|
# # # #
Recently a lot of media attention has been focused on a new study that reported the replacement of lost dopamine neurons in a mouse model of Parkinson’s, which resulted in the correction the associated behavioural/motor issues.
The researchers involved achieved this amazing feat by simply reducing a single protein in a special type of helper cell in the brain, called astrocytes. By lowering the levels of the protein, they were able to transform the astrocytes into dopamine neurons.
Intriguingly, the study represented independent replication of a previous study that demonstrated a similar result – transformation of astrocytes inside a mouse brain into dopamine neurons by reducing a single protein.
The protein in both studies is called Ptbp1, and in today’s post we will discuss what this protein does, what the new study found, and what the implications of this work could be.
# # # #
|
 Source: Howtogeek
Source: Howtogeek
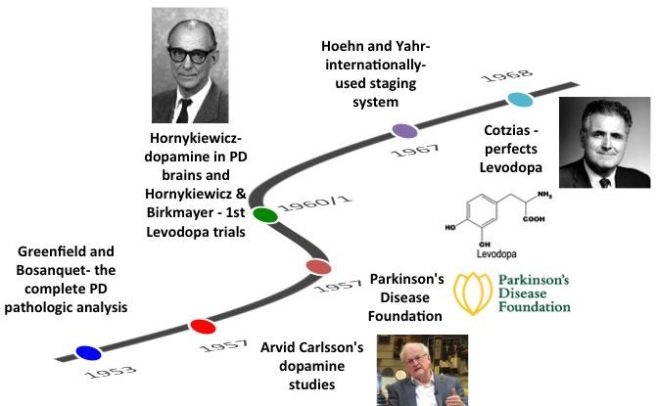
Earlier this year, I stated in my 2020 wish list for Parkinson’s research (Click here to read that post) that one of the big themes I was hoping to see more of was further research on regenerative approaches for the condition.
We have discussed this a few times, but any “curative” treatment for Parkinson’s will require 3 components:
- A disease halting mechanism – to slow/stop the progression of the disease
- A neuroprotective agent – to protect the remaining cells & provide a nurturing environment for,
- Some form of restorative/regenerative therapy – replacing what has been lost
Now the encouraging news is that if you look at the SoPD “The Road Ahead: 2020” post, you will see that there is a great deal of research being conducted on all three of these components at the clinical stage (in addition to vast amounts of work on the preclinical level).
But it is fair to say that the bulk of the clinical research being conducted on restorative therapy for Parkinson’s is centred around the transplantation of stem cell-derived dopamine neurons to replace the cells that have been lost in Parkinson’s (click here to read a recent SoPD post on this topic).
 Embryonic stem cells in a petridish. Source: Wikipedia
Embryonic stem cells in a petridish. Source: Wikipedia
In my wish list for 2020, I was hoping to see regenerative approaches beyond the well trodden path of cell transplantation (growing cells in culture and then injecting them into the brain).
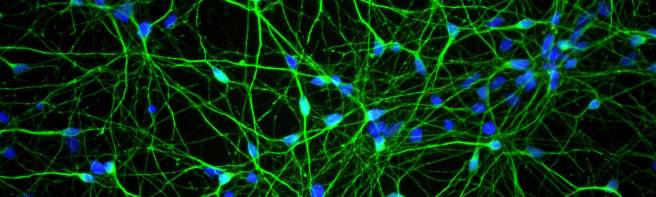 Dopamine neurons (green) in cell culture. Source: Axolbio
Dopamine neurons (green) in cell culture. Source: Axolbio
Rather, I was hoping to see more research on new regenerative approaches that target/manipulate endogenous pathways in the brain – forcing changes within the central nervous system itself.
I didn’t have high expectations in this department, but I have to admit that now I have been pleasantly surprised by the number of research reports that have been published thus far this year highlighting novel regenerative approaches. We have discussed several of them here on the SoPD already (Click here and here for examples), and today we are going to review another which was recently published in the prestigious scientific journal Nature.
This is what all the news papers have been talking about?
Indeed. There has been a lot of media attention focused on this research report.
So what does the new study report?
Continue reading “Ptbp1: “One and done”(?)” →
 Wadlow (back row on the left). Source: Telegraph
Wadlow (back row on the left). Source: Telegraph Source: Businessinsider
Source: Businessinsider