
|
Dipeptidyl peptidase-4 (or DPP-4) is an enzyme that breaks down the protein (GLP-1) that stimulates insulin release in your body. Inhibitors of DPP-4 are used in the treatment of Type 2 diabetes, because they help increase insulin levels in the body. Recently some Swedish researchers noticed something curious about DPP-4 inhibitors: They appear to reduce the risk of developing Parkinson’s disease. In today’s post, we will review what DPP-4 inhibitors do and look at how this could be reducing the risk of Parkinson’s disease. |
Sitagliptin. Source: Diabetesmedicine
Last year an interesting research report about a class of medications that could possibly reduce the risk of developing Parkinson’s disease was published in the journal Movement disorders:

Title: Reduced incidence of Parkinson’s disease after dipeptidyl peptidase-4 inhibitors-A nationwide case-control study.
Authors: Svenningsson P, Wirdefeldt K, Yin L, Fang F, Markaki I, Efendic S, Ludvigsson JF.
Journal: Movement Disorders 2016 Jul 19.
PMID: 27431803
In this study, the investigators used the Swedish Patient Register, to find the medical records of 980 people who were diagnosed with Parkinson’s disease but also had type 2 diabetes. Importantly, all of the subjects had been treated with Type 2 diabetes medication for at least 6 months prior to the date of Parkinson’s being diagnosed.
For comparative sake, they selected 5 controls (non-Parkinsonian) with Type 2 diabetes (n = 4,900) for each of their Parkinsonian+diabetes subjects. They next looked at whether GLP-1R agonists (such as Exenatide), Dipeptidyl peptidase-4 (or DPP-4) inhibitors, or any other oral Type 2 diabetic medication can influence the incidence of Parkinson’s disease.
Now, if all things are considered equal, then when looking at each diabetes medication there should be 1 person in the Parkinson’s disease + Type 2 diabetes for every 5 people in the Type 2 diabetes control group taking each medication right? That is because there are almost 1000 people in the first group and 5000 in the second group.
But this is not what the researchers found.
Early use of GLP-1R agonists was seen in just 1 person with Parkinson’s disease + Type 2 diabetes and 7 Type 2 diabetes controls. Given the low number of people using this class of drug, the investigators were reluctant to conclude very much from this result. But the really interesting discovery was that early use of DPP-4 inhibitors was seen in 3 people with Parkinson’s disease + Type 2 diabetes and 64 Type 2 diabetes controls.
I think you’ll agree that a 3:64 (or 1:21) ratio is slightly larger than a 1:5 ratio.
While the researcher were again caution about concluding too much from this result, they still suggested that there is a significant decrease in the incidence of Parkinson’s disease among individuals with a history of DPP-4 inhibitor intake. They finished by suggesting that future studies with a larger sample sizes and longer follow-up period should be conducted to confirm this observation.
Now this result by itself is pretty weak due to the low numbers. But it is supported by other research that has been conducted on DPP-4, which agrees that there could be something interesting going on here.
Hmm, intriguing. But what is DPP-4?
Dipeptidyl peptidase-4 (also known as DPP-4, or adenosine deaminase complexing protein 2 or CD26) is an enzyme that sits on the surface of most types of cells in your body.

