A biotech company in Australasia got the green light for the next round in a clinical trial two weeks ago.
Their product: tiny cylinders filled with pig cells.
Their mission: to treat Parkinson’s disease with the regenerative healing properties of naturally occurring cells.
In today’s post we will look at what the company is doing and what will happen next.

Source: ProactiveInvestors
We have been contacted by several readers asking for a post on the press release last week regarding the clinical trial being conducted by Living Cell Technologies Limited (LCT).
Two weeks ago LCT received approval to commence the treatment of 6 patients in their third group of subjects in a Phase IIb clinical trial of NTCELL® for Parkinson’s disease, at Auckland City Hospital in New Zealand (Click here for the press release).
The company completed treatment of all six patients in ‘group 2’ of the Phase IIb clinical trial of NTCELL for Parkinson’s disease at the end of 2016. Four patients in the trial had 40 NTCELL microcapsules implanted into the putamen on each side of their brain, and two patients had sham surgery with no NTCELL implanted. They now have approval to repeat this in a third group of subjects.
What do we know about the company?
Founded in 1999, the initial goal of the company was to develop regenerative cell therapies. This goal was to be achieved by transplanting cells from Auckland Island pigs into humans.
The first disease considered for this approach was type 1 diabetes, which is now being pursued by a joint venture company in the US while LCT focuses its attention on Parkinson’s disease.
What are NTCELL microcapsules?
NTCELL is an a tiny capsule, that contains choroid plexus cells (taken from pigs). The capsule is made of a semi permeable membrane that allows all of the good chemicals and nutrients (that the cells are producing) to escape into the surrounding environment. At the same time it doesn’t let the cells escape, nor does it allow negative elements into the capsule. In addition, the bodies immune system can’t get at the foreign cells and remove them due to the membrane surrounding the capsule.

An example of encapsulated cells. Source: LEN
These capsules can be transplanted into the brain of people with neurodegenerative conditions, providing the brains of those individuals with the benefits of supportive chemicals and nutrients.

A brain scan of NTCELL capsules transplanted in the human brain. Source: LCT
Interesting, but what are choroid plexus cells?
Believe it or not, there are some empty spaces inside your brain. Spaces where there are no brain cells (neurons).
These spaces are called the ‘ventricles‘.
In the human brain there are 4 basic divisions of the ventricles as you can see in the image below (the ventricles are the yellow space):

The ventricles and choroid Plexus in the human brain (red coloured regions). Source: PhysRev
The ventricles are filled up with a solution called cerebrospinal fluid. Cerebrospinal fluid is very similar to the liquid portion of blood (or plasma – if you remove the cells from blood, it’s called plasma), except that cerebrospinal fluid is nearly devoid of protein. It is actually made from plasma, but it only contains 0.3% of plasma proteins and about 2/3 of the glucose of blood.
The choroid plexus cells are one of the primary sources of production for the cerebrospinal fluid. That production is actually great – total volume of cerebrospinal fluid in the the average human being turns over almost 4 times per day. Choroid plexus cells can be found in all 4 divisions of the ventricular system (the choroid plexus cells are indicated with red/brown colouring in the image above).
And, um… why pigs?
The choroid plexus cells are sourced from a unique herd of pigs that have been designated pathogen-free. They were originally sourced from the remote sub-Antarctic Auckland Islands, where they have been running around in isolation since 1807.
The not-so-tropical Auckland Islands, south of NZ. Source: Sciblogs
That isolation has made them ‘pathogen free’ – basically there is a reduced likelihood of endogenous infectious agents (eg. porine (pig) retrovirus (or PERVs)) in the cells – which is a good thing when you are planning to stick something in the brain.
What research has been done on NTCELL?
Firstly, regarding the capsules, the company published this report in 2009:

Title: Encapsulated living choroid plexus cells: potential long-term treatments for central nervous system disease and trauma.
Authors: Skinner SJ, Geaney MS, Lin H, Muzina M, Anal AK, Elliott RB, Tan PL.
Journal: J Neural Eng. 2009 Dec;6(6):065001.
PMID: 19850973
In this study, the company looked at the utility of the capsules in rodent brains. One important aspect that they wanted to address was how well the cells survive inside the capsules when placed in the brain. They found that the capsules effectively protected the cells from the host immune system, and they survived for the length of the 6 months study without causing any adverse effects.
The capsules were retrieved from the brains of the rats at the end of the study and the viability of cells was analysed. The researchers found that there was no difference in the production of nutrients from the cells in the capsules at 4 months post implantation, but they did see a decrease of 33% at 6 months. In addition, the number of cells decreased to approximately 40% of the pre-implantation values at 6 months.
We are unsure whether the capsules have been altered for the clinical trial.
The researchers followed this research up in 2013 by publishing this paper:

Title: Recovery of neurological functions in non-human primate model of Parkinson’s disease bytransplantation of encapsulated neonatal porcine choroid plexus cells.
Authors: Luo XM, Lin H, Wang W, Geaney MS, Law L, Wynyard S, Shaikh SB, Waldvogel H, Faull RL, Elliott RB, Skinner SJ, Lee JE, Tan PL.
Journal: J Parkinsons Dis. 2013 Jan 1;3(3):275-91. doi: 10.3233/JPD-130214.
PMID: 24002224 (This article is OPEN ACCESS if you would like to read it)
The researchers wanted to test the capsules in non-human pre-clinical trials. For this purpose they induced Parkinson’s disease in 15 monkeys using the neurotoxin MPTP, waited 10 weeks and then implanted their capsules. Six monkeys were implanted with the NTCELL capsules, 6 were implanted with empty capsules, and 3 received no capsules. The animals were then tested out to 24 weeks post implantation.
The behavioural response was dramatic. Most of the primates with the NTCELL capsules demonstrated positive behavioural benefits by 2 weeks post implantation (becoming statistically significant by 4 weeks), while the controls and empty capsule groups exhibited no behavioural recovery at all across the entire 24 weeks.
In addition to behavioural benefits, the investigators found significantly more dopamine neurons in the brains of the monkeys implanted with the NTCELL capsules when compared to the controls.
These findings were used by the company to justify moving towards clinical trials in humans.

Source: Healthline
And what do we know about the clinical trial for Parkinson’s disease?
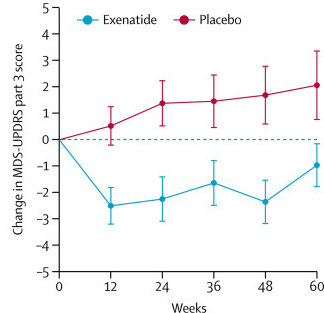
A Phase I/IIa NTCELL clinical trial for the treatment of Parkinson’s disease was completed in June 2015. It was an open label investigation of the safety and clinical effect of NTCELL in 4 people who had been diagnosed with Parkinson’s disease for at least five years.
The trial “met the primary endpoint of safety” and “reversed progression of the disease two years after implant” (as measured by the Unified Parkinson’s Disease Rating Scale (UPDRS)). The NTCELL implantation was well tolerated, with “no adverse events considered to be related to NTCELL”. The results of the trial have not been published, but the press release can be found here.
The results from that trial were used to justify and design a larger Phase IIb trial.
What does Phase IIb mean?
Phase II studies, which are designed to address clinical efficacy and biological activity, can be divided into IIA or IIB, and while there is no stated definition for these labels it is generally agreed that:
- Phase IIA studies are usually pilot studies designed to demonstrate clinical efficacy or biological activity (‘proof of concept’ studies);
- Phase IIB studies look to find the optimum dose at which the drug shows biological activity with minimal side-effects (‘definite dose-finding’ studies) – (Source: Wikipedia).
The goal of this Phase IIb LCT clinical study is to “confirm the most effective dose of NTCELL, define any placebo component of the response and further identify the initial target Parkinson’s disease patient sub group”.
A total of 18 patients under 65 years old are taking part in the trial being conducted at Auckland Hospital and Mercy Ascot Hospital in New Zealand. The company will have to wait 26 weeks until after the last patient is implanted to know whether it has been successful in meeting regulator’s conditions on quality, safety, and efficacy. At the 26 weeks mark, the subjects that received the placebo (empty capsules) will be given the NTCELL capsules.
If the current Phase IIb trial is successful, Living Cell Technologies Limited will be looking to “apply for provisional consent to treat paying patients in New Zealand and launch NTCELL® as the first disease modifying treatment for Parkinson’s disease, in 2017” (Source: Ltcglobal). We will wait to see the results of the current study before passing judgement on whether this situation is likely, though it does seem premature given that by the end of the phase IIb trial only 20 people with Parkinson’s disease will have received the NTCELL treatment. A larger phase III trial may be required. Alternatively, if the results of the current trial are truly spectacular, the company may be able to propose a Phase IV style of trial (also called a ‘post-marketing surveillance’ trial) which would allow them to market their product, but they would be required to maintain a strict program of safety surveillance and ongoing technical analysis of the treatment.
Are other companies trying to do something similar?

Source: NSgene
Another company, NSgene (in Denmark) has a similar sort of experimental product called NsG0301 which is encapsulated human cells that express the neuroprotective protein, GDNF. NsG0301 is still in preclinical testing however, with the Michael J Fox Foundation helping the company to get the clinical trials started.
Sounds very interesting, but what does it all mean?
So in summary, the biotech company LCT have been given permission to continue with their phase II clinical trial which involves placing tiny capsules which contain cells that release beneficial nutrients into the brains of people with Parkinson’s disease. The company will be blind to which individuals are receiving the capsules with cells in them or empty capsules. They should know later in the year if the trials is successful.
One positive feature of this idea is that immune-suppressant treatments are not required as they are with other forms of transplantation therapies. This means that the patient doesn’t need to take medication which stops the immune system from attacking the foreign cells, because the cells are protected by the capsule membrane. Such medication can leave subjects with reduced immune system responses to illness and thus vulnerable.
Having said that, we are a little concerned that the NTCELL product has not been tested thoroughly enough in Parkinson’s disease for the company to be proposing it for commercial use later this year. For example, the phase I open label results could easily be the result of the placebo effect in practise (as all 4 participants knew they were receiving the capsules. This issue could be resolved with DATscan brain imaging of the first 4 subjects (in the phase I trial).
In addition, we would be interested to know how long the cells inside the capsules keep producing cerebrospinal fluid and other beneficial nutrients once inside the human brain. The rodent study (reviewed above) suggested reductions in production from the cells after just 6 months.
While the NTCELL capsules have been tested in many different models of neurological conditions (see the LCT’s publication page for more on this), the company suffered a set back in 2014 when they retracted one of their key pieces of research which demonstrated the use of NTCELL in a rodent model of Parkinson’s disease (Click here for more on this). The study in question was conducted by LCT between 2007 – 2009, and the results were published in The Journal of Regenerative Medicine in 2011. The study was retracted, however, because “the efficacy conclusions in the publication cannot be confirmed”.
To be fair, the company requested the retraction themselves – which is to their credit – and as a precautionary measure LCT placed a hold on any further patient recruitment in their Phase I/IIa clinical study that was underway at the time. But with this study retracted, the published preclinical research for NTCELL in Parkinson’s disease is largely limited to the primate study reviewed above (we are happy to be corrected on this).
We will be intrigued to see the results of the phase II trial, and (if all goes well) whether the New Zealand regulators will be happy for the product to be made commercially available. Depending on the results, they may request further studies. It will definitely be interesting to follow up long-term the 20 subjects who will have received the NTCELL product by that time.
We watch and wait.
UPDATE FROM 1st MAY 2017:
Today Living Cell Technologies Limited posted the following press release:
Treatment completed for all patients in Parkinson’s trial
Living Cell Technologies Limited has completed treatment of all six patients in the third and final group of patients in the Phase IIb clinical trial of NTCELL® for Parkinson’s disease, at Auckland City Hospital.
Four patients had 120 NTCELL microcapsules implanted into the putamen on each side of their brain, and two patients had sham surgery with no NTCELL implanted. To date there are no safety issues in any of the six patients.
The company is blind to the results until 26 weeks after the completion of group 3 of the trial. The results will then be analysed in accordance with the statistical plan and the conclusions announced. This is anticipated to occur in November 2017. Thereafter the patients who received the placebo will receive the optimal treatment.
The Phase IIb trial aims to confirm the most effective dose of NTCELL, define any placebo component of the response and further identify the initial target Parkinson’s disease patient sub group. Providing the trial is successful, the company will apply for provisional consent in Q4 2017 with a view to treating paying patients in New Zealand in 2018.
“The completion of treatment for the patients in group 3 brings us a step closer to our goals of obtaining provisional consent and launching NTCELL as the first disease modifying treatment for Parkinson’s disease,” says Dr Ken Taylor, CEO of Living Cell Technologies.
FULL DISCLOSURE: Living Cell Technologies Limited (LCT) is an Australasian biotechnology company that is publicly listed on the ASX and NSgene is a privately owned company. Under no circumstances should investment decisions be made based on the information provided here. In addition, SoPD has no financial or beneficial connection to either company. We have not been approached/contacted by either company to produce this post. We are simply presenting this information here following requests from our readers and because we thought the science of what the company is doing might be of interest to other readers. The author of this blog is associated with an individual contracted by LCT, but that individual did not request nor was not made aware of this post before publication.
The banner for today’s post was sourced from the Planner
 I am the green string. Barry is everything else. Source: Philiphemme
I am the green string. Barry is everything else. Source: Philiphemme