|
“Repurposing” in medicine refers to taking drugs that are already approved for the treatment of one condition and testing them to see if they are safe and effective in treating other diseases. Given that these clinically available drugs have already been shown to be safe in humans, repurposing represents a method of rapidly acquiring new potential therapeutics for a particular condition. The antidepressant, Trazodone, has recently been proposed for repurposing to neurodegenerative conditions, such as Parkinson’s. In today’s post we will look at what Trazodone is, why it is being considered for repurposing, and we will review the results of a new primate study that suggests it may not be ideal for the task. |
Opinions. Everyone has them. Source: Creativereview
I am regularly asked by readers to give an opinion on specific drugs and supplements.
And I usually cut and paste in my standard response: I can not answer these sorts of questions as I am just a research scientist not a clinician; and even if I was a clinician, it would be unethical for me to comment as I have no idea of your medical history.
In many of these cases, there simply isn’t much proof that the drug/supplement has any effect in Parkinson’s, so it is hard to provide any kind of “opinion”. But even if there was proof, I don’t like to give opinions.
Eleven out of every ten opinions are usually wrong (except in the head of the beholder) so why would my opinion be any better? And each individual is so different, why would one particular drug/supplement work the same for everyone?
In offering an answer to “my opinion” questions, I prefer to stick to the “Just the facts, ma’am” approach and I focus solely on the research evidence that we have available (Useless pub quiz fact: this catchphrase “Just the facts, ma’am” is often credited to Detective Joe Friday from the TV series Dragnet, and yet he never actually said it during any episode! – Source).
Detective Joe Friday. Source: Wikipedia
Now, having said all of that, there is one drug in particularly that is a regular topic of inquiry (literally, not a week goes by without someone asking about): an antidepressant called Trazodone.
What is Trazodone?
Trazodone is an anti-depressant that was first approved for medical use in the United States in 1981.
It is well absorbed after oral administration, and it reaches a mean peak blood levels at approximately one hour after ingestion. Trazodone is a member of a different class of anti-depressant from the tricyclic antidepressants which we recently reviewed in a previous post (Click here to read that post).
Source: Rivopharm
Trazodone is a triazolopyridine-based anti-depressant. Triazolopyridine is a class of antidepressants that are structurally and pharmacologically unrelated to other antidepressants.
Trazodone. Source: Wikipedia
It is marketed around the world under different brands, such as Trittico and Desyrel (worldwide), Molipaxin (UK), and Trazorel & Oleptro (USA).
How does Trazodone work?
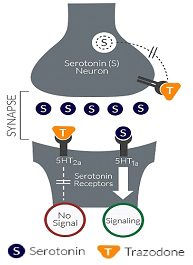
Trazodone is an antidepressant with a dual mechanism of action. Like most anti-depressants, Trazodone blocks the reabsorption of the neurotransmitter serotonin by inhibiting the serotonin transporter (or SERT) – a pump that collects used serotonin from outside of the cell. But Trazodone also binds to and blocks the serotonin type 2 receptor (both the 5-HT2A and 5-HT2C receptors). This ‘mechanism of action’ puts Trazodone into a class of drugs that are referred to as Serotonin antagonist and reuptake inhibitor (or SARI). SARIs leave more of the ‘happy’ neurotransmitter’ (serotonin) floating around in the brain and binding to serotonin type 1 receptors.
How Trazodone works. Source: Tonixpharma
In addition to this serotonergic mechanism, Trazodone has other functions in the body. For example, it is a α1-adrenergic receptor antagonist, and it has weak antihistamine or histamine H1 receptor inverse agonist properties (Click here for a good review on how Trazodone works).
The drug has also recently been shown to have beneficial effects in certain models of neurodegeneration, and this has resulted in the idea that Trazodone could be an interesting drug for repurposing for neurodegenerative conditions, like Parkinson’s.
What is “repurposing”?
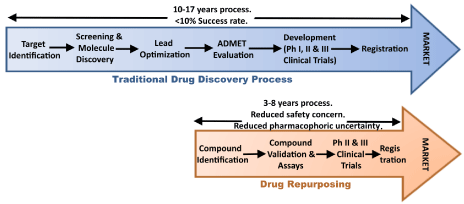
The repurposing of clinically available drugs is the focus of much attention within the Parkinson’s community as it represents a means of bringing new treatments to the clinic faster.
The traditional clinical trial process that is required in the development of new medications takes too long. A new drug to market for neurodegeneration can take up to 15 years to pass through all the phases of development and clinical testing (and some of the trials run over several years each).