The structure of DPP-4. Source: Wikipedia
It functions as a rather indiscriminate enzyme, breaking down a wide range of proteins. A particular protein will do its job and then DPP-4 will chew it up and get rid of it. The proteins targeted by DPP-4 include:
- Brain natriuretic peptide (BNP)
- Erythropoietin
- Eotaxin
- Gastrin-releasing peptide (GRP)
- Glucagon
- Glucagon-like peptide-1 (GLP-1)
- Glucagon-like peptide-2 (GLP-2)
- Glucose-dependent insulinotropic polypeptide (GIP)
- Granulocyte colony-stimulating factor (G-CSF)
- Granulocyte-macrophage colony-stimulating factor (GM-CSF)
- GHRH and IGF-1
- High-mobility group box 1 (HMGB1)
- Macrophage-derived chemokine (MDC)
- Macrophage inflammatory protein-1 α (MIP-1 α), chemokine (C-C motif) ligand 3-like 1 (CCL3L1), or LD78β
- Oxyntomodulin
- Pituitary adenylate cyclase-activating polypeptide (PACAP)
- Neuropeptide Y (NPY)
- Peptide tyrosine tyrosine (PYY)
- Regulated on activation, normal T cell expressed and secreted (Rantes)
- Stromal cell-derived factor-1 (SDF-1)
- Substance P (SP)
Click here for a very good OPEN ACCESS review of DPP-4 and all of the proteins that it interacts with.
One particular protein on that list that is of interest to us in the Parkinson’s community is GLP-1.
Drugs based on GLP-1 are used in treatment of Type II diabetes. GLP-1 is a hormone that stimulates insulin production while blocking glucagon release. Insulin instructs cells to take in and use glucose from the blood (which helps lower blood sugar levels) while the hormone Glucagon has the opposite effect – it tells the body to release glucose into the blood and raises blood sugar levels.
The function of GLP-1. Source: Wikipedia
GLP-1-based drugs have been in the news recently as a phase II clinical trial of the GLP-1-based drug Exenatide demonstrated some positive benefits in Parkinson’s disease (see a previous post on this). Thus, raising GLP-1 levels is being further investigated as a future therapy for this condition.
There are two ways of achieving increased levels of GLP-1 though. You could keep injecting synthetic GLP-1 into the body, OR you could simply use DPP-4 inhibitors.
And what are DPP-4 inhibitors?
DPP-4 inhibitors block the enzyme DPP-4 from deactivating the naturally occurring GLP-1 (and all of the other proteins on that list above). And there is a orally administered class of drug, called DPP-4 inhibitors that can do this very well. They allow naturally occurring levels of GLP-1 to rise (as much as twofold).
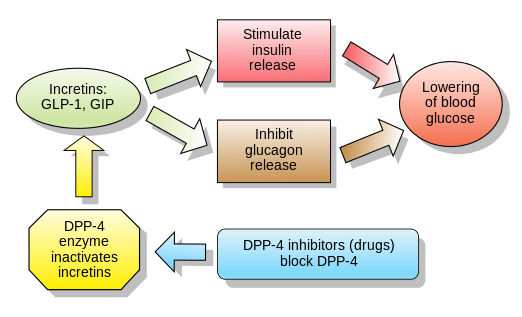
How DDP-4 inhibitors work. Source: Wikipedia
And GLP-1 is not the only interesting protein on this long list of DPP-4 interacting proteins.
Observant readers might have notice another protein called ‘Granulocyte-macrophage colony-stimulating factor‘ (GM-CSF) which we have discussed in a previous post dealing with a clinical trial that was conducted in Nebraska (Click here to read that post). Granulocyte-macrophage colony-stimulating factor stimulates regulatory T cells.
What are regulatory T cells?
Regulatory T cells maintain law and order in the immune system. They do this by enforcing a dominant negative regulation of other immune cells, particularly other T-cells. T-cells are a type of white blood cell that circulate around our bodies, scanning for cellular abnormalities and infections.
Think of T-cells as the inquisitive neighbours curious about and snooping around a local crime scene, and then imagine that Regulatory T cells are the police telling them “nothing to see here, move along”.

Tregs maintaining order. Source: Keywordsuggestions
By stimulating Regulatory T cells, Granulocyte-macrophage colony-stimulating factor is able to reduce the immune systems response to the cell death occurring in the brain (limiting further cell loss). And a recent randomised, double-blind phase I clinical trial of a clinically available drug called Sargramostim – which is a synthetic version Granulocyte-macrophage colony-stimulating factor – demonstrated some positive benefits in people with Parkinson’s disease (Click here to read more on this).
DPP-4 deactivates Granulocyte-macrophage colony-stimulating factor.
Thus by using DPP-4 inhibitors, levels of Granulocyte-macrophage colony-stimulating factor increase:

Title: Dipeptidylpeptidase 4 negatively regulates colony-stimulating factor activity and stress hematopoiesis.
Authors: Broxmeyer HE, Hoggatt J, O’Leary HA, Mantel C, Chitteti BR, Cooper S, Messina-Graham S, Hangoc G, Farag S, Rohrabaugh SL, Ou X, Speth J, Pelus LM, Srour EF, Campbell TB.
Journal: Nat Med. 2012 Dec;18(12):1786-96.
PMID: 23160239 (This article is OPEN ACCESS if you would like to read it)
The investigators that conducted this study looked at Granulocyte-macrophage colony-stimulating factor levels in mice that had no DPP-4 and also in normal mice that had been treated with DPP-4 inhibitors. They found that levels of Granulocyte-macrophage colony-stimulating factor were significantly higher than normal in both sets of mice.
So DPP-4 inhibitors increase levels of naturally occurring GLP-1 and Granulocyte-macrophage colony-stimulating factor.
Two birds, one stone I hear someone in the back row saying?
Ok, but what actual Parkinson’s research has been conducted on DPP-4?
Admittedly, not much (and part of the purpose of this post is to encourage more!). There is a great deal we do not know about this enzyme in Parkinson’s disease. But there have been a few recent studies looking at DPP-4 inhibitors in models of Parkinson’s disease:

Title: Neuroprotective effects of vildagliptin in rat rotenone Parkinson’s disease model: role of RAGE-NFκB and Nrf2-antioxidant signaling pathways.
Authors: Abdelsalam RM, Safar MM.
Journal: J Neurochem. 2015 Jun;133(5):700-7.
PMID: 25752913 (This article is OPEN ACCESS if you would like to read it)
In this study, the Egyptian investigators investigated the anti-Parkinsonian effect of Vildagliptin (a DPP-4 inhibitor) in a rat model of Parkinson’s disease. They randomly allocated rats into three groups – a control group and two additional groups that received the neurotoxin (Rotenone). The control group received no treatment, while one of the neurotoxin groups received Vildagliptin once per day for three weeks. The Vildagliptin treatment markedly improved the motor performance of the rats and resulted in an improvement in dopamine content in the brain. The DPP-4 inhibitor treatment also resulted in reduced levels of inflammation in the brain. Despite observing some weight loss in the Vildagliptin treated animals, the investigators concluded that “Vildagliptin is endowed with various neuroprotective effects and thus can be a promising candidate for the management of Parkinson’s disease”.
And this first result was recently supported by the findings of another study conducted by Jordanian scientists:

Title: Sitagliptin and liraglutide reversed nigrostriatal degeneration of rodent brain in rotenone-induced Parkinson’s disease.
Authors: Badawi GA, Abd El Fattah MA, Zaki HF, El Sayed MI.
Journal: Inflammopharmacology. 2017 Jun;25(3):369-382.
PMID: 28258522
The researchers behind this study conducted a very similar experiment to the previous study, except that they delayed the DPP-4 inhibitor (Sitagliptin) treatment until 16 days after the neurotoxin was given. They also compared the DPP-4 inhibitor Sitagliptin with a drug called Liraglutide which is a GLP-1 agonist similar to Exenatide. They used three different doses of the drugs and found both of the drugs to provide neuroprotective effects – reducing dopamine neuronal loss and markedly reducing inflammation in the brain (admittedly, Sitagliptin was used at much higher doses than Liraglutide in this study).
And the two independent research groups that conducted these studies are not the only folks interested in the idea of using DPP-4 inhibitors for Parkinson’s disease.
The DPP-4 inhibitor Sitagliptin was the topic of a Michael J Fox research grant in 2014 (which was a follow on research grant from a previous MJFF grant looking at the use of Pfizer experimental drug PF-00734,200 – a potent DPP-4 inhibitor – in Parkinson’s disease). The outcome report of that grant suggested were apparently interesting enough that Sitagliptin is now being tested in non-human primate models of Parkinson’s disease. Dr Nigel H Greig of the NIH Translational Gerontology Branch of the National Institute on Aging in Baltimore is named on both of the MJFF research grants and he is also named as one of the investigators in the Exenatide clinical trial research report we have reviewed in a previous post (Click here to read that post). Thus, we will hopefully see a lot more research coming down the pipe very soon further investigating the use of Sitagliptin in Parkinson’s disease.
So should I tell my doctor that I want to start taking DPP-4 inhibitors?
No, you shouldn’t. These potent drugs have not been tested clinically in people with Parkinson’s disease, and we have no idea what they would do. And these drugs are associated with serious side effects.
Adverse effects of DPP-4 inhibitors include:
- gastrointestinal problems – including nausea, diarrhoea and stomach pain
- flu-like symptoms – headache, runny nose, sore throat
- skin reactions – painful skin followed by a red or purple rash
In addition, DPP-4 inhibitors have been linked with an increased risk of pancreatitis (inflammation of the pancreas – Click here to read more about this).
But there are natural forms of DPP-4 inhibitors that have a more mild effect that could be considered.
Natural forms of DPP-4 inhibitors???
Yes, there are natural forms of DPP-4 inhibitors.
Berberine, for example, is a common herbal dietary supplement. It has been shown to lower blood sugar, which it partly does by inhibiting DPP-4:

Title: Inhibition of dipeptidyl peptidase IV (DPP IV) is one of the mechanisms explaining the hypoglycemic effect of berberine.
Authors: Al-masri IM, Mohammad MK, Tahaa MO.
Journal: J Enzyme Inhib Med Chem. 2009 Oct;24(5):1061-6.
PMID: 19640223
Berberine is a bitter-tasting, yellow-coloured compound found in plants. It has a long history of medical use in traditional Chinese and Ayurvedic medicine. Berberine is generally found in the roots and stem bark of many plants, including goldenseal, coptis or goldenthread, Oregon grape, barberry, and tree turmeric. In this study, the investigators found that Berberine readily fit within the binding pocket of of the DPP-4 enzyme, blocking DPP-4 from doing its job. Experimentally, this interaction resulted in significantly reduced levels of DPP-4 activity.
For more information of Berberine (doses and side effects), please click here.
Please see addendum below before asking your physician about Berberine
Another source of DPP-4 inhibition is Lupeol:

Title: Flavonoids with prolyl oligopeptidase inhibitory activity isolated from Scutellaria racemosa Pers
Authors: Marques MR, Stüker C, Kichik N, Tarragó T, Giralt E, Morel AF, Dalcol II.
Journal: Fitoterapia. 2010 Sep;81(6):552-6.
PMID: 20117183
Lupeol is a triterpene (a class of chemical compound) that is found in white cabbage, green pepper, strawberry, olive, mangoes and grapes. In this study, the investigators from Brazil found that the Lupeol exhibits robust DPP-4 inhibitory activity at high concentrations (in the millimolar range).
So what does it all mean?
With the recent excitement surrounding the Exenatide clinical trial results, the last thing I wanted to do was write another Exenatide-related post. But after reading into the topic of DPP-4, I figured that this ‘Exenatide-related’ protein is actually quite interesting and it would be good should share this information.
Drugs blocking the enzyme DPP-4 – which breaks down certain proteins after they have served their function – appear to have beneficial effects on models of Parkinson’s disease and reduce the risk of Parkinson’s disease within one particular population. I would like to reiterate, however, that while DPP-4 is associated with some research suggesting beneficial impact in the area of Parkinson’s disease, more investigations are required. The association between DPP-4 and a reduced risk of Parkinson’s disease within the Swedish diabetic community involved a very small number of cases. Equally, the pre-clinical investigations of DPP-4 inhibitors in animal models of Parkinson’s disease need to be followed up with larger studies by different research groups, investigating many different aspects of their use (such as the effect of longer use of these drugs).
As I mentioned further above, the point of this post was to encourage further investigations within the Parkinson’s research community into this interesting topic. DPP-4 activity affects a lot of different proteins, and as a result DPP-4 inhibitors have some serious side effects. It would be interesting to know, however, if specific functions of DPP-4 could be targeted using carefully designed DPP-4 inhibitors, limiting the number of potential side effects. It’s just an idea.
This is the last Exenatide-related post for a while. Back to other Parkinson’s disease-associated topics in the next post.
ADDENDUM 15/08/2017:
Some readers have pointed out (both in the comment section below and via email) that Berberine is known to have interactions with various medications – some of them serious. They are right, and this could be particularly relevant to Parkinson’s disease so caution should be taken and consulting one’s physician before considering any kind of treatment regime change is essential.
In truth, the research investigating Berberine in models of Parkinson’s disease has been conflicting to date. And it is largely a Korean affair.
In 2010, a research group in Korea published this research report:

Title: Effects of berberine on 6-hydroxydopamine-induced neurotoxicity in PC12 cells and a rat model of Parkinson’s disease
Authors: Kwon IH, Choi HS, Shin KS, Lee BK, Lee CK, Hwang BY, Lim SC, Lee MK.
Journal: Neurosci Lett. 2010 Dec 3;486(1):29-33.
PMID: 20851167
In this study, the investigators found that berberine was having a toxic effect in models of Parkinson’s disease, both cell-culture and rodent models. The researchers treated the rats with berberine once daily for 21 days, and they found a marked reduction in the number of dopamine neurons in the substantia nigra when compared to Parkinsonian rats that did not recieve berberine.
And in to 2013, they followed up that first report with this subsequent study:

Title: Neurotoxic effects of berberine on long-term L-DOPA administration in 6-hydroxydopamine-lesioned rat model of Parkinson’s disease.
Authors: Shin KS, Choi HS, Zhao TT, Suh KH, Kwon IH, Choi SO, Lee MK.
Journal: Arch Pharm Res. 2013 Jun;36(6):759-67.
PMID: 23539311
In this follow-up study, the researchers repeated their previous study with the difference of including Levodopa treatment to determine if this made any difference in the outcome. The investigators modelled Parkinson’s disease in rats and then treated them with berberine and/or Levodopa once daily for 21 days. Again the researchers found that berberine administration to neurotoxin treated animals exaggerated dopamine cell loss when compared to the neurotoxin treated control animals, but with the addition of Levodopa to berberine treatment, the loss of dopamine neurons was made worse.
Sounds bad right?
But several months before that second study was made available, a second Korean research team published a research report that suggested that berberine was actually neuroprotective in models of Parkinson’s disease:

Title: Berberine protects 6-hydroxydopamine-induced human dopaminergic neuronal cell death through the induction of heme oxygenase-1.
Authors: Bae J, Lee D, Kim YK, Gil M, Lee JY, Lee KJ.
Journal: Mol Cells. 2013 Feb;35(2):151-7.
PMID: 23329300 (This article is OPEN ACCESS if you would like to read it)
In this study, the investigators pretreated cells in culture with berberine before exposing them to a neurotoxin (6-OHDA) and they found significantly better survival of cells in the berberine treated group. They found that the positive effect was partly due to the an increase in a protein called heme oxygenase 1 (HO-1), which is known to protect dopaminergic neurons against neurotoxicity (Click here for more on this).
This study was subsequently follow up by a third Korean group who also found the berberine has a positive neuroprotective effect in a mouse model of Parkinson’s disease:

Title: Berberine prevents nigrostriatal dopaminergic neuronal loss and suppresses hippocampal apoptosis in mice with Parkinson’s disease.
Authors: Kim M, Cho KH, Shin MS, Lee JM, Cho HS, Kim CJ, Shin DH, Yang HJ.
Journal: Int J Mol Med. 2014 Apr;33(4):870-8.
PMID: 24535622
The mice in this study were orally administered berberine once a day for a total of 5 weeks, and the researchers found that treatment with berberine enhanced motor skills and coordination by preventing dopamine neuron loss and damage. They concluded that “Based on these data, treatment with berberine may serve as a potential therapeutic strategy for the alleviation of memory impairment and motor dysfunction in patients with Parkinson’s disease”.
Confused? Me too.
A more recent study, however, has highlighted the need for further safety testing of berberine in Parkinson’s disease, especially due to its tendency to accumulate in the brain:

Title: Mitochondria and NMDA receptor-dependent toxicity of berberine sensitizes neurons to glutamate and rotenone injury.
Authors: Kysenius K, Brunello CA, Huttunen HJ.
Journal: PLoS One. 2014 Sep 5;9(9):e107129.
PMID: 25192195 (This article is OPEN ACCESS if you would like to read it)
In this study, the investigators looked at what berberine does inside cells and they found that berberine causes rapid mitochondria-dependent toxicity in neurons, characterised by mitochondrial swelling and dysfunction.
Thus at this point, given the ambiguity in the results from berberine in models of Parkinson’s disease it is probably best to avoid this supplement at least until we have a better idea of what is happening.
EDITORIAL NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. Please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from CSIRO
Berberine. Lupeol. Interesting. I suppose the natural dpp-4 inhibitors are a crap shoot. Unknown quality control, unknown side effects and unknown actual intended effect. Just typing out loud, not asking for recommendations.
LikeLike
Examine.com
Berberine has a high potential to interact with a medications, and some interactions may be serious.
Berberine is one of the few supplements in the Examine.com database with human evidence that establishes it to be as effective as pharmaceuticals
Fwiw
LikeLike
Berberine is POSSIBLY SAFE for most adults for short-term use when taken by mouth or applied to the skin.
Webmd.com
Not sure these comments are appropriate. Feel free to delete.
LikeLike
Awesome post! I’m going to go with: Where there is smoke, there is fire. Hopefully we’ll see more good news from data mining on these populations soon. Seems like we can learn more than waiting until 2020 of so for the next glp-1 trial data. Besides, We need more content for Simon to explain to us. 😊
LikeLike
Hi Double,
Thanks for the comment and kind words. Glad you liked the post. Yeah, the data mining studies of medical records are fantastic. A wealth of information. Lots of other interesting bits and pieces in there that I’ll be posting about in the future. Plus there will be more coming soon on DPP-4 inhibitors.
Kind regards,
Simon
LikeLike
Looking forward to the next update. Also the addendum to this post was very informative. I think I did the same googling as the other folks and had the same question on Berberine.
LikeLike