|
Many members of the Parkinson’s community will have heard of deep brain stimulation – a surgical procedure that is offered to individuals with particularly bad tremor or dyskinesias – but there is now another form of stimulation that is now being tested in people with PD. Spinal cord stimulation has long been used as a therapy for back pain and research groups have recently been asking if this technique could be applied to Parkinson’s. In today’s post we will discuss some recently published data that points towards certain aspects of the motor features of Parkinson’s that could benefit from spinal cord stimulation, particularly freezing of gait. |
Deep brain stimulation electrodes implanted in the brain. Source: 2ndFriday
Deep brain stimulation (or DBS) has now become a routine treatment option for those in the Parkinson’s community with particularly debilitating motor features (such as severe tremor or dyskinesias).
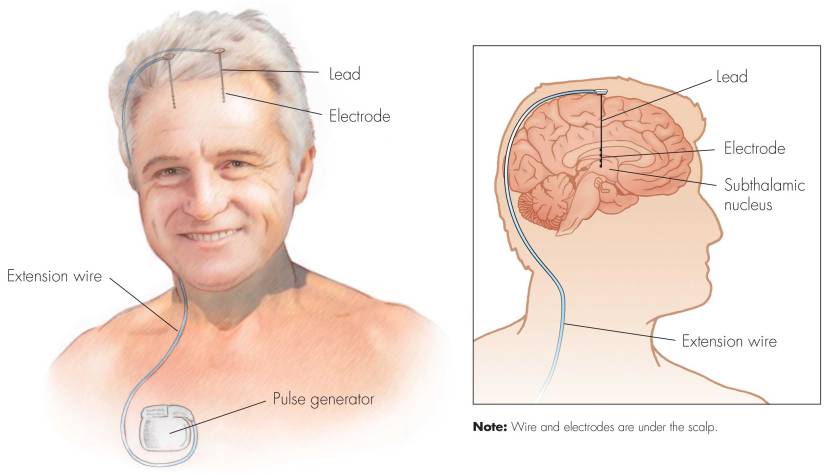
First introduced in 1987, deep brain stimulation consists of three components: the pulse generator, an extension wire, and the leads (which the electrodes are attached to). All of these components are implanted inside the body. Similar to a pace maker for the heart, the DBS system is turned on, programmed and turned off remotely.
Source: Ucdmc
The electrodes that are implanted deep in the brain are tiny, and the very tip of the electrode has small metal plates (each less than a mm in width) which provide the pulses that will help mediate the activity in the brain.

DBS electrode tip. Source: Oxford
Interesting. How does it work?
The electrode extends up into the leads (or extension wire) which continue up and out of the brain, across the top of the skull, down the neck and to the pulse generator which is generally located on the chest.

Xray image demonstrating the leads. Source: Fineartamerica
There are many different brands/types of pulse generators, but they all largely do the same basic function. They are titanium packets containing the electronics and power supply for the leads and electrodes. They are implanted in a subcutaneous spot, usually located under the collar bone on the chest. And some of the batteries in these generators now have 25 year life spans.

Pulse generators. Source: Cambridge (also a good read on DBS)
The pulse generator is programmable (which the doctor usually does several weeks post surgery). A small hand held device is used to turn on/off the generator, generally by holding the device over it.

Remote control. Source: Cambridge
And while all of this is interesting, it is not the topic of today’s post – just the intro. If you would like to read more about DBS and how it affects the brain – click here to read a previous SoPD post on the topic.
So what is today’s post about then?
A different kind of stimulation: Spinal cord stimulation
What is spinal cord stimulation?
Spinal cord stimulation is a form of implantable neuromodulation. Similar to deep brain stimulation, it involves using electrical signals to modulate neural activity. But rather than electrodes being placed into the brain (in the case of DBS), spinal cord stimulation involves – as the label on the can suggests – specific areas of the spinal cord being stimulated. And this has primarily been done in the past for the treatment of certain types of pain.

An x-ray of the spine with a stimulator implanted (towards the top of the image, and connecting cords leading off to the bottom left). Source: Wikipedia
A spinal cord stimulator involves a small device being used to apply pulsed electrical signals to the spinal cord. It is generally used for pain relief, but it has recently been tested in a variety of other medical conditions.
The device is a column of stimulating electrodes that is surgically implanted in the epidural space of the spine. And before you ask: the epidural space is the area between the outer protective skin of the spinal cord (called the dura mater) and the surrounding vertebrae. So the device lies against the spinal cord, and is protected by the bones that make up the spine (as shown in the image below).
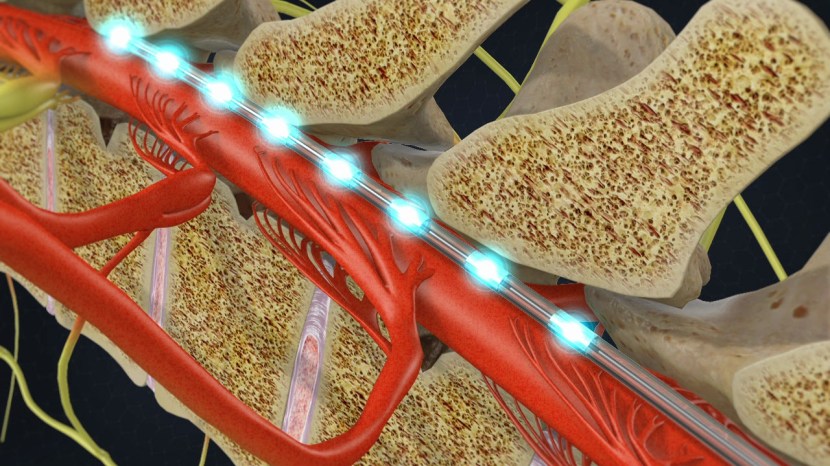
The stimulating electrodes within the epidural space. Source: SpineOne
An electrical pulse generator is implanted in the lower abdomen and conducting wires are connected between the electrodes to the generator. Much like deep brain stimulation, the system is entirely enclosed in the body and operated with a remote control.
How does spinal cord stimulation work?
The stimulation basically interrupts the feeling of pain – blocking it from reaching the brain – substituting it with a more pleasing sensation called paresthesia (a kind of tingling or numbness).

Source: MayoClinic
The stimulation does not eliminate the source of pain, it simply masks it by interfering with the signal going to the brain. As a result the amount of relief from pain varies from person to person. In general, spinal cord stimulation resulting in a 50-70% reduction in pain.
But Parkinson’s results from inability to move, how would spinal cord stimulation work in Parkinson’s?
Yeah, this is a good question and the answer is not entirely clear, but the researchers (behind the research we discuss below) suggest that beneficial effects from spinal cord stimulation in Parkinson’s could be coming from direct activation of ascending pathways reaching thalamic nuclei and the cerebral cortex. That is to say (in plain English): activation of the spinal cord results in a signal going up into the brain where it alters the interaction between two of the regions involved in the initiation of movement (the thalamus and the cortex). And as we shall discuss below, there is evidence backing this idea.
Ok, so how much research has been done on spinal cord stimulation for Parkinson’s?
Actually quite a bit (in fact, for a good early review on the topic – click here).
The first real attempt at spinal cord stimulation for Parkinson’s was this report here:

Title: Spinal Cord Stimulation Restores Locomotion in Animal Models of Parkinson’s Disease
Authors: Fuentes, R., Petersson, P., Siesser, W. B., Caron, M. G., & Nicolelis, M. A. L.
Journal: Science (2009) 323(5921), 1578-1582.
PMID: 19299613 (This article is OPEN ACCESS if you would like to read it)
It was conducted by Prof Miguel Nicolelis and his colleagues at Duke University. Duke were kind enough to make this short video about the research:
In their research report, the scientists injected mice with a drug (the tyrosine hydroxylase inhibitor alpha-methyl-para-tyrosine or AMPT) which reduced the level of dopamine in the brain. Similar to Parkinson’s, this resulted in a significant reduction in the movements of those mice. It also resulted in changes in the neuronal activity patterns of cells in an area of the brain called the motor cortex (we have talked about the motor cortex in a previous post). When the researchers then conducted spinal cord stimulation on these mice, they found that stimulation corrected both the loss of movement and the altered activity in the motor cortex.
The researchers then tested spinal cord stimulation in rats which had their dopamine system severely depleted (using the neurotoxin 6-OHDA), and they again found that the treatment could rescue the loss of locomotor ability. Curiously, spinal cord stimulation in the rats also caused an increase in locomotion activity after the stimulation period had stopped. On top of this, the researchers found that spinal cord stimulation aided the effect of L-dopa, allowing lower doses of L-dopa to achieve the same behavioural results as higher doses in animals not receiving spinal cord stimulation.
These initial results were then replicated in primates:

Title: Spinal cord stimulation alleviates motor deficits in a primate model of Parkinson disease.
Authors: Santana MB, Halje P, Simplício H, Richter U, Freire MA, Petersson P, Fuentes R, Nicolelis MA.
Journal: Neuron. 2014 Nov 19;84(4):716-22.
PMID: 25447740 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers modelled Parkinson’s in five adult marmosets using the neurotoxin 6-OHDA, which resulted in a reduction in spontaneous behaviour and a significant loss of dopamine neurons in the brain. They then implanted a spinal cord stimulator in each of the animals, which once activated resulted in a 200% improvement in some aspects of behavioural activity. Improvements observed in Parkinson’s-like features included freezing (31%), posture (23%), and bradykinesia (21%) as assessed by investigators who were blind to the treatment conditions of each subject.
In the brain, the researchers found that spinal cord stimulation resulted in similar improvements in neural activity as that seen with L-dopa treatment. Given all of these results, the investigators concluded that spinal cord stimulation “should be further tested in clinical studies aimed at measuring its long-term efficacy as a less invasive, long-term therapy for” people with Parkinson’s.
And it was not just Prof Nicolelis’ group that has achieved these results. Japanese researchers have also reported spinal cord stimulation having beneficial effects in models of Parkinson’s:

Title: Spinal cord stimulation exerts neuroprotective effects against experimental Parkinson’s disease.
Authors: Shinko A, Agari T, Kameda M, Yasuhara T, Kondo A, Tayra JT, Sato K, Sasaki T, Sasada S, Takeuchi H, Wakamori T, Borlongan CV, Date I.
Journal: PLoS One. 2014 Jul 10;9(7):e101468.
PMID: 25009993 (This article is OPEN ACCESS if you would like to read it)
In this report, the researchers actually found that spinal cord stimulation resulted in neuroprotection in a classical model of Parkinson’s (rodent 6-OHDA striatal delivery). Across three different levels of stimulation, the researchers reported better rescue of motor deficits and protection of dopamine neurons (particularly for 50Hz stimulation). The researchers also provided evidence suggesting that the neuroprotective effect might have something to do with a protein called Vascular endothelial growth factor (or VEGF). Interestingly, they found that the neuroprotective protein GDNF (that we have discussed before – click here for that post) was not involved.
So has this spinal stimulation procedure ever been conducted in humans with Parkinson’s before?
Yes, it has. But the results have been a bit mixed:

Title: Spinal cord stimulation failed to relieve akinesia or restore locomotion in Parkinson disease.
Authors: Thevathasan W, Mazzone P, Jha A, Djamshidian A, Dileone M, Di Lazzaro V, Brown P.
Journal: Neurology. 2010 Apr 20;74(16):1325-7.
PMID: 20404313 (This article is OPEN ACCESS if you would like to read it)
In this first, very small clinical study, just two people (both 75+ years of age) with Parkinson’s were fitted with spinal cord stimulators. Ten days after the surgery, the subjects participated in a blind analysis of the motor effects of spinal stimulation (blind analysis meaning that the assessors were not aware of their surgical treatment). The assessors, however, found no improvements as a result of the stimulation treatment.
This report lead to a letter to the journal from Prof Nicolelis and his colleagues:

In their letter, Prof Nicolelis and co. point out several issues with the clinical study that may impact the final results (such as the tiny size of the study (only two participants) and the fact that the electrodes were located at a high cervical level, while in the rodent study they were located at a high thoracic level). In addition, the commercially available electrodes used in the human clinical study did not match the relative size or orientation of the electrodes used in the rodent study.
The researchers of the clinical study suggested that the beneficial motor effect described in the rodent study may be due to an increase in arousal (as a result of higher stimulation). But Prof Nicolelis and colleagues pointed out in their letter that their rodent study included three control experiments (including air puffs, trigeminal stimulation at the highest intensity tolerated by the animals, and direct measurements of changes in heart rate following spinal stimulation) which did not find a strong connection between arousal response and recovery seen in the level of locomotion.
The letter concluded that the results of the small clinical trial were inconclusive, and that further research in nonhuman primate models of Parkinson’s are required to determine the effects of electrode design and stimulation parameters. The doctors behind the clinical study agreed that more research is required.
This pilot study demonstrated that spinal cord stimulation is safe for people with Parkinson’s, which was supported by several individual case studies (Click here and here for examples). These pilot results led to a larger study of spinal cord stimulation in Parkinson’s which suggested that beneficial effects could be had from this method:
 Title: Spinal cord stimulation for the treatment of abnormal posture and gait disorder in patients withParkinson’s disease.
Title: Spinal cord stimulation for the treatment of abnormal posture and gait disorder in patients withParkinson’s disease.
Authors: Agari T, Date I.
Journal: Neurol Med Chir (Tokyo). 2012;52(7):470-4.
PMID: 22850494 (This article is OPEN ACCESS if you would like to read it)
This study investigated the effect of spinal cord stimulation on posture and gait in 15 people (5 men & 10 women) with Parkinson’s in Japan. The results of the study suggested that both posture was improved at 3 months after surgery, while measures of gait and a timed 10-meter walk tests were found to be significantly improved at both 3 months and 1 year after surgery. The researchers concluded that spinal cord stimulation is expected to lead to both the amelioration of pain (a common feature of advanced PD) and improvement of motor function in folks with Parkinson’s.
Those early clinical studies were followed more recently by a study conducted by a research group in London (Canada) who published this report earlier this year:
 Title: Spinal cord stimulation therapy for gait dysfunction in advanced Parkinson’s disease patients.
Title: Spinal cord stimulation therapy for gait dysfunction in advanced Parkinson’s disease patients.
Authors: Samotus O, Parrent A, Jog M.
Journal: Mov Disord. 2018 Feb 14. [Epub ahead of print]
PMID: 29442369
In this pilot study, five males with Parkinson’s suffering from severe gait disturbances and freezing of gait episodes underwent midthoracic spinal cord stimulation for a 1- to 4-month period.
Thoracic vertebrae. Source: Wikipedia
After the operation, clinic visits were made every two weeks for 3 months. At each visit, the subjects were single-blinded and randomly selected given a stimulation combination. Eleven frequency (30, 60, and 130 Hz) and pulse width (200, 300, 400, and 500 microseconds) stimulation combinations were tested (the device did not allow 500 microseconds/130 Hz to be programmed).
At the end of the week 16 visit, the stimulation settings that produce the best motor response were selected by the objective measurements made by the clinical assessors (using gait analysis measures such as Protokinetics Walkway and Unified Parkinson’s Disease Rating Scale (or UPDRS) motor items) and by the participant subjective assessments (such as Freezing of Gait Questionnaire or FOG-Q).
Protokinetics Walkway. Source: Protokinetics
SIDE NOTE: Related to self monitoring – I really like this form of gait monitoring: a mat that measures all aspects of how one walks and feeds that information to a nearby computer.
At the 6 months visit, the clinical evaluations and the effects of spinal cord stimulation were collected and assessed. The researchers found that spinal cord stimulation setting combinations of 300-400 μs/30-130 Hz provided gait improvements. They reported that the mean step length, stride velocity, and sit-to-stand improved by 38.8%, 42.3%, and 50.3%, respectively. In addition, the mean UPDRS and FOG-Q scores improved by 33.5% and 26.8% respectively.
Rather impressively, when the investigators looked at the mean number of freezing-of-gait episodes, they found a significant reduction from 16 episodes (pre-surgery) to 0 at 6 months post surgery (and this was while participants were on L-dopa and off stimulation).
The researchers openly admit that this pilot study has some flaws in its design. For example, it was open-label and un-blinded, meaning that the participants involved in the study were aware that they were having spinal cord stimulation. Thus the improvements in motor and gait could have been a placebo response.
But they also believe that the results justify a larger phase I/II clinical study which is currently underway in Canada. That study will involve 25 participants who will be given dorsal thoracic spinal cord stimulation (I am curious as to why the location of the stimulation in the spinal cord has been shifted from the mid to the dorsal thoracic region). There will be no masking (blinding) in this study either, as this will just be a further characterisation and safety study (Click here to read more about that trial). The results of that study should be available in late 2020.
So what was the research published this week?
This report was published:
Title: Effects of spinal cord stimulation on postural control in Parkinson’s disease patients with freezing of gait.
Authors: de Lima-Pardini AC, Coelho DB, Souza CP, Souza CO, Ghilardi MGDS, Garcia T, Voos M, Milosevic M, Hamani C, Teixeira LA, Fonoff ET.
Journal: Elife. 2018 Aug 2;7. pii: e37727.
PMID: 30070204 (This report is OPEN ACCESS if you would like to read it)
This study was an international collaboration between Brazilian and Canadian researchers who desired to assess the effects of spinal cord stimulation on freezing of gait and certain aspects of postural control.
What is freezing of gait?
I have briefly mentioned freezing of gait (or FOG) above, but I didn’t really explain it.
Freezing of gait is a sudden disturbance or interrupting of walking. Specific to the issue is the sudden and transient nature of this debilitating aspect of Parkinson’s. During an episode of FOG, an individual often perceives complete inability to continue with locomotion (which can result in falls).
This video (kindly provided by the medical journal the Lancet) offers examples of an individual suffering from freezing of gait:
The researchers recruited four individuals with Parkinson’s who suffered badly from episodes of freezing of gait. These participants had been previously implanted with a spinal cord stimulation system in the upper thoracic spine as part of another study (Click here to read about the study and click here to read more about the results of that study). Anticipatory postural adjustment (APA), reactive postural responses, gait and FoG were then biomechanically assessed
What are Anticipatory postural adjustment and reactive postural responses?
A necessary aspect of normal walking is an exact coordination of postural adjