around this. After the trial, the results will be analysed (1-2 months). In many modern clinical trials an independent monitoring committee will be actively assessing the results as the study is ongoing, but the finally tallying of results at the end of the study will still take several weeks.
These results are written up and submitted to the regulator for their approval – a process that will take 6 months of peer-review, etc. And then (all things going well) the regulator will say ‘yay’ as opposed to ‘ney’ for the treatment to enter clinical usage. But the process does not stop there. Adoption of the new treatment by clinicians can be a slow process (let’s say 3-6 months).
This is what Barry and James mapped out.
A small segment of the clinical trial process. And it is basic – it doesn’t take into account additional matters such as contracts between research centres and funders (a very slow process). But at least a general overview/structure was there.
What happened next?
Next, we organised a meetup.
On Wednesday 13th February, the “SEAM #22 – Cure Parkinson’s Trust : Enterprise Agility in Practise” was held at the Stack Exchange headquarters in London.
About 30 people met ate pizza and chatted. And after some initial networking, everyone gathered in front of a projector and then Barry, James and yours truly spelt out “What is Parkinson’s and how we clinically test new treatments“. We explain the clinical trial process outlined above, and I was impressed that the ideas started flowing before we even finished the presentation. Comments from the audience included:
“What do you mean there’s no national database of people wanting to take part in a clinical trial?!? That’s idiotic! Why wouldn’t you have a database?!?”
“What about using AI to check and speed up the funding and regulatory/ethics applications?”
“Why can’t you just have more regular committee meetings?”
You get the idea.
We concluded the presentation by asking the attendees to shift to a series of tables and to start brain storming ideas for changes that could help speed up the clinical trial process.
Then what happened?
Well, the attendees shifted to a series of tables and they started brain storming ideas for changes that could help speed up the clinical trial process.
The situation was very dynamic with groups forming, then dividing and then reforming. Satellite groups would pop up off to one side, and disintegrate as quickly as they had formed. I was really intrigued with it all. And I should add that this was the first one of these events I had ever been to, but I really liked it.
 I was hanging around by the pizzas while everyone else was working!
I was hanging around by the pizzas while everyone else was working!
One observation from the evening: The level and intensity of focus from the participants was amazing!
Another observation from the evening: Some of the ideas were pretty cool!
After about 40 minutes of banging ideas around, all of the various groups elected one individual to readout and feedback the ideas to everyone in attendance. The ideas were many.
Can you give me some examples?
Absolutely.
Some of the ideas were well trodden ground, but with clever new twists (for example, using centralised platforms for people to register willingness to take part in trials and provide medical professionals access to their information, which could include a mobile-app (this latter part is already being worked on – right Claire?))
 Source: Dealnews
Source: Dealnews
A few ideas were grand is their scope and ambition (for example, invest in research clinics in large metropolitan areas in developing countries).
Other ideas were a wee bit unethical (for example, incentivise participants to take part and stay in the study until the end – this is a non-starter, as clinical trials would simply start ‘buying’ participants with incentives, making the process even more expensive and resulting in less treatments being tested).
 Not going to happen. Source: thenest
Not going to happen. Source: thenest
Some ideas suggested a better understanding of the actual participant experience of the process (for example, understand the reasons why Parkinsons suffers don’t take part in trials)
And other ideas were certainly worthy of further investigation (for example, building a community platform to promote trials and share experiences after completing the trial – which could feed back to make trials better in the future (could be something for the PRIG group on Facebook to consider?)
All of the ideas/thoughts/comments were written down and collected by Barry and James for further consideration.
Sounds great, but what happens next?
I am not entirely clear on this.
There will definitely be a meeting on how to proceed (we are all too busy this week), and I am certainly keen to follow up a number of the ideas. But I am simply an observer/passenger in this journey, and I am awaiting guidance from Barry and James with regards to next steps.
