|
Given that the condition is defined by the loss of specific types of neurons in the brain, any future therapy for Parkinson’s should include some form of restorative treatment. Much of the current clinical research exploring restoration in Parkinson’s is focused on cell transplantation – replacing the cells that have been lost in the brain. But the adult brain is very different to the developing brain. While youngsters have lots of nurturing and supportive protein floating around – encouraging plasticity and survival – once we reach adulthood, our brains appear to be full of inhibitory molecules that reduce rejuvenation in the case of injury. What if we could re-introduce some of those supportive factors and remove the inhibitory proteins? Could this help with restorative therapies for Parkinson’s? In today’s post, we will look at new research exploring how we may be able to reduce some of those inhibitory factors and provide a more supportive environment for restorative therapy in Parkinson’s.
|
 Source: restorativejustice
Source: restorativejustice
As we regularly state here on the SoPD, any ‘curative therapy’ for Parkinson’s is going to require three core components:
- A disease halting mechanism
- A neuroprotective agent
- Some form of restorative therapy
Now, the bad news is (as far as I am aware) there is no single treatment currently available (or being tested) that can do all three of these things. By this I mean that there is no disease halting mechanism therapy that can also replace lost brain cells. Nor is there a restorative therapy that stop the progression of the condition.
That statement can obviously be read as bad news, but it shouldn’t.
Let me explain:
A curative therapy for Parkinson’s is going to need to be personalised to each individual, with varying levels of each of the three component listed above. It will be a multi-modal approach designed for each individual’s needs.
 Making things personal. Source: Flickr
Making things personal. Source: Flickr
By this I mean, there is a great deal of heterogeneity (or variability) between individuals with regards to their symptoms and the amount of time that they have had the condition. Some folks are more tremor dominant, while others do not experience tremor at all. Likewise, some individuals have only just been diagnosed, while others have lived with the condition for many years.
The treatment needs of each individual will be different, and thus what we will require is different amounts of the disease halting mechanism component, the neuroprotection component, and the restorative therapy components for each affected person.
In today’s post we are going to explore some alternative approaches being tested for restorative therapy.
What do you mean ‘alternative’?
Most of the ‘restorative’ approaches currently being clinically tested for Parkinson’s focus on replacing the cells that have been lost in the development of Parkinson’s – specifically the dopamine producing neurons.
Researchers have developed protocols for producing vast amounts of dopamine neurons from stem cells grown in cell culture. Millions of stem cells can be grown in petri dishes, providing a ready supply of tissue for transplanting into the brain.
 Embryonic stem cells in a petridish. Source: Wikipedia
Embryonic stem cells in a petridish. Source: Wikipedia
These expanded populations of cells can then be encouraged to become dopamine neurons – a type of brain cell that is badly affected in Parkinson’s – which can then be prepared for transplantation into the brain.
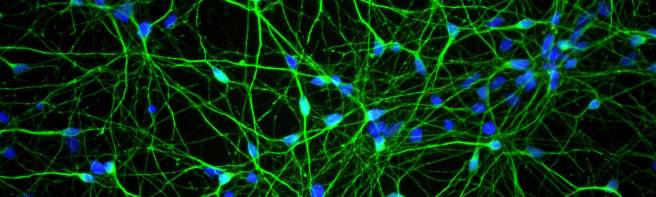 Dopamine neurons (green) in cell culture. Source: Axolbio
Dopamine neurons (green) in cell culture. Source: Axolbio
For the actual transplant procedures, multiple injections of cells are usually made into an area of the brain called the putamen. These multiple sites allow for the transplanted cells to produce dopamine across the entire extent of the putamen. And the cells should remain localised to the putamen, so that they are not producing dopamine in areas of the brain where it is not desired (possibly leading to side effects).

Targeting transplants into the putamen. Source: Intechopen
Postmortem analysis of the brains of individuals who have previously received transplants of dopamine neurons (and then subsequently died from natural causes) has revealed that the transplanted cells can survive the surgical procedure and integrate into the host brain. In the image below, you can see rich brown areas of the putamen in panel A. These brown areas are the dopamine producing cells. A magnified image of individual dopamine producing neurons (their circular bodies and their branches are stained in brown) can be seen in panel B:

Transplanted dopamine neurons. Source: Sciencedirect
The transplanted cells take several years to develop into mature neurons after the transplantation surgery. This means that the actually benefits of the transplantation technique will not be apparent for some time (2-3 years on average). Once mature, however, it has also been demonstrated (using brain imaging techniques) that these transplanted cells can produce dopamine.