Shortening the wait. Source: Austinpublishing
By testing existing clinically approved drugs on conditions like Parkinson’s, this process can be sped up. Given that the safety profile of the drug will be well established after years of clinical usage, repurposing that drug for a new condition basically involves determining whether it can demonstrate efficacy in treating the condition.
A good example of this is the work currently being done on the diabetes drug Exenatide/Bydureon. This drug has been clinically tested on people with Parkinson’s and it has been found to have an effect on the motor features of the condition (Click here and here to read more about this).

A Bydureon pen containing Exenatide. Source: Businesswire
Other clinically available drugs are also being tested on Parkinson’s, such as the respiratory drug Ambroxol. There are two clinical trials testing Ambroxol in Parkinson’s in two different “Londons” (London, UK and London, Canada).
London, Canada. Source: Pinterest
The first study (named AiM-PD) is a phase IIA Prospective, Single-Centre, Open Label Clinical Trial to evaluate the safety, tolerability and pharmacodynamic effects of Ambroxol in Parkinson’s (Click here to read more about this trial). This trial, which is funded by the Cure Parkinson’s Trust and the Van Andel Research Institute(USA), is being conducted at the Royal Free Hospital in London (UK). We are expecting results from the AiM-PD study in 2018.
The second study is being conducted by the Lawson Health Research Institute (and the Weston Foundation) in Canada, and it is a phase II, 52 week trial of Ambroxol in 75 people with Parkinson’s Dementia (Click here to read more about this trial). While this second trial will finish in the second half of 2018, we are not expecting results from this study until 2019.
So why is Trazodone being proposed for repurposing in Parkinson’s?
Midway through 2017, this report was published:

Title: Repurposed drugs targeting eIF2α-P-mediated translational repression prevent neurodegeneration in mice
Authors: Halliday M, Radford H, Zents KAM, Molloy C, Moreno JA, Verity NC, Smith E, Ortori CA, Barrett DA, Bushell M, Mallucci GR.
Journal: Brain, 2017 Jun 1;140(6):1768-1783.
PMID: 28430857 (This article is OPEN ACCESS if you would like to read it)
In this study, the investigators tested 1,040 compounds (that represent many of the clinically available drugs we have) on tiny microscopic worms (called C. Elegans). These worms represent a useful experimental model for screening drugs as many aspects of their biology can be examined. In this study, the worms were exposed to one of the 1040 compounds and a chemical called tunicamycin.
Tunicamycin causes cells to undergo the unfolded protein response – a protective mechanism triggered by rising levels of misfolded proteins, and results in cells shutting protein production, degrading & disposing of any misfolded proteins, and then re-activating proteins involved with increasing the protein production again. When the unfolded protein response is too strong, however, cells will die.
Misfolded proteins are suspected of being responsible for many of the neurodegenerative conditions (such as Parkinson’s), and this may be causing a dysfunctional unfolded protein response. Thus, finding drugs that can control the unfolded protein response could be very useful for future treatment of these conditions.
Of the 1040 compounds tested, the investigators identified five drugs that provided the best protection to the worms:
- Azadirachtin – which is the active ingredient in many pesticides.
- Dibenzoylmethane – a cancer treatment.
- Proguanil – a medication used to treat and prevent malaria.
- Trazodone – an antidepressant used to treat depression and anxiety disorders.
- Trifluoperazine – an antipsychotic of the phenothiazine chemical class.
The investigators decided not to further investigate Azadirachtin as it is a pesticide and displays a poor ability to penetrant the blood-brain-barrier – the protective membrane surrounding the brain. They also rejected Proguanil because while it is safe to use in humans, it is toxic in mice which made further investigation problematic. And finally Trifluoperazine was eliminated as it should not be used in the elderly populations (apparently it ‘increases the risk of death’!), which obviously limited its further utility given that age is a major determinant of neurodegeneration.
This selection process left the researchers with Dibenzoylmethane and Trazodone.
The researchers found that both of these drugs can cross the blood-brain-barrier and were able to prevent neurodegeneration and rescue behavioural deficits in a mouse model of neurodegeneration (prion disease). And they observed no toxic effects of these treatments in other organs (such as the pancreas).
Taking the study one step further, the investigators asked if the drugs could be effective in a model of another neurodegenerative condition, such as Alzheimer’s disease. To investigated this, they treated rTg4510 mice with both of the drugs. rTg4510 mice produce a lot of a human protein (called tau) that has a particular mutation (known as P301L), which results in the onset of Alzheimer’s like pathology at an early age. The rTg4510 mice received either Trazodone or Dibenzoylmethane on a daily basis from 4 months of age and were examined at 8 months of age. The researchers found significantly less cell loss and shrinkage in the brains of the mice treated with one of the two drugs when compared to rTg4510 mice that received no treatment.
