|
This week a new clinical trial was registered which caught our attention here at the SoPD HQ. It is being sponsored by a small biotech called Neuraly and involves a drug called NLY01. NLY01 is a GLP-1R agonist – that is a molecule that binds to the Glucagon-like peptide-1 receptor and activates it. Other GLP-1R agonists include Exenatide (also called Bydureon) which is also also about to start a Phase III clinical trial in Parkinson’s (Click here to read a previous SoPD post about this). There is a lot of activity in the Parkinson’s research world on GLP-1R agonists at the moment. In today’s post, we will discuss what a GLP-1R agonist is, what we know about NLY01, and what the new clinical trial involves.
|
Every week there are new clinical studies being announced for Parkinson’s.
Many of them are registered on the Clinicaltrials.gov website. Here at the SoPD, we try to keep track of new trials being registered (the SoPD Twitter account highlights the more interesting trials).
This week one particular newly registered clinical trial stood out. It involves a small biotech company Neuraly (which is owned by parent company D&D PharmaTech).
 And the drug being tested in the Neuraly clinical trial is a GLP-1R agonist.
And the drug being tested in the Neuraly clinical trial is a GLP-1R agonist.
What is a GLP-1R agonist?
Before I answer that, let’s start with glucose.
Glucose?
Yes, glucose.
Glucose is a great source of energy for the cells in our bodies. After eating food, our body releases a protein called insulin which then attaches to cells and signals to those cells to absorb the new glucose that is flooding our bloodstream. Without insulin, our cells have a really hard time absorbing glucose.
Think of insulin as a “key” which unlocks cells to allow glucose to enter the cell.
 Insulin. Source: Wikipedia
Insulin. Source: Wikipedia
The pancreas is the organ in your body that produces insulin, which is critical for maintaining normal glucose levels in our bodies. Insulin instructs cells to take in and use glucose from the blood. This has the effect of lowering blood sugar. The hormone Glucagon has the opposite effect – it tells the body to release glucose into the blood to raise sugar levels.
Now we get to the GLP-1 part of your question.
Glucagon-like peptide-1 (or GLP-1) is a hormone that stimulates insulin production while blocking glucagon release.

The function of GLP-1. Source: Wikipedia
But what is a GLP-1R agonist?
On the surface of cells there are small proteins called receptors, which act like switches for certain biological processes. Receptors will wait for another protein to come along and bind to them. In binding to the receptor, the protein will either activate the receptor or inhibite it.
The proteins that activate the receptors are called agonists, while the inhibitors are called antagonists.
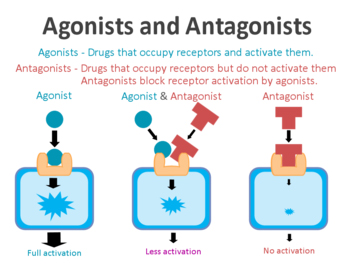
Agonist vs antagonist. Source: Psychonautwiki
So GLP-1R agonists are molecules that activate the GLP-1 receptor (GLP-1R).
Activation of the GLP-1 receptor by a GLP-1 receptor agonist results in the activation of many different biological pathways within a cell:

The GLP-1 signalling pathway. Source: Sciencedirect
Of particular importance is that GLP-1 receptor activation inhibits certain “cell death” pathways, reduces inflammation, reduces oxidative stress, and increases neurotransmitter release.
All pretty positive stuff really.
For a recent and very good OPEN ACCESS review of the GLP-1-related Parkinson’s research field, click here.
Ok. So NLY01 is a GLP-1R agonist. Is it similar to other GLP-1R agonists like exenatide?
NLY01 is a PEGylated form of exenatide.
What does that mean? Pegylated?
PEGylation describes the attachment of polyethylene glycol (PEG; which is a non-toxic and non-immunogenic molecule) to a protein. This process can help improve the therapeutic properties of the protein being PEGylated.
But understand that NLY01 is essentially very similar to exenatide, which has been tested in people with Parkinson’s (Click here to read a previous SoPD post about this).
What do we know about NLY01 in Parkinson’s?
In the middle of last year, this report was published:
 Title: Block of A1 astrocyte conversion by microglia is neuroprotective in models of Parkinson’s disease
Title: Block of A1 astrocyte conversion by microglia is neuroprotective in models of Parkinson’s disease
Authors: Yun SP, Kam TI, Panicker N, Kim S, Oh Y, Park JS, Kwon SH, Park YJ, Karuppagounder SS, Park H, Kim S, Oh N, Kim NA, Lee S, Brahmachari S, Mao X, Lee JH, Kumar M, An D, Kang SU, Lee Y, Lee KC, Na DH, Kim D, Lee SH, Roschke VV, Liddelow SA, Mari Z, Barres BA, Dawson VL, Lee S, Dawson TM, Ko HS
Journal: Nature Medicine, 2018 Jul;24(7):931-938.
