|
New research from multiple independent research groups proposes that one Parkinson’s associated protein (LRRK2) may be affecting the activity of another Parkinson’s associated protein (GCase). Specifically, when LRRK2 becomes hyperactive (as is the situation in some cases of Parkinson’s), it causes is associated with a reduction in the amount of GCase activity. In today’s post, we will discuss what LRRK2 and GCase both do, what the new research suggests, and how this news could influence efforts to treat Parkinson’s in the future.
|
 Connections. Source: Philiphemme
Connections. Source: Philiphemme
For a long time, the Parkinson’s research community had a set of disconnected genetic risk factors – tiny errors in particular regions of DNA that were associated with an increased risk of developing Parkinson’s – but there seemed to be little in the way of common connections between them.
 Known genetic associations with PD. Source: PMC
Known genetic associations with PD. Source: PMC
The researchers studied the biological pathways associated with these risk factors, trying to identify potential therapeutic angles as well as looking for connections between them.
The therapies are currently being clinically tested (Click here to read more about these), but the connections have taken a lot longer to find.
Recently one important connection has been identified by several research groups and it could have important implications for how Parkinson’s will be treated in the future.
What’s the connection?
Researchers have found that the Parkinson’s associated protein LRRK2 is influencing the Parkinson’s associated protein GCase.
Ok. Lots of questions here. Let’s start with: What is LRRK2?
Leucine-rich repeat kinase 2 (or LRRK2 – pronounced ‘lark 2’) – also known as ‘Dardarin‘ (from the Basque word “dardara” which means “trembling”) – is an enzyme that has many functions within a cell – from supporting efforts to move things around inside the cell to helping to keep the power on (involved with mitochondrial function).
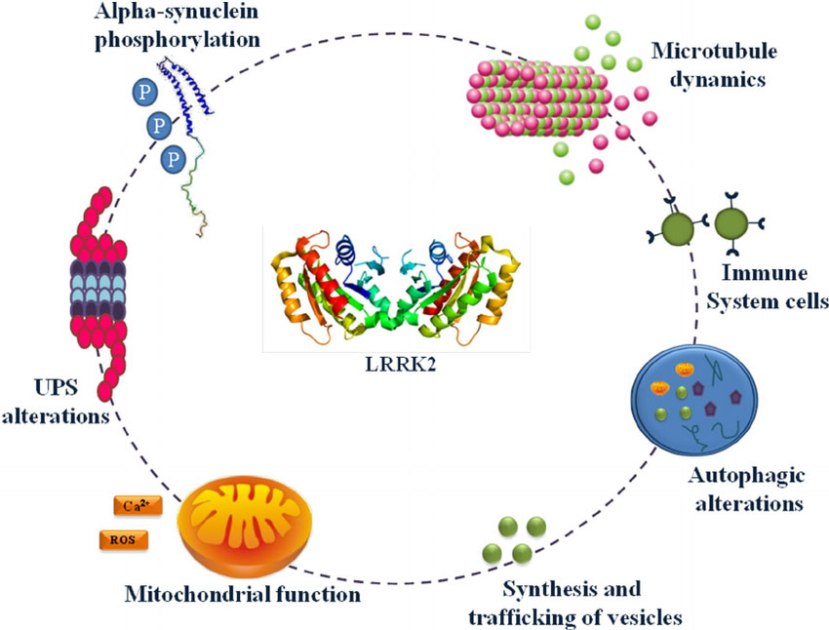
The many jobs of LRRK2. Source: Researchgate
The LRRK2 gene is made up of many different regions. Each of those regions is involved with the different functions of the eventual protein. As you can see in the image below, the regions of the LRRK2 gene have a variety of different functions:
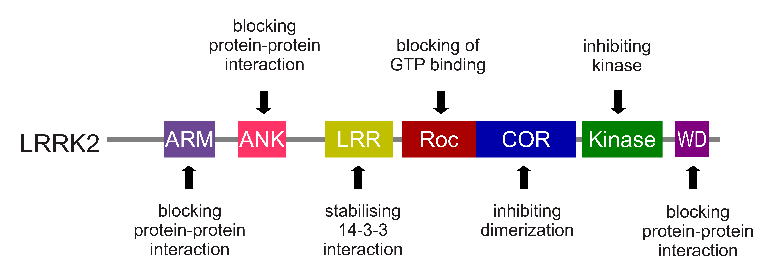
The regions and associated functions of the LRRK2 gene. Source: Intechopen
Genetic errors or variations within the LRRK2 gene are recognised as being some of the most common with regards to increasing ones risk of developing Parkinson’s (LRRK2 variants are present in approximately 1-2% of all cases of Parkinson’s).
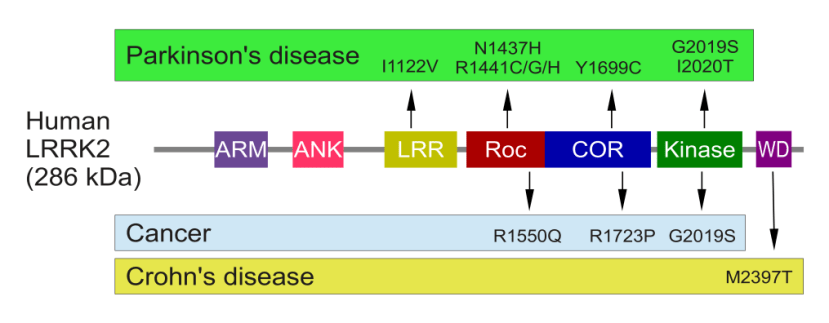
The structure of Lrrk2 and where various mutations lie. Source: Intech
As the image above suggests, mutations in the PARK8 gene are also associated with Crohn’s disease (Click here and here for more on this) – though that mutation is in a different location to those associated with Parkinson’s. And one particularly common Parkinson’s-associated LRRK2 mutation – called G2019S – is also associated with increased risk of certain types of cancer, especially for hormone-related cancer and breast cancer in women – Click here to read more about this. If you have a G2019S mutation, no reason to panic – but it is good to be aware of this association and have regular check ups.
The G2019S mutation (the name designates its location on the gene) is the most common LRRK2 mutation. In some populations of people it can be found in 40% of people with Parkinson’s (Click here to read more about this). But what is interesting about this mutation is that it gives rise to a LRRK2 enzyme that is hyperactive.
 LRRK2 protein. Source: Youtube
LRRK2 protein. Source: Youtube
As a protein, LRRK2 interacts with many different types of other proteins, and you can imagine that in a finely balanced environment like the cells that a mutant ‘hyperactive’ form of LRRK2 is going to cause problems. The consequences of this constantly active form of LRRK2 protein is believed to be influential in the cell death in LRRK2-associated Parkinson’s.
This has led to the development of treatments that inhibit the hyperactive version of LRRK2. Some of these treatments are now being clinically tested, and leading the pack here is a biotech company called Denali Therapeutics.
Founded in 2013, by a group of former Genentech executives, this San Francisco-based biotech company now has two drugs – LRRK2 inhibitors called DNL-201 and DNL-151 – in Phase I safety/tolerability clinical testing for Parkinson’s (Click here and here to read more about these trials).
In addition, a biotech company is called Ionis Pharmaceuticals is developing a novel LRRK2 approach.
Previously known as Isis Pharmaceuticals (until it changed this name in December 2015 when the label ISIS took on negative connotations), Ionis is collaborating with the pharmaceutical company Biogen to target LRRK2 in Parkinson’s.
 The two companies have recently registered a Phase I clinical trial to evaluate their lead candidate, called BIIB094. BIIB094 is an antisense oligonucleotide, which is a method of blocking LRRK2 RNA before it can be used to make LRRK2 protein (we have discussed antisense oligonucleotides in a previous SoPD post – click here to read that post). The trial is a Phase I clinical trial to evaluate the safety and tolerability of single and multiple doses of BIIB094 (Click here to learn more about that trial).
The two companies have recently registered a Phase I clinical trial to evaluate their lead candidate, called BIIB094. BIIB094 is an antisense oligonucleotide, which is a method of blocking LRRK2 RNA before it can be used to make LRRK2 protein (we have discussed antisense oligonucleotides in a previous SoPD post – click here to read that post). The trial is a Phase I clinical trial to evaluate the safety and tolerability of single and multiple doses of BIIB094 (Click here to learn more about that trial).
In addition, pharmaceutical company GlaxoSmithKline has a LRRK2 inhibitor research program and they appear to be gearing up for clinical trials, as they have paired up with genotyping company 23andMe to help identify individuals with LRRK2 genetic variants (Click here to read more about this).
There are also other biotech firms who are pursuing LRRK2 inhibitor research programs, such as Pfizer-spin off Cerevel Therapeutics, San Francisco-based E-Scape Bio (more on them in an upcoming SoPD post), and the French biotech company Servier.
Needless to say, there is a great deal of research focused on LRRK2 inhibition.
|
RECAP #1: LRRK2 is a Parkinson’s-associated gene (functional region of DNA). The LRRK2 gene provides instructions for producing a multi-functional protein. When mutated, the gene provides instructions for a hyperactive form of the LRRK2 protein, which is believed to disrupt normal cellular function. Multiple biotech companies are developing therapies that inhibit the hyperactive form of the LRRK2 protein.
|
Ok, so you said that this Parkinson’s associated protein LRRK2 is influencing another Parkinson’s associated protein?
Yes. There is growing evidence that this is the case.
Late last year, this report was published:
 Title: LRRK2 inhibition prevents endolysosomal deficits seen in human Parkinson’s disease.
Title: LRRK2 inhibition prevents endolysosomal deficits seen in human Parkinson’s disease.
Authors: Rocha EM, De Miranda BR, Castro S, Drolet R, Hatcher NG, Yao L, Smith SM, Keeney MT, Di Maio R, Kofler J, Hastings TG, Greenamyre JT.
Journal: Neurobiol Dis. 2019 Oct 13;134:104626.
PMID: 31618685 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers wanted to characterise the lysosomal and autophagic defects in the surviving dopamine neurons in postmortem human brain samples from people who passed away with Parkinson’s.
Hang on a sec: what is meant by “lysosomal and autophagic defects”?
Autophagy (from the Ancient Greek αὐτόφαγος autóphagos, meaning “self-devouring”) is an absolutely essential function in a cell. Think of it as the waste disposal process in each cell of your body.
Without autophagy, old proteins would pile up, making a cell sick and eventually causing it to die. Through the process of autophagy, the cell can break down the old protein, clearing the way for fresh new proteins to do their job.

The process of autophagy. Source: Wormbook
Waste material inside a cell is collected in membranes that form sacs (called vesicles). These vesicles then bind to another sac (called a lysosome) which contains enzymes that will breakdown and degrade the waste material – the same way enzymes in your washing powder break down muck on your dirty clothes). The degraded waste material can then be recycled or disposed of by spitting it out of the cell.
“Lysosomal and autophagic defects” refer to problems in this complicated, but very necessary process.
The researchers conducting this study found that this waste disposal system was disrupted in postmortem sections of brain from people with Parkinson’s, but also in a rodent neurotoxin (rotenone) model of Parkinson’s.
In both cases, they found reductions in Glucocerebrosidase levels.
Hang on a second. What exactly is gluco…cere…bro…si…dase?
Glucocerebrosidase (also known as GCase) is an enzyme that helps with the digestion and recycling of various proteins (particularly glucocerebrosides) inside cells.
The enzyme is located and active inside small bags of degradative enzymes. These bags are the lysosomes and they can be found floating around inside of cells.
You mentioned lysosomes above. How exactly do these lysosomes work?
On a relatively continual basis, small parts of a cell membrane are being brought inside the cell. This is a process called endocytosis.
It occurs when the cell needs to consume resources from the outside world in order to find what it requires to function and survive. As a section of cell membrane is brought into the cell, it forms what is called a vesicle (which is a term used to refer to small spherical bags of stuff inside cells). Given the process by which these outer membrane vesicles are formed, they is referred to as endosomes (sometimes it is also called a vacuole).
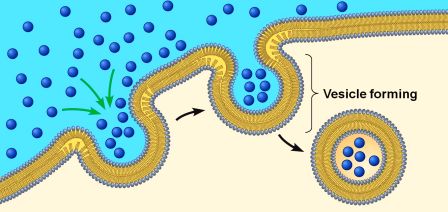 Source: Socratic
Source: Socratic
Once the endosome is inside the cell and detached from the rest of the membrane, it will bind to another vesicle which is called a lysosome. And as I mentioned above, lysosome is a small bag that is full of digestive enzymes, which help to break down the contents of the endosome.

How lysosomes work. Source: Prezi
The lysosome will fuse with the endosome/vacuole and the enzymes from the lysosome will mix with the material in the vacuole and digest it (or it break down into more manageable components).
This enzymatic process works in a very similar fashion to the commercial products that you use for washing your clothes.
 Enzymatic degradation. Source: Samvirke
Enzymatic degradation. Source: Samvirke
The reagents that you put into the washing machine with your clothes contain a multitude of enzymes, which help to break down the dirty, bacteria, flakes of skin, etc that cling to your clothes. Each enzyme breaks down a particular protein, fat or such like. And this is very similar to the collection of enzymes in the lysosome. All of them are needed to break down all of the contents of the endosome.
And if one of those enzymes – such as glucocerebrosidase/GCase – is faulty (due to a genetic mutation), then the enzymatic process is disrupted, which could result in the build up of un-degraded material over time.
Ok, but why did the researchers look at GCase? Why is it important to Parkinson’s?
The enzyme GCase is produced by cells using instructions provided by the GBA gene (a gene is a section of DNA that provides the instructions for making a particular protein).
Genetic variations in the GBA gene are associated with an increased risk of developing Parkinson’s.
 The GBA1 gene and mutations associated with Parkinson’s. Source: Neurology
The GBA1 gene and mutations associated with Parkinson’s. Source: Neurology
In fact, they are one of the most common genetic risk factors for the condition. It is believed that approximately 5%–8% of people with Parkinson’s have a genetic mutation in their GBA gene (Click here and here to read more about this).
Interestingly, this is not the first time researchers have found that people with Parkinson’s but without GBA mutations have lower levels of GCase activity (Click here to read a previous SoPD post about this).
But what they found next was particularly interesting.
|
RECAP #2: Autophagy (the waste disposal system of a cell) is a critical part of cellular function. The researchers reported a disruption to autophagy in both a rodent model of Parkinson’s and postmortem brain tissue from people with Parkinson’s. They found that the Parkinson’s-associated protein glucocerebrosidase (GCase) was also reduced in both cases.
|
What did they find next?
They found that LRRK2 inhibition (that is, blocking the activity of the LRRK2 protein) prevented the autophagy issues in the rodent neurotoxin model.
You will remember we were discussing LRRK2 near the top of this post, how the protein becomes hyperactive in some cases of Parkinson’s, and how the research community is developing/testing pharmacological inhibitors of LRRK2. Well, when the researchers treated their Parkinson’s model with a LRRK2 inhibitor, they found a normalisation of the autophagy problems (such as GCase levels).
This suggested to the researchers that increased LRRK2 activity may impair the autophagy process, resulting in lysosomal dysfunction and causing waste to build up in cells.
Interesting, but does this mean that LRRK2 and GCase are directly interacting?
This is where a second report from an independent research group comes into the story.
Here is that second report:
 Title: LRRK2 kinase activity regulates lysosomal glucocerebrosidase in neurons derived from Parkinson’s disease patients.
Title: LRRK2 kinase activity regulates lysosomal glucocerebrosidase in neurons derived from Parkinson’s disease patients.
Authors: Ysselstein D, Nguyen M, Young TJ, Severino A, Schwake M, Merchant K, Krainc D.
Journal: Nat Commun. 2019 Dec 5;10(1):5570.
PMID: 31804465 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers examined GCase enzyme activity in dopamine neurons derived from people with Parkinson’s. The scientists collected skins cells (fibroblasts) from participants and converted those cells into induced pluripotent stem cells using a method of molecular biology magic (Click here to read a previous SoPD post on this topic).
These patient derived ‘stem cells’ could be grown in cell culture and encouraged to develop into dopamine neurons – a type of brain cell severely affected in Parkinson’s. Some of the cells came from people with LRRK2-associated Parkinson’s, and the researchers found that the LRRK2 mutations resulted in reduced levels of GCase activity in the converted dopamine neurons.
And just like the previous report, when they treated their LRRK2 mutant cells with a LRRK2 inhibitor, the investogators witnessed a restoration of GCase activity.
But then they did the opposite experiment and investigated dopamine neurons derived from people with GBA mutations, and they found that LRRK2 inhibitor treatment in these also……
…..restored of GCase activity.
What?!?
They reported that reducing LRRK2 activity with a LRRK2 inhibitor increased GCase activity in dopamine neurons with GBA mutations. And interestingly, there was no increase in actual LRRK2 activity in the dopamine neurons that carried GBA mutations.
And this correction also rescued some of the Parkinson’s-associated cellular features (such as an accumulation of the protein alpha synuclein). These findings suggested that GCase activity is somehow being negatively controlled by LRRK2 activity.
Further analysis allowed the researchers to determine that this regulation was being controlled by a protein called RAB10.
What is Rab10?
RAB proteins are a group of proteins that sit on the inside of cell membranes. There are many types of RAB proteins and each of them has unique subcellular membrane distribution and take part in various functions.
LRRK2 is known to act on RAB proteins, but particularly RAB10 – and this interaction has been the focus of a lot of recent Parkinson’s research (Click here to read a previous SoPD post on RAB10 which explains how it functions).
Importantly, the researchers in this new study found that increasing levels of RAB10 protein resulted in an increase in GCase activity, regardless of LRRK2 or GBA genetic mutations that cells might be carrying. This finding not only provides a link between two Parkinson’s-associated proteins, but also points to a novel therapeutic angle.
There are already preclinical efforts being made to identify molecules that can increase levels of RAB10 ( (Click here to read more about this), but as discussed at the top of this post there are also clinical trials focused on inhibiting LRRK2 activity. And this new research report provides further evidence that LRRK2 inhibitor therapies may not only be useful for people with LRRK2 genetic variants, but they could also potentially be tested on people with other types of Parkinson’s (perhaps even GBA-associated Parkinson’s).
|
RECAP #3: LRRK2 inhibition was found to restore GCase activity in both of the research reports. The second research report found that a protein called RAB10 may be the connection between the two Parkinson’s-associated proteins. All three proteins are now the targets of therapeutic research for Parkinson’s.
|
So what we require is a therapy that reduces LRRK2 activity. But are there also drugs that can increase GCase levels?
Yes there are.
And as we discussed above there are clinical trial efforts exploring LRRK2 inhibition, but there are also efforts to boost GCase levels as well.
We have recently discussed the results of the ambroxol clinical trial (Click here to read that post), which provided interesting results indicating that this clinically available drug can boost GCase levels.
Ambroxol is a commonly used medication in Europe. It is used for the treatment for respiratory diseases (the respiratory system involving the lungs and related components required for breathing). Ambroxol promotes the clearance of mucus and eases coughing.
 Ambroxol. Source: Skinflint
Ambroxol. Source: Skinflint
FULL DISCLOSURE: The author of this blog is an employee of The Cure Parkinson’s Trust, which co-funded the ambroxol clinical trial (along with the Van Andel Institute and the John Black Charitable Foundation).
In addition, there are biotech firms that have been set up to investigate enhancing GBA activity, such as Lysosomal Therapeutics.
 This company has just completed the Phase 1a clinical studies of their experimental drug LTI-291, which is an activator of the GCase enzyme (Click here to read a previous SoPD post on this topic). LTI-291 is targeted at GBA-associated Parkinson’s, but the first clinical study involved healthy volunteers (Click here to read more about this study – some knowledge of Dutch may help with regards to the details here).
This company has just completed the Phase 1a clinical studies of their experimental drug LTI-291, which is an activator of the GCase enzyme (Click here to read a previous SoPD post on this topic). LTI-291 is targeted at GBA-associated Parkinson’s, but the first clinical study involved healthy volunteers (Click here to read more about this study – some knowledge of Dutch may help with regards to the details here).
The results have not been published yet, but the company’s website suggests that the “study established an excellent safety and tolerability profile in normal human volunteers” (Click here to read more). They have now begun the Phase 1b clinical trial to test safety and tolerability of LTI-291 in 15 people with Parkinson’s who carry a heterozygous mutation in the GBA1 gene (Click here to read more about this trial).
And there is also another biotech company called Prevail Therapeutics.
 This company is taking a gene therapy approach towards treating GBA-associated Parkinson’s (Click here to read a recent SoPD post covering Prevail’s work).
This company is taking a gene therapy approach towards treating GBA-associated Parkinson’s (Click here to read a recent SoPD post covering Prevail’s work).
Both Lysosomal Therapeutics and Prevail have been supported by the Silverstein Foundation – which is focused on finding curative treatments for GBA-associated Parkinson’s (Click here to read more about the Foundation).
And finally (but wait there’s still more), another contender in the GBA-associated Parkinson’s arena is a new Chicago-based biotech company called Surmount Bio.
 Late last year, researchers associated with the company published this report:
Late last year, researchers associated with the company published this report:
 Title: A modulator of wild-type glucocerebrosidase improves pathogenic phenotypes in dopaminergic neuronal models of Parkinson’s disease.
Title: A modulator of wild-type glucocerebrosidase improves pathogenic phenotypes in dopaminergic neuronal models of Parkinson’s disease.
Authors: Burbulla LF, Jeon S, Zheng J, Song P, Silverman RB, Krainc D.
Journal: Sci Transl Med. 2019 Oct 16;11(514). pii: eaau6870. doi: 10.1126/scitranslmed.aau6870.
PMID: 31619543
In this study, the investigators presened a new small-molecule modulator of GCase, which they called S-181. The drug improved the Parkinson’s associated features in human cell culture and mouse models of GBA-associated Parkinsons. Treatment with the drug also improved the GCase functioning of neurons carrying other Parkinson’s-associated genetic variants, such as LRRK2, DJ-1, & PARKIN variants.
I understand that Surmount Bio. has been set up to further develop S-181 for future clinical testing.
So what does it all mean?
There is now a lot of research accumulating that is converging around certain biological pathways associated with Parkinson’s (for example, autophagy and lysosomal dysfunction). But more encouraging is the fact that very specific aspects of those pathways are now being targeted in clinical trials, and over the next few years we should start to see how much of a role some of these proteins may play in the condition.
It is equally pleasing to see that independent research groups are generating data that compliments, supports, and extends on previous findings, even when they come at the problem from different starting points. Things would be much more complicated if the results were differing dramatically.
New research suggests that the Parkinson’s-associated protein LRRK2 is negatively influencing another Parkinson’s-associated protein called GCase (via a third protein called RAB10). Further research in this area is being carefully watched by us here at the SoPD.
Expect updates.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
Further, the author of this post is an employee of the Cure Parkinson’s Trust. The Trust has not asked for this post to be written, and there has been no effort to highlight the work of the Trust over others (perceptions of any bias should be directed to the author). This post has been written by the author solely for the purpose of sharing what the author considers interesting information.
The banner for today’s post was sourced from Sciencefriday




One thought on “Too much LRRK2 begets too little GCase?”