The researchers concluded that “these compounds therefore represent potential new disease-modifying treatments for dementia. Trazodone in particular, a licensed drug, should now be tested in clinical trials in patients”.
Naturally these results got a lot of people in the neurodegenerative communities excited and hence the regular stream of emails asking for “an opinion” on Trazodone.
So what research has been conducted on Trazodone and Parkinson’s?
There is remarkably little preclinical data on Trazodone in models of Parkinson’s.
There may well be some research groups looking at it now (given the results in 2017 that we have just discussed above), but most of the work that has been conducted thus far has looked at the effect of Trazodone on specific aspects of Parkinson’s.
Very recently, a research report was published in which the investigators looked at the use of Trazodone in a primate model of Parkinson’s with L-dopa induced dyskinesias:
Title: Trazodone alleviates both dyskinesia and psychosis in the parkinsonian marmoset model ofParkinson’s disease
Authors: Hamadjida A, Nuara SG, Gourdon JC, Huot P.
Journal: J Neural Transm (Vienna). 2017 Dec 15.
PMID: 29247391
In this study (published in December 2017), the researchers investigated the anti-dyskinetic and anti-psychotic effects of Trazodone in a primate model of Parkinson’s. Six marmosets were treated with a neurotoxin (MPTP) that causes the loss of dopamine neurons and appearance of Parkinson’s-like motor issues, and they were then treated with high doses of L-dopa to cause L-dopa-induced dyskinesias. The primates were next treated with Trazodone (at either 0.1, 1 or 10 mg/kg) or a placebo.
The investigators found that co-administration of 10 mg/kg of Trazodone with L-DOPA was able to reduce to peak dose dyskinesias by approximately 40%. BUT, this benefit came at a cost: Trazodone significantly decreased the duration of ON-time (by approximately 20%).
That is to say, Trazodone hindered the ability of L-dopa to correct the motor issues in these primates.
The researchers concluded “that Trazodone may be effective in alleviating L-DOPA-induced dyskinesia and psychosis in Parkinson’s, but its deleterious effect on motor function is a concern and may limit its tolerability and usefulness in clinical settings”.
Previous research suggests that Trazodone has very little affinity for dopamine receptors (Source), so the effect that was observed in this study is unlikely to be due to any kind of dopamine receptor inhibition. Thus, the effect must be coming from other aspect of Trazodone function – exactly how this is working will need to be investigated in future studies.
Beyond this, there is little pre-clinical data for Trazodone in models of Parkinson’s.
Ok, but have there ever been any clinical trials of Trazodone in Parkinson’s?
Yes, there have been several studies and case reports.
There is clinical evidence (case study reports) which suggests that Trazodone could be having an effect on dyskinesias (supporting the primate study mentioned above):
Title: Response of tardive and L-dopa-induced dyskinesias to antidepressants.
Authors: el-Awar M, Freedman M, Seeman P, Goldenberg L, Little J, Solomon P.
Journal: Can J Neurol Sci. 1987 Nov;14(4):629-31.
PMID: 3690436
In this report, the investigators described a 61-year-old man with Parkinson’s, depression and L-dopa-induced dyskinesias. His depression was treated first with desipramine (a tricyclic antidepressant) and later with Trazodone, and it was noted that his dyskinesias improved significantly with both drugs. The doctors speculated that the response of the dyskinesias to the antidepressant medication may have been due to the fact that antidepressants decrease beta-adrenoreceptor sensitivity (we have previously discussed beta-adrenoreceptors and their possible association with Parkinson’s – Click here to read that post). This decrease, in turn, may result in a diminished release of dopamine as beta-adrenoceptors mediate the noradrenaline-stimulated release of dopamine (don’t worry if that sounds complicated, the doctors were just speculating).
A more recent clinical study has also suggested some interesting possibilities for Trazodone:
Title: The use of an antagonist 5-HT2a/c for depression and motor function in Parkinson’ disease.
Authors: Werneck AL, Rosso AL, Vincent MB.
Journal: Arq Neuropsiquiatr. 2009 Jun;67(2B):407-12.
PMID: 19623435 (This article is OPEN ACCESS if you would like to read it)
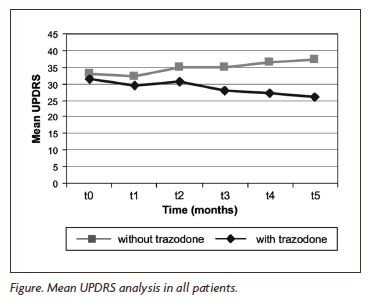
In this study, 20 people with Parkinson’s (with and without depression) were randomly assigned to receive Trazodone (group 1) or not (group 2). Fifteen men and five women participated in the study, aged between 45 and 75 years old. Eight were given the drug (50 mg trazodone orally twice a day), while the other twelve were not treated with anything. The study ran for 5 months and once per month the Parkinson’s features were assessed blindly by two assessors.
All of the participants involved in the study took at least one of the following antiparkinsonian drugs: Carbidopa + levodopa, Amantadine and/or Pramipexole. Unfortunately, in the Trazodone treated group, three patients dropped out during the course of the study and were not included in the analysis performed (two due to sleepiness and one because of postural vertiginous sensation). These drop outs left the study extremely underpowered (with only 5 people in the Trazodone treated group), and it was difficult to conclude much from the final results.
Having said that, there was a trend towards Trazodone stabilising or improving Parkinson’s symptoms over the 5 months of the study (see graph below). Further data analysis, however, demonstrated that Trazodone significantly improved levels of depression, but that the motor function improvements were only observed in the depressed participants.
Their results (and a review of previous work) led the researchers to conclude that “Trazodone significantly improved motor symptoms only in depressed patients”. One important consideration in this study, however, is that it was an “open-label” study for the participants. Those who were being treated with Trazodone knew that they were being treated with Trazodone, while the comparison group knew that they were not receiving any treatment. Thus a placebo effect could have been occurring (in such a situation, the effect may have no pharmacological explanation, and it can only be attributed to the patient’s belief in the treatment).
And this last idea is supported by a previous double blind study in which Trazodone was tested and found to have very limited effect:
Title: Therapeutic effects of trazodone in the treatment of tremor. Multicentric double-blind study
Authors: Sanson F, Schergna E, Semenzato D, Trevisan CP, Bizzarini M, Violante F, Santagostino I, Ravenna C, Maccarone G.
Journal: Riv Neurol. 1986 Nov-Dec;56(6):358-64.
PMID: 3296121
(FULL DISCLOSURE: I do not have access to the full report of this study and I am simply relying on the online abstract and what others have written about it).
In this study, 47 individuals suffering from different types of tremor were blindly treated for 30 days with Trazodone. No significant improvements were detected in individuals suffering from Parkinsonian syndromes, multiple sclerosis, psychogenic tremor, or hyperthyroidism. A significant improvement was, however, observed in 5 out of 6 patients with essential tremor (while the score did not change in the 3 patients with essential tremor who were treated with placebo).
In addition to these studies, there have been other case reports suggesting that Trazodone may actually cause Parkinson’s-like motor-related issues in the elderly.
Such as this one:

Title: Can trazodone induce parkinsonism?
Authors: Albanese A, Rossi P, Altavista MC.
Journal: Clin Neuropharmacol. 1988 Apr;11(2):180-2.
PMID: 3378227
This report was a single case study of a 74 year old lady who developed depression after losing her sister with whom she lived. She was prescribed Trazodone, which was effective in improving her mood. Just several months later, however, she began presenting Parkinsonian symptoms.
It began with the onset of a resting tremor in the left arm, then a slowing of movement and a masking of the face. The attending physician withdrew the Trazodone treatment and within two months the symptoms began to disappear, with no symptoms apparent 12 months later. And unfortunately this is not an isolated case – other periodic reports of Trazodone-induced motor issues have been reported (Click here and here for examples).
Thus, there is currently mixed results with regards the use of Trazodone in Parkinson’s and we may need to proceed with caution in any further testing of this drug in this condition.
So what does it all mean?
This post has been written in response to many inquires regarding the antidepressant Trazodone and its potential use for Parkinson’s.
There has been some preclinical research suggesting that this drug could be useful in neurodegenerative conditions, but to date there has been limited investigations of it in models of Parkinson’s. The clinical data that we have available is mixed and suggestive that some caution is required. Obviously further research is needed before we can provide any definitive ‘opinions’ on Trazodone.
Given how widely Trazodone has been used since it was approved for clinical use in 1981, it would be interesting to search some of the large medical databases and determine whether its use has resulted in a reduction in neurodegenerative conditions such as Alzheimer’s or Parkinson’s (for those prescribed the drug). This analysis will be difficult (due to the potential for other complications in those individuals treated with the drug), but the data may be informative.
