|
The results of a small clinical study evaluating the safety and tolerability of Ursodeoxycholic acid (or UDCA) in people with Parkinson’s have recently been published. UDCA is a naturally occurring bile acid that is used in the treatment of gallstones. More recently, however, researchers have reported that this clinically available medication has beneficial effects in models of Parkinson’s. The clinical study that has recently been published suggests that UDCA is safe and well tolerated in people with Parkinson’s, and warrants further investigation in larger clinical trials. In today’s post, we will discuss what UDCA is, we will consider some of the previous research in models of Parkinson’s, we will review the results of the clinical trial, and then we will discuss what may happen next.
|
 Source: Youtube
Source: Youtube
How often do you consider your gallbladder?
Excuse me?
Your gallbladder. How often do you think about it?
Uhh,….never?
And I would believe that. It is one of the less appreciated organs. A pear-shaped, hollow organ located just under your liver and on the right side of your body. Its primary function is to store and concentrate your bile. What is bile you ask? Bile is a yellow-brown digestive enzyme – made and released by the liver – which helps with the digestion of fats in your small intestine (the duodenum).
 Source: Mayoclinic
Source: Mayoclinic
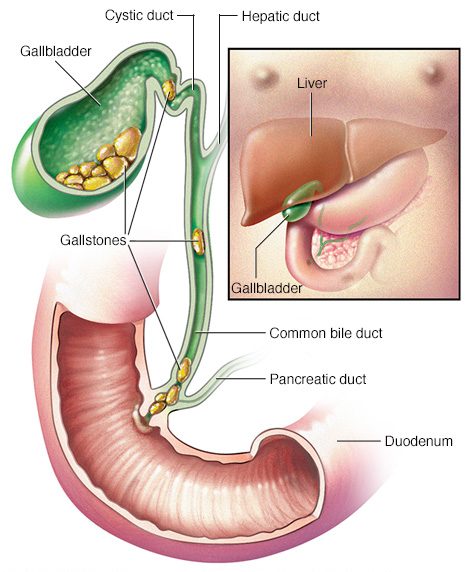
One of the down sides of having a gall bladder: gallstones.
Gallstones are hardened deposits that can form in your gallbladder. About 80% of your average gallstone is cholesterol. The remaining 20% of a gallstone is made of calcium salts and bilirubin. Bilirubin is the yellow pigment in bile. When the body produces too much Bilirubin or cholesterol, gallstones can develop.
 Gallstones – ouch! Source: Healthline
Gallstones – ouch! Source: Healthline
About 10-20% of the population have gallstones (Source), but the vast majority experience no symptoms and need no treatment.
Interesting intro, but what does any of this have to do with Parkinson’s?
One of the treatments for gallstones is called UDCA. And recently we learned the results of a clinical trial in which UDCA is being “repurposing” as a treatment for Parkinson’s.
What is UDCA?