PMID: 29892066
(Before we start here, I just want to say: This report is a beast! It is massive – so much work has been done in the one report).
In this study, the researchers were investigating the use of NLY01 in models of Parkinson’s. They started their analysis by treating normal mice and primates with the drug to assess the pharmacodynamics and pharmacokinetic of the drug.
What does that mean?
Pharmacodynamics explores how a drug affects an organism (eg. mechanism of action), while pharmacokinetics looks at how the organism affects the drug (eg. how well is it aborbed and excreted).
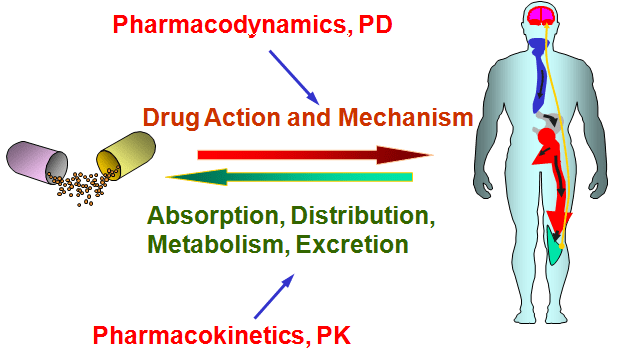 Source: Pinterest
Source: Pinterest
NLY01 had a very impressive half-life of 38 hours in mice and 88 hours (in primates). The half-life of a drug is the time required for the concentration of the drug to decrease by half in the body.
NLY01 also did not increase the risk of hypoglycemia (or low blood sugar) when administered in the animals. And it reached the brain very efficiently – having no issues with the blood brain barrier (the protective membrane surrounding the brain).
Next, the researchers tested the drug on two different mouse models of Parkinson’s:
- the alpha synuclein preformed fibril model
- the hA53T genetically engineered mouse
What are alpha synuclein preformed fibrils?
As regular readers will be aware alpha synuclein is considered to be one of the main trouble makers in Parkinson’s. It may sound like a distant galaxy, but it is an extremely abundant protein in our brains – making up about 1% of all the proteins floating around in each neuron.
When alpha synuclein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure. Alone, it will look like this:
 Alpha synuclein. Source: Wikipedia
Alpha synuclein. Source: Wikipedia
By itself, alpha synuclein is considered a monomer, or a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). In Parkinson’s, alpha synuclein also aggregates to form what are called ‘fibrils’.

Microscopic images of Alpha Synuclein (AS) monomers, oligomers and fibrils. Source: Brain
And over the last few years research labs have been taking alpha synuclein and turning them into fibrils, and then using those ‘preformed fibrils’ in models of Parkinson’s. For example:
 Title: Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice.
Title: Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice.
Authors: Luk KC, Kehm V, Carroll J, Zhang B, O’Brien P, Trojanowski JQ, Lee VM.
Journal: Science. 2012 Nov 16;338(6109):949-53.
PMID: 23161999 (This report is OPEN ACCESS if you would like to read it)
In this study, when the researchers injected preformed fibrils of alpha synuclein into the mice, they started to observed alpha synuclein protein aggregation in the brain approximately one month later, and the mice started to exhibit motor problems 3-6 months later.
In the NLY01 study, the researchers injected their preformed fibrils into mice and they waited one month before initiating treatment of the mice with either NLY01 or a placebo treatment. They treated the mice for twice per week for 5 months.
The results found that NLY01 treatment significantly reduced the amount of alpha synuclein aggregating and also reduced the loss of dopamine neurons. The drug also rescued the motor deficits across a range of behaviour tests.
Next the researchers tested NLY01 on the hA53T genetically engineered mouse.
And what is the hA53T genetically engineered mouse?
There is a region of your DNA that provides the instructions for making alpha syncuclein. That region of DNA is called SNCA. And there are several genetic variations (or mutations) inside of SNCA that are associated with an increased risk of developing Parkinson’s.
A53T is the name of one of those genetic variations.
As you can see in the image below, A53T lies in the red (Amphipathic) region of SNCA along with several other genetic variants, such as A30P and E46K:
Mice have been genetically engineered to carry the SNCA gene with the A53T genetic mutation (Click here to read the original report). These mice exhibit hyperactivity and then start to display signs of alpha synuclein protein aggregation at about four to six months of age. They also pass away earlier than normal mice (12-14 months of age, compared to 20+ months for normal mice).
In the NLY01 study, the investigators started treating the A53T mice at 6 months of age with either NLY01 or a placebo control, and they reported that NLY01 significantly prolonged the lifespan of the A53T mice by over 3 months. In addition to this prolonged survival, the researchers also found a reduction in levels of aggregated alpha synuclein in the brains of the NLY01-treated A53T mice.
