|
Deep brain stimulation (DBS) has now become a standard treatment option for people with Parkinson’s (Click here to read more about DBS), but recently researchers have been investigating a whole new form of stimulation to further help alleviate the symptom of the condition. Spinal cord stimulation – the electrical modulation of the spinal cord – has been tested in models of Parkinson’s in laboratories for the last decade, and this week we saw the publication of the results of a pilot clinical study testing this approach in humans with Parkinson’s. In today’s post we will discuss what spinal cord stimulation is, review the results of this pilot study, and discuss what could happen next for this new treatment approach. |
Source: Videoblocks
2017 was the 200th anniversary of the first report of Parkinson’s by one James Parkinson in 1817 (Click here to read a previous post on this), and the 20th anniversary of the discovery of the first genetic mutation associated with Parkinson’s (Click here to read more about this).
It was also the 50th anniversary of the first use of a technique called spinal cord stimulation.
What is spinal cord stimulation?
Spinal cord stimulation is a form of implantable neuromodulation. Similar to deep brain stimulation (or DBS), it involves using electrical signals to modulate neural activity. But rather than electrodes being placed into the brain (in the case of DBA), spinal cord stimulation involves – as the label on the can suggests – specific areas of the spinal cord being stimulated for the treatment of certain types of pain.
The treatment involves a column of stimulating electrodes that is surgically implanted in the epidural space of the spine. And before you ask: the epidural space is the area between the outer protective skin of the spinal cord (called the dura mater) and the surrounding vertebrae. So the device lies against the spinal cord, and is protected by the bones that make up the spine (as shown in the image below).
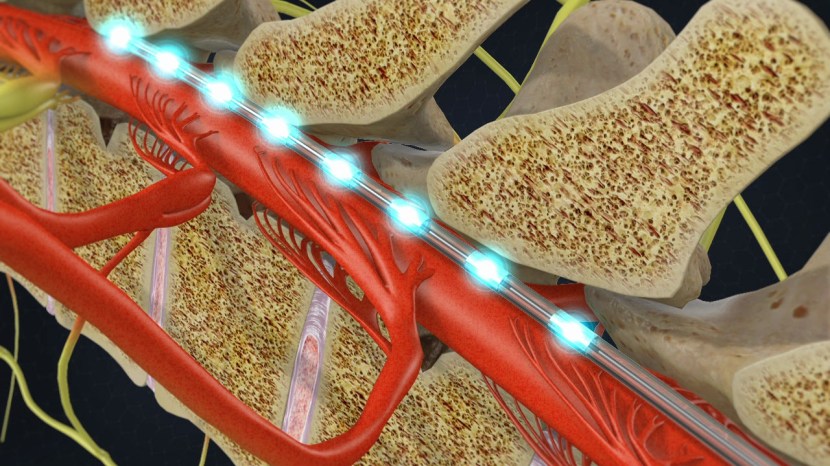
The stimulating electrodes within the epidural space. Source: SpineOne
An electrical pulse generator is implanted in the lower abdomen and conducting wires are connected between the electrodes to the generator. Much like deep brain stimulation, the system is entirely enclosed in the body and operated with a remote control.

An x-ray of the spine with a stimulator implanted (towards the top of the image, and cords leading off to the bottom left). Source:Wikipedia
How does spinal cord stimulation work?
The stimulation basically interrupts the feeling of pain – blocking it from reaching the brain – substituting it with a more pleasing sensation called paresthesia (a kind of tingling or numbness).

Source: MayoClinic
The stimulation does not eliminate the source of pain, it simply masks it by interfering with the signal going to the brain. As a result the amount of relief from pain varies from person to person. In general, spinal cord stimulation resulting in a 50-70% reduction in pain.
But Parkinson’s results from inability to move, how could spinal cord stimulation help in Parkinson’s?
Yeah, this is a good question and the answer is not entirely clear.
The scientists behind the research that we will be discussing below suggest that the beneficial effects from spinal cord stimulation in Parkinson’s could be resulting from activation of the ascending pathways reaching thalamic nuclei and the cerebral cortex. That is to say (in plain English): stimulation of the spinal cord results in a signal going up into the brain where it alters the interaction between two of the regions involved in the initiation of movement (the thalamus and the cortex). And as we shall discuss below, there is evidence backing this idea.
Ok, so how much research has been done on spinal cord stimulation for Parkinson’s?
Actually quite a bit (in fact, for a good early review on the topic – click here).
The first real attempt at spinal cord stimulation in a model of Parkinson’s was reported in this report:

Title: Spinal Cord Stimulation Restores Locomotion in Animal Models of Parkinson’s Disease
Authors: Fuentes, R., Petersson, P., Siesser, W. B., Caron, M. G., & Nicolelis, M. A. L.
Journal: Science (2009) 323(5921), 1578-1582.
PMID: 19299613 (This article is OPEN ACCESS if you would like to read it)
This study was conducted by Prof Miguel Nicolelis and his colleagues at Duke University, and Duke were kind enough to make this short video about the research:
In their study, the scientists injected mice with a drug that reduced the level of dopamine in the brain (the tyrosine hydroxylase inhibitor alpha-methyl-para-tyrosine or AMPT). Similar to Parkinson’s, this resulted in a significant reduction in the ability of these mice to move properly. It also resulted in changes in the neuronal activity patterns of cells in an area of the brain called the motor cortex (we have discussed the motor cortex in a previous post). When the researchers then conducted spinal cord stimulation on these mice, they found that stimulation corrected both the loss of movement and the altered activity in the motor cortex.
The human motor cortex (red). Source: Pinterest
The researchers then tested spinal cord stimulation in rats which had their dopamine system severely depleted (using the neurotoxin 6-OHDA which kills dopamine neurons), and they again found that the treatment could rescue the loss of locomotor ability. Curiously, spinal cord stimulation in the rats also caused an increase in locomotion activity after the stimulation period had stopped – that is to say, when the stimulation was turned off, the rats were still moving around just fine. On top of this, the researchers found that spinal cord stimulation aided the effect of L-dopa treatment in these animals, allowing lower doses of L-dopa to achieve the same behavioural results as higher doses in animals not receiving spinal cord stimulation.
These initial results were then replicated in primates:

Title: Spinal cord stimulation alleviates motor deficits in a primate model of Parkinson disease.
Authors: Santana MB, Halje P, Simplício H, Richter U, Freire MA, Petersson P, Fuentes R, Nicolelis MA.
Journal: Neuron. 2014 Nov 19;84(4):716-22.
PMID: 25447740 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers modelled Parkinson’s in five adult marmosets using the neurotoxin 6-OHDA, which resulted in a reduction in spontaneous behaviour and a significant loss of dopamine neurons in the brain. They then implanted a spinal cord stimulator in each of the animals, which once activated resulted in improvements in Parkinson’s-like features included freezing (31%), hypokinesia (23%), posture (23%), and bradykinesia (21%) – as determined by investigators who were blind to the treatment conditions of each subject.
In the brain, the researchers found that spinal cord stimulation resulted in similar improvements in neural activity as that seen with L-dopa treatment. Given all of these results, the investigators concluded that spinal cord stimulation “should be further tested in clinical studies aimed at measuring its long-term efficacy as a less invasive, long-term therapy for” people with Parkinson’s disease.
And it was not just Prof Nicolelis’ group that has achieved these results. Japanese researchers have also reported spinal cord stimulation having beneficial effects in models of Parkinson’s disease:

Title: Spinal cord stimulation exerts neuroprotective effects against experimental Parkinson’s disease.
Authors: Shinko A, Agari T, Kameda M, Yasuhara T, Kondo A, Tayra JT, Sato K, Sasaki T, Sasada S, Takeuchi H, Wakamori T, Borlongan CV, Date I.
Journal: PLoS One. 2014 Jul 10;9(7):e101468.
PMID: 25009993 (This article is OPEN ACCESS if you would like to read it)
In this report, the researchers actually found that spinal cord stimulation resulted in neuroprotection in a classical model of Parkinson’s disease (rodent 6-OHDA striatal delivery). Across three different levels of stimulation, the researchers reported the rescue of motor deficits and protection of dopamine neurons (particularly for 50Hz stimulation). The researchers also provided evidence suggesting that the neuroprotective effect might have something to do with a protein called Vascular Endothelial Growth Factor (or VEGF). Interestingly, they found that the neuroprotective protein GDNF (that we have discussed before – click here for that post) was not involved.
Interesting. Has anyone ever tested spinal cord stimulation in people with Parkinson’s?
Yes, and this is where we get to the new research. This week, the results of a small clinical study were published:
 Title: Spinal cord stimulation therapy for gait dysfunction in advanced Parkinson’s disease patients.
Title: Spinal cord stimulation therapy for gait dysfunction in advanced Parkinson’s disease patients.
Authors: Samotus O, Parrent A, Jog M.
Journal: Mov Disord. 2018 Feb 14. [Epub ahead of print]
PMID: 29442369
In this pilot study, five males with Parkinson’s suffering from severe gait disturbances and freezing of gait episodes underwent midthoracic spinal cord stimulation for a 1- to 4-month period.
Thoracic vertebrae. Source: Wikipedia
After the operation, clinic visits were made every two weeks for 3 months. At each visit, the subjects were single-blinded and randomly selected given a stimulation combination. Eleven frequency (30, 60, and 130 Hz) and pulse width (200, 300, 400, and 500 microseconds) stimulation combinations were tested (the device did not allow 500 microseconds/130 Hz to be programmed).
At the end of the week 16 visit, the stimulation settings that produce the best motor response were selected by the objective measurements made by the clinical assessors (using gait analysis measures such as Protokinetics Walkway and Unified Parkinson’s Disease Rating Scale (or UPDRS) motor items) and by the participant subjective assessments (such as Freezing of Gait Questionnaire or FOG-Q).
At the 6 months visit, the clinical evaluations and the effects of spinal cord stimulation were collected and assessed. The researchers found that spinal cord stimulation setting combinations of 300-400 μs/30-130 Hz provided gait improvements. They reported that the mean step length, stride velocity, and sit-to-stand improved by 38.8%, 42.3%, and 50.3%, respectively. In addition, the mean UPDRS and FOG-Q scores improved by 33.5% and 26.8% respectively.
Rather impressively, when the investigators looked at the mean number of freezing-of-gait episodes, they found a significant reduction from 16 episodes (pre-surgery) to 0 at 6 months post surgery (and this was while participants were on L-dopa and off stimulation).
The researchers openly admit that this pilot study has flaws in its design. For example, it was open-label and unblinded meaning that the participants involved in the study were aware that they were having spinal cord stimulation. Thus the improvements in motor and gait could have been a placebo response. But they also believe that the results justify a larger phase I/II clinical study which is currently underway in London, Canada. That study will involve 25 participants who will be given dorsal thoracic spinal cord stimulation (I am curious as to why the location of the stimulation in the spinal cord has been shifted from the mid to the dorsal thoracic region). There will be no masking (blinding) in this study either, as this will just be a further characterisation and safety study (Click here to read more about that trial). The results of that study should be available in late 2020.
Importantly, the pilot study demonstrated that spinal cord stimulation is safe for people with Parkinson’s, which agrees with previous individual case studies (Click here, here and here for examples). These pilot results are also in agreement with a previous study of spinal cord stimulation which suggests that beneficial effects could be had from this approach:
 Title: Spinal cord stimulation for the treatment of abnormal posture and gait disorder in patients withParkinson’s disease.
Title: Spinal cord stimulation for the treatment of abnormal posture and gait disorder in patients withParkinson’s disease.
Authors: Agari T, Date I.
Journal: Neurol Med Chir (Tokyo). 2012;52(7):470-4.
PMID: 22850494 (This article is OPEN ACCESS if you would like to read it)
This study investigated the effect of spinal cord stimulation on posture and gait in 15 people (5 men & 10 women) with Parkinson’s in Japan. The results of the study suggested that both posture was improved at 3 months after surgery, while measures of gait and a timed 10-meter walk tests were found to be significantly improved at both 3 months and 1 year after surgery. The researchers concluded that spinal cord stimulation is expected to lead to both the amelioration of pain (a common feature of advanced PD) and improvement of motor function in folks with Parkinson’s.
Further (blinded) research is required to determine just how effective this approach could be.
What does it all mean?
Spinal cord stimulation involves placing electrodes on the spinal column and modulating the neural activity. This approach has been used for many decades to treat chronic pain issues.
Recently spinal cord stimulation has been tested as an experimental approach for alleviating the motor features of Parkinson’s. The results of a recent pilot study suggest that the technique is safe and provide intriguing indications that there could be positive benefits. But further research is certainly required. Critically, the next step will be testing the efficacy of this approach under double blind circumstances.
When such results are available, we will have another look at this stimulating research.
The banner for todays post was sourced from Bostonscientific and paulchristomd



This is fascinating as I’ve been looking at SCS for my husband (PD x 19 years). He had Boston Scientific’s Vercise DBS in 2012; can’t imagine life without it honestly! But his parasomnias and PLMD have been violent enough to cause a rotation in his lumbar spine; SCS would be the answer!
LikeLike
Hi Lisa,
Thanks for your comment – glad you found the post useful. It would certainly be very interesting to test if SCS is an option for folks with restless leg, PLMD, etc. Having said that the technique is still very experimental. If you are interested in more information, you could reach out and contact Dr Mandar Jog who is conducting this research (http://www.uwo.ca/physpharm/jog/). Kind regards,
Simon
LikeLike
Thanks Simon!
BSC have had CE approval for their 1st SCS since 2012, then another later. I don’t really know if hubby would be applicable because of the MRI-guidance v his Vercise, but I am curious with his Pisa syndrome. I have heard of other PwP having SCS over DBS…just can’t track them down at present. I’ll keep you posted. Many thanks!
LikeLike
Hello. I read with a great your articles and i am also reading Dr Miguel nicolelis fameuse book the true creator of eveything. I am french and m’y wife hasard been suffering of parkinson symptômes for 18 years. Couleur you please indicates us people worling on spinal corps stimulation in France or Europe ?
LikeLike