In future, when I am asked by email for an opinion on Trazodone, I will point those inquiries towards this page. And if you have received such a response from me and are still reading this, I hope this page has been useful.
Good post?
I look forward to your opinion.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Reference







At least this is an affordable option so why not spend a little more time on it? The analysis you suggest sounds like a good starting point.
LikeLike
Hi Diana,
Thanks for your comment. Yes, it is an affordable option, but there is also a sedative effect with Trazodone that we didn’t get into in this post. It will need a lot more research before I’d be comfortable providing an ‘opinion’ on it.
Kind regards,
Simon
LikeLike
I have a fairly mild essential tremor, and because my wife told me that I sometimes move in my sleep while dreaming (RBD, perhaps) and am a bit of a worry wart, I’ve been evaluated by neurologists several times over the years for Parkinsonian type diseases. So far, nothing, thank God, but I’m writing because my local doctor prescribed Trazadone for sleeping for me. I noticed that when I took it, my anxieties would magically disappear, but the drug would not keep me asleep all night. So, I got the time release version, which works really well, but puts me to sleep all day. My doctor has now given me an alternative, which also works as an antihistamine and helps with my sleep, along with Ambien and melatonin.
I did notice a reduction in my ET while taking Trazadone. I may want to rethink it, if the drug can be proven to be neuroprotective. After all, my dad developed Parkinson’s in his late 70’s, and I’d just as soon avoid it, or something worse. MSA gives me the heebie-jeebies!
LikeLike
Hi WorryWart,
Thanks for the comment and for sharing your experience. As I said in a comment above there is a sedative effect with Trazodone that I didn’t really touch on in the post. But I think a lot more research is required on this drug before we get too excited about it. Thankfully that research is probably already on going. I’ll review it on the site when it becomes available.
Kind regards,
Simon
LikeLike
Thanks, I was one of many asking I think.
LikeLike
I have Parkinson’s and essential tremors I’ve been on trazodone for about six years It must work for me I’ll sleep good with it
LikeLike
Hi Charles,
Thanks for sharing – very interesting.
Kind regards,
Simon
LikeLike
I was diagnosed with PD 3 years ago, For most of that time I’ve been on 5mg of Trazadone nightly. Reading these not very encouraging reports, I began to wheen myself off of it and, in a week was taking very little when my family members told me that my tremor was worse. So now I’m on the full 5 mg again. But I’d like to know what is the dosage for maximum benefit–though few seem ready to say that there is any benefit.
LikeLike
Thomas, sorry about your DX. My Dr. prescribed Trazadone for sleeping (I do not have PD). I take a time released version dosage of 75mg at night, at it keeps me asleep through the morning, as well as canceling out my anxiety attacks that usually wake me up between 3-4 AM. This amount does not cause drowsiness during the day, so I am pretty happy with it.
LikeLike
I have been on Trazadone for almost 14 years as part of my treatment for depression, I started with a small hand tremor a couple of years back. Since increasing my dose now 250mg each night my shaking is really bad and I sleep 12hours each night.
LikeLike
Hello, very nice and complete review, congratulations. Since serotonin medications lower dopamine, isn’t it contra productive to use this kind of medication for Parkinson disease?
Thank you
LikeLike
Hi Simon, I hope you have a moment to read this. First of all thank you so much for offering so much detailed information regarding Trazodone, super informative.
My mom is 90 y/o who was originally diagnosed with Parkinsonism and at this point it was never made clear if she has Parkinsons or Parkinsonism. She takes Carbidopa Levodopa 25mg-100mg. Also, she takes Trazodone 50mg to help her sleep. The last dose of Carbidopa Levodopa is at 7:30pm followed by the Trazodone at 8:30pm. She gets sleepy within 1/2 hour but not enough to fall asleep, even though the Trazodone dosage was increased at one point but did not change anything so we decreased back to 50mg and this same thing that I will describe now happens every night; At about 11:30pm she gets involuntary movements and her left leg is stiff with cramp like symptoms along with up down movements and during this she also has rapid eye movements with lip smacking movements. This goes on for about 25-45 minutes and then she falls asleep. The oddest thing is that this is very punctual at the same starting time anywhere from 11:30pm – 12:30 every night and ending 25-45 min, and then she falls asleep with no further movements until morning hours. I don’t know if she needs the trazodone or not or if she is having withdrawal symptoms from no Carbidopa after 7:30pm. I would greatly appreciate any type of input from you. Thank you very much.
LikeLike