As you can see in the images below, there is less dopamine being processed (indicated in red) in the putamen of the Parkinsonian brain on the left than the brain on the right (several years after bilateral – both sides of the brain – transplants):

Brain imaging of dopamine processing before and after a successful transplantation. Source: NIH
For those interested in stem cell research for Parkinson’s, click here to read a previous SoPD post on the topic.
Interesting. But if that is what most ‘restorative’ approaches are doing, what is the ‘alternative’ you were talking about?
So it is one thing to stick cells into the brain to replace the lost population, but what if the brain is not a nuturing environment for such cells? What if the Parkinsonian brain is a difficult situation for transplanted cells to survive in?
Perhaps we need to think about provide a more supportive environment?
Supportive how?
Recently some Australian researchers explored this idea in rodent models of Parkinson’s.
This is the report of their research:
 Title: Viral Delivery of GDNF Promotes Functional Integration of Human Stem Cell Grafts in Parkinson’s Disease.
Title: Viral Delivery of GDNF Promotes Functional Integration of Human Stem Cell Grafts in Parkinson’s Disease.
Authors: Gantner CW, de Luzy IR, Kauhausen JA, Moriarty N, Niclis JC, Bye CR, Penna V, Hunt CPJ, Ermine CM, Pouton CW, Kirik D, Thompson LH, Parish CL.
Journal: Cell Stem Cell. 2020 Feb 10. pii: S1934-5909(20)30010-2.
PMID: 32059808
In this study, the researchers wanted to test whether delivery of a neurotrophic factor called glial cell line-derived neurotrophic factor (or GDNF) could improve the survival and connections of transplanted dopamine neurons.
Hang on a second. What is a neurotrophic factor? And what is GDNF?
Neurotrophic factors (neurotrophic = Greek: neuron – nerve; trophikós – pertaining to food/to feed) are proteins that nurture neurons and support their growth. There are many types of neurotrophic factors, some having more beneficial effects on certain types of neurons and not other. GDNF is one of these neurotrophic factors, and it has very beneficial properties when it comes to dopamine neurons. There is a long history of research on GDNF in the context of Parkinson’s (Click here to read a previous SoPD post on GDNF).
 The structure of GDNF protein. Source: Wikipedia
The structure of GDNF protein. Source: Wikipedia
Given this supportive effect of GDNF on dopamine neurons, the researchers wanted to see if introducing it into a rodent model of Parkinson’s 3 weeks before or 3 weeks after cell transplantation would make any difference regarding how the transplanted cells survive and integrate.
Interestingly, they found that GDNF only enhanced the survival of the transplanted dopamine neurons when it was introduced 3 weeks before the cells were transplanted. GDNF did not increase survival when administered 3 weeks after the transplant.
BUT, the researchers also reported that GDNF only increased connectivity in the brain where the GDNF was given 3 weeks after the transplant took place, not before. That is to say, the presence of GDNF only caused more sprouting of the neurons branches when GDNF was delivered after the neurons were transplanted.
So perhaps one can make the environment in the brain too supportive for the transplanted cells – to the point where they will won’t try too hard to make connections.
Interesting. So what does it all mean?
No, we’re not summing up yet.
There is another alternative route towards restorative therapy for the Parkinson’s brain that I’d like to discuss.
Which is?
It involves removing some of the inhibitory stuff in the brain.
Que?
As mature from childhood to adulthood, our brains become less plastic. Less adaptive. And more effort is required to change in response to experience.
We become fixed in our ways.
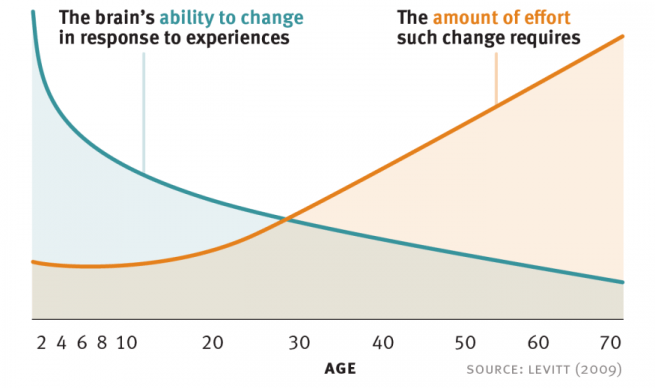 Source: Harvard
Source: Harvard
I guess it is an important part of storing important neurological functions (like the mechanics of how we talk or talk) for long periods of time. Groups of neurons become specialised in tasks and then they become ‘hard-wired’. To stop them from suddenly changing their assigned function, the brain appears to produce lots of inhibitory proteins that reduce plasticity.
One group of these proteins is called Chondroitin Sulfate Proteoglycans (or CSPGs).
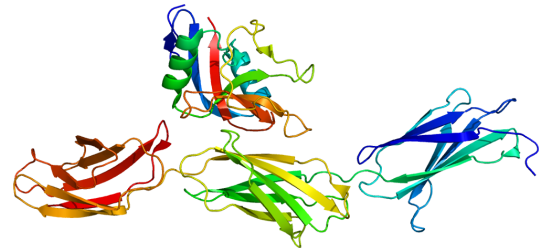 CSPGs. Source: Wikipedia
CSPGs. Source: Wikipedia
CSPGs have a wide range of functions in the brain, but they are known to inhibit the regeneration of neuronal branches (or axons) when they are damaged. And when the brain is injured (such as in the case of a spinal cord injury), support cells in the brain called glial cells will overproduce CSPGs, which reduces the brains ability to repair itself.
And we know this because by introducing an enzyme called Chondroitinase ABC – which breaks down CSPGs – researchers have been able to encourage spinal cord repair (Click here to read more about this).
Interesting. Do you think this Chondroitinase ABC stuff could help in Parkinson’s?
It’s a really good question.
One that has recently been tested – this research report was published at the end of last year:
 Title: Chondroitinase ABC reduces dopaminergic nigral cell death and striatal terminal loss in a 6-hydroxydopamine partial lesion mouse model of Parkinson’s disease.
Title: Chondroitinase ABC reduces dopaminergic nigral cell death and striatal terminal loss in a 6-hydroxydopamine partial lesion mouse model of Parkinson’s disease.
Authors: Fletcher EJR, Moon LDF, Duty S.
Journal: BMC Neurosci. 2019 Dec 20;20(1):61.
PMID: 31862005 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers introduced chondroitinase ABC treatment at the same time as a neurotoxin that causes Parkinson’s-like symptoms in rodents. They used two different models – one partial lesion (to emulate an early stage of PD) and a more severe lesion (replicating later stages of the condition).
The investigators found that the chondroitinase ABC treatment in severe cases provided no beneficial effect, but in the partial model the treatment had a protective effect on the dopamine neurons – in terms of both the number of cells surviving and the preservation of dopamine branches (axons). Unfortunately, this effect did not show itself in any improvement in motor outcomes, but is interesting that removal of CSPGs is having an effect in models of Parkinson’s.
And perhaps removal of inhibitory molecules alone is not enough. Maybe some growth-inducing factors like GDNF are also required – in the form of a combination therapy approach.
Has anyone ever looked at chondroitinase ABC treatment in Parkinson’s models before?
Yes, researchers have.
In 2001, this report was published:
 Title: Regeneration of CNS axons back to their target following treatment of adult rat brain with chondroitinase ABC.
Title: Regeneration of CNS axons back to their target following treatment of adult rat brain with chondroitinase ABC.
Authors: Moon LD, Asher RA, Rhodes KE, Fawcett JW.
Journal: Nat Neurosci. 2001 May;4(5):465-6.
PMID: 11319553
In this study, the reseachers demonstrated that degradation of CSPGs using chondroitinase ABC enhanced regeneration of damaged dopamine fibres in the rat brain. The difference between this study and the more recent study mentioned above is that this older study used physical damage to the neuron to study the effects of chondroitinase ABC, while the more recent study used a neurotoxin.
The results of this older study have been replicated by the same research group (Click here to read more about that) and an independent research group (Click here to read more about that).
Is anyone exploring chondroitinase ABC in the clinic?
Researchers are developing it for potential clinical use, but currently not for Parkinson’s.
Chondroitinase ABC is being actively investigated for spinal cord injury. For example, click here to read about the CHASE IT project, or watch this video:
So what does it all mean?
Elsewhere on this website, we have explored many of the different approaches being taken to try and deal with Parkinson’s. From anti-inflammatory therapies (click here for a SoPD post on this) to spinal cord stimulation (click here to read more about this), there are a lot of varying therapeutic methods being explored.
While this is encouraging, I am always on the look out for new and novel techniques/treatments focused on restorative therapies, which is an area of Parkinson’s research that needs more attention.
Many in the Parkinson’s community have a lot of hope and faith placed in cell transplantation as a means of restorative therapy. But alternative approaches towards this goal warrant more investigation. And as I mentioned on the “2020 wish list” post, the exploration of restorative research will be a theme throughout 2020 .
In this post, we have reviewed enhancing the survival and intergration of transplanted cells using supportive factors like GDNF, and we have considered the removal of inhibitory elements which might be blocking the brain from healing itself. I am intrigued as to how these two approaches could be combined or adapted to encourage more of an effect, and will be watching for further research in this area.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like!
The banner for today’s post was sourced from Youtube

4 thoughts on “A case for chondroitinase?”