 Labmouse. Source: PBS
Labmouse. Source: PBS
Next, the researchers wanted to try and determine how NLY01 was having this effect (the ‘mechanism of action’), and they started by labelling GLP-1 receptors in cell cultures of difference types of brain cells (astrocytes, microglia and neurons) and then they looked to see where GLP-1 was most abundant.
Interestingly, they found that microglia cells have the highest levels of GLP-1 receptors.
What exactly are microglia?
Microglia are some of the helper cells in the brain. They act as the resident immune cells. When infection or damage occurs, the microglia become ‘activated’ and start cleaning up the area.
 Different types of cells in the brain. Source: Dreamstime
Different types of cells in the brain. Source: Dreamstime
Also of interest: the researcher reported that treating the cells with preformed fibrils of alpha synuclein decrease GLP-1 levels in neurons and increased GLP-1 levels almost two fold in microglia. And when they looked at postmortem tissue, the investigators found a 10x increase in the levels of GLP-1 in the substantia nigra of people who passed away with Parkinson’s (compared to healthy controls samples). The substantia nigra is the region of the brain where the dopamine neurons reside, and it is badly affected in Parkinson’s.
The finding that microglia have higher levels of GLP-1 protein led the researchers to test the idea that perhaps NLY01 was having its effect via the microglia cells rather than the neurons. So they treated cultures of just dopamine neurons with preformed fibrils of alpha synuclein and the investigators found that NLY01 treatment had no protective effect on the cells.
 Dopamine neurons (green) in cell culture. Source: Axolbio
Dopamine neurons (green) in cell culture. Source: Axolbio
Previously the researchers conducting this study had demonstrated that activation of microglia can cause them to release all sorts of nasty, inflammatory chemicals. They do this to alert other microglia to a potential problem. But these chemicals can also cause astrocytes to shift from a neutral state to a highly reactive state (Click here to read more about this).
 An astrocyte. Source: Wikipedia
An astrocyte. Source: Wikipedia
Astrocytes, like microglia, are helper cells in the brain. They provide nutrients to neurons and make sure the environment surrounding the neurons is balanced and supportive. When astrocytes shift from a neutral state to a reactive state, however, it can be very bad news for any neurons nearby. And given this scenario, the researchers began to wonder whether the interactions between microglial cells and astrocytes could be involved in the neuroprotective effects of NLY01.
They decided to test this idea by firstly treating microglia with preformed fibrils of alpha synuclein. This got the microglia nice and activated, and the researchers then split the microglia in to two groups: one group was treated with NLY01 and the other group was not. Next the investigators collected the solutions from these two groups of microglia cultures, and they applied those solutions to two groups of astrocytes in cell culture. Astrocytes given the NLY01-treated microglial solution did not become reactive.
And subsequent experiments demonstrated that this reduction in reactive astrocytes was neuroprotective for dopamine neurons when they were grown in culture solution from the NLY01-treated activated microglia and reactive astrocytes.
I hope all of this makes a little bit of sense.
Long story short/the take home message:
- Preformed fibrils of alpha synuclein activate microglia
- Activated microglia spit out nasty, inflammatory chemicals
- Astrocytes exposed to those chemicals get reactive
- Reactive astrocytes are bad news for nearby neurons
And NLY01 limits the activated microglia from spitting out too much of the nasty, inflammatory chemicals, thus lowering the number of reactive astrocytes.
NLY01 appears to be having its effect via the microglial cells.
All clear?
Interestingly, when the researchers removed astrocytes from the equation by exposing the neurons to solution collected from activated microglia, there is some cell death in the neuronal population, but it is not as much as that seen in the cultures including reactive astrocyte solution.
Thus, reactive astrocytes seem to be critical to the neuronal cell death.
And when the researchers looked at the brains of the ‘preformed fibrils of alpha synuclein’ injected mice that had been treated with NLY01 for 5 months (which we discussed above), they found a reduced number of activated microglia.
These results led the researchers to conclude that “NLY01 exhibits protective effects primarily through microglial GLP1R“. And because of this specific mechanism, the investigators suggest that “NLY01 may have broad neuroprotective properties in a variety of neurodegenerative disorders and neurologic injuries“, beyond just Parkinson’s.
And obviously, after the results of this study, the researchers were keen to test NLY01 is a clinical trial.
So has NLY01 ever been clinically tested in humans?
Clinical testing is a multiple phase process.
NLY01 has already been tested in a Phase I safety/tolerability trial involving 96 healthy individuals. That study in involved two parts:
In Part A of that study, the investigators tested 5 single doses. Each dose was tested on one group of participants. The doses increased across the groups (from a low dose group to a high dose group). The participants in each group were randomised to receive either NLY01 or placebo.
In Part B of the study, the participants received once-weekly doses of NLY01 (or placebo) for 4 weeks. While part A of the study was a single dose study, Part B was a multi dose study. Three groups of participants were used, each group receiving either 15%, 35% or 70% of the single-dose maximum tolerated dose from Part A of the study. All of the Part B groups received a fixed dose of NLY01 (or placebo) once-weekly for the 4 week treatment period.
This Phase I study is now complete (Click here to read more about the structure of the study). We are still waiting to see the publication of the results of that Phase I safety/tolerability trial, but given that a Phase II trial has now been initiated, we can assume that the folks at Neuraly (and the regulators) here happy with the initial results to proceed to the next phase of clinical testing.
So what is the next phase?
Neuraly has just registered a Phase II study designed to assess the safety, tolerability and efficacy of NLY01 in participants with early untreated Parkinson’s. So while the Phase I study evaluated NLY01 in healthy individuals, this new trial will be assessing the drug in the cohort of interest: people with Parkinson’s.
The new Phase II trial will involve 240 participants in a multi-center study. The recruited subjects will be randomised into 3 groups – 2.5 mg injection (yes, NLY01 is an injected treatment, just like exenatide), 5.0 mg injection, or placebo. All of the treatments will be self administered once per week for 36 weeks.
The primary end point of the study (the measure by which the drug is tested) will be the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS parts II and III). The trial will also be double-blind, meaning that no one involved (participants or clinicians) will know who is being treated with what until the end of the 36 week study.
The results will be available after March 2021 (Click here to read more about the trial)
What do they mean by “participants with early untreated Parkinson’s”?
This means recently diagnosed individuals. In the “Exclusion Criteria” of the study it says that individuals with any “prior use of dopaminergic treatment or MAO-B inhibitors for more than 28 days” will not be considered for this study. This detail differs from the Phase III exenatide study which is about to start – the Phase III study is recruiting individuals who have been on “dopaminergic treatment for at least 4 weeks before enrolment” (Click here to read the details of that study).
What does it all mean?
Efforts to repurpose GLP-1R agonists – a class of drugs which are used for treating diabetes – to Parkinson’s has recently gone up a gear with a number of new clinical trials about to get underway.
for those keeping score, there is the:
- Phase I Exenatide in Parkinson’s study in Florida (USA – click here to read more about this).
- Phase II Liraglutide in Parkinson’s study in Los Angeles (Click here to read more about this).
- Phase II Lixisenatide in Parkinson’s study in France (Click here to read more about this).
- Phase II Semaglutide in Parkinson’s study in Norway (Click here to read more about this).
- Phase II NLY01 in Parkinson’s study (Re-read this entire post for more about this).
- Phase III Exenatide in Parkinson’s study in the UK (Click here to read more about this).
All of this GLP-1R clinical research will build a considerable body of data once the results are in. A lot of ‘expectations’ have been built up around this class of drug for Parkinson’s since the 2017 publication of the results of the exenatide Phase II trial. But in order to build that ‘considerable body of data’, we need to manage those expectations and conduct these trials in an unbiased fashion so as to truly determine if GLP-1R agonists could really be a disease modifying treatment for Parkinson’s.
The registration of the next phase of NLY01 clinical testing is a positive new development, and here at the SoPD we will be watching closely as the trial gets underway.
The banner for today’s post was sourced from BusinessWire



What happened to the Ambroxol studies ? I thought in 2019 we would get results ? 2019 is almost over.
LikeLike
Hi Diego,
As I understand things the results are about to be published.
Kind regards,
Simon
LikeLike
Thanks for the info.
LikeLike
Simon, sorry for the late read on this one. This explains how the drug prevents microglia activation. However once a neuron has already been infected how does the synuclein clearance take place?
LikeLike
Hmm, OK. So Neuraly’s PEGylated exenatide essentially works by preventing microglia from entering the destructive form of activation at one time referred to as M1. But given that curcumin and a number of other substances also can do this, why the big fuss over exenatide?
I mean, it is cool that they could show that putting some microglia all alone in a test tube and then adding alpha synuclein pre-formed fibrils activated them, and that adding exenatide reversed that activation. But those microglia also have receptors that respond to neuromelanin that is dumped from dying neurons, and curcumin and baicalein/baicalin can antagonize those receptors, as well as receptors that respond to aggregatedalpha-synuclein, which also reduces the likelihood of M1 activation.
So is exenatide just one additional anti-inflammatory in an arsenal of such that already includes several commonly-available supplements? If not, then what makes it special?
LikeLike
Interesting also that curcumin is shown to stimulate the release of actual GLP-1. I mean, a receptor agonist is great, but ain’t nothing like the real thing…
https://www.sciencedirect.com/science/article/abs/pii/S0300908419302457
LikeLike