That said, the day after the SEAM meetup evening, I asked Barry if he would mind if I write a post for the SoPD website about the event. I explained that I would like to let others know about the idea, allow them to follow our progress (oh yes, there will be future posts on this), and hopefully pick up the ball themselves and run with some ideas.
Barry had absolutely no objections.
So all I will say for now is watch this space for updates!
So what does it all mean?
Long time readers will know that I am not the emotional type. I haven’t got time for feelings. But I have to say, that my faith in mankind was slightly restored at the SEAM meetup evening. I mean, here were a group of people with no association what-so-ever (in most cases) with Parkinson’s, and they had just finished a long day at work, but they were prepared to give up their evening to have a go at proposing some solutions to a complicated problem. They could have done whatever they wanted – they could have come along to the meetup, eaten pizza and then legged it.
But they didn’t. I was really inspired.
And there were no vested interests.
Except of course for Barry.
But I have a lot of respect for the proactive individuals in the community who are seeking to improve things, not only for themselves but also for others. Using the skills that they have, to help make progress in the world.
EDITOR’S NOTE – The author of this post is an employee of the Cure Parkinson’s Trust which was associated with the first SEAM meetup focused on clinical trials for Parkinson’s. Neither the SEAM team nor CPT has asked for this post to be written. This post has been provided by the author solely for the purpose of sharing what he considers a really interesting idea – in the hope that it may inspire others to replicate it.
The banner for today’s post was sourced from Meetup






Really inspiring! I like the ideas generated but implementation will be more difficult.
The national database is a sound idea but several already exist . I have been signed up to many and not one has resulted in an opportunity for particpation in research..
LikeLike
Hi Keith,
Yes, implementation and follow through was discussed in the pub after the event. There would need to be a lot of buy in from all stakeholders (clinicians, affected community, PD nurses network, charities, pharma, etc), but it would be in everyone’s interest. Another issue is that NHS needs to step up with genetics testing, etc – at present many trusts simply can’t afford to offer much in this regard – this type of information would be critical in any database.
A daunting task, but worth following up.
Kind regards,
Simon
LikeLike
Interesting blog – Peasant Farmer spot on, though, about signing up to databases. Only one that seems genuinely active is Fox Finder, and even then have not been matched to anything that has progressed.
The PRIG also sounds useful but, alas, have wrested myself free from Facebook.
I really appreciate your blogs – thanks.
LikeLike
Great idea. Maybe find a good trial with promise and run your process improvements on it as a “process improvement” project. Get the low hanging fruit that we control and are not dependent on someone else to change. FoxFinder is us! If we are part of the delay then no excuses if it is in our control as a community to change.
LikeLike
Hi Len,
Thanks for your comment. I completely agree with your thoughts. I was wondering in the days after the event if getting buy-in from some pharmas would a first step once changes are ready to implement. They also have a vested interest in speeding up the process, can bring the required resources to the table, and can probably change clinician mindset better than we can.
But getting the PD community involved is the first step!
Kind regards,
Simon
LikeLiked by 1 person
Hi Jellywoman,
You are welcome & thanks for the interesting comment (I too avoid FB!).
What is the rate limiting step in the database process at the moment? If you sign up for Fox Finder, do they regularly contact you saying there is A,B or C options? Or do they take your information and say “we’ll be in touch”? It would be an interesting exercise to see the database situation/experience from the patient perspective.
Kind regards,
Simon
LikeLike
Thanks Simon.
I have always felt that PwP, their families and friends (and now we can add in young professionals) could be organized to do useful research.
Since we are spread around the world, how about an on-line meetup?
LikeLike
Hi John,
An online meetup would be a fantastic idea! What would be the topic?
Kind regards,
Simon
LikeLike
The best meetup would include PwP, clinicans, researchers and young professionals. It would, at least, deliver prototype solutions. The topic should be general enough to gain from both technical and non-technical responses.
The proposition is: more knowledge, more data, better outcomes.
My suggestion for the topic would be “Small data: making the most of patient experience”. This might include discussion of:
– how to measure the user experience of PD;
– educating PwP;
– how to run a DIY n of 1 trial;
– how to optimize your drug regimen, diet and exercise;
– measuring the effect of supplements;
– blinding a n of 1 trial;
– the role of an ethics committee;
– the information content of high frequency testing;
– post hoc statistics;
– Bayesian statistics;
– meta-statistics of m n of 1 trials;
– database of heterogeneous n of 1 trial data.
LikeLike
Simon, John this on line meet up is an excellent idea to build upon. What are the next steps?
LikeLike
Hi Simon,
Thanks for this post. This looks like much needed work, and not just for Parkinson’s.
My husband took eight months to get onto a trial after diagnosis – there was some kind of mix up and he ended up being on a different trial to the one he was originally booked for. Reading other posts here, his was not an isolated experience.
The process map in your post was a “helicopter view”, done from the perspective of the drug. Was there also a map done from the perspective of a patient walking the process?
Regards,
Paula
LikeLike
Hi Paula,
Thanks for your interesting comment. No, the perspective of the patient was not there on the night (beyond Barry, who has not been in a trial, but is keen to take part). The SEAM event was very much about the outside, independent view point. The patient perspective is a really interesting idea though. Perhaps this could be part of John’s online meetup idea (see other comments).
Kind regards,
Simon
LikeLike
Hello Simon
I am a fairly active responder to requests for participation in on-line contributions to research as well as being on databases for matching. I find:-
It’s easy to get confused about which surveys/questionnaires one has completed. My data might be counted twice!
I answer the same set of questions (or very similar) over and over and over and over again – small wonder if people give up.
Combining a single database that collects the standard elements that the BIG data type of projects can use with a matching service for more specific candidates for trials would make a lot of sense.
Also standardizing the questions might remove the impression I sometimes get of “These people just don’t get it”
LikeLike
Hi Eirwen,
I hope all is well. Thanks for the interesting comment. I know it is annoying to have the same question over and over again, but there is data in the answering of that question. How consistent is the participant with their answers and how does that change over time? I like the idea of combining a single database (Fox insights perhaps?) with a matching service. Will note that down.
Kind regards,
Simon
LikeLike
Hi again
What I was trying to say was that different studies all ask the same basic questions ( quite understandably) and if those basic answers were collected together on one database that researchers could draw on it would save respondents providing the answers more than necessary, so we could do the basics once every 3 months not every 3 months for every study we’re involved with, and then each study could ask just their “special” questions.
Eirwen
LikeLike
Simon,
This progressive approach to shortening the lead time for drug trials is a great idea. I am a Master Black Belt, Lean Six Sigma. We lead teams that are created to study a process to reduce cost, improve quality, increase capacity and reduce the overall cycle time. While improving small steps is a good first step, setting lofty goals usually gets better results. I once read an advertisement for a snow mobile. It said, ‘Industry expert said that reducing the weight of our snow mobile by 20% would be unprecedented, so we shot for 40% and got 50%.’ I was talking with some manufacturing engineers at a Houston Oil Field tool manufacturer. They were distressed that the new VP of Manufacturing had set a goal for reducing tool delivery time from 16 weeks (industry standard) to 4 weeks, in three months. I asked where the project was now. They said that with 3 weeks to go, delivery was at 6 weeks and they hoped to get to 5 weeks by the target date. I suggested that they go to the VP and advise him of their progress and ask for an extension of the due date, to meet the original goal. The VP was quite happy to grant the extension, since they had done such an outstanding job.
The tools of a Continuous Improvement project team are Gant Charts that chart the Critical Path for completion and help to convert the usual series completion process to parallel steps, to shorten the project length. To get around the ‘Not Invented Here’ and ‘We’ve always done it that way’ arguments, we use the ‘5 Whys’. When someone objects to parallel steps, we ask WHY. To the answer, we ask WHY. By the fifth why, you have usually eliminated the objection. If not, you have discovered the critical path for that process.
Best regards,
Joe Cambell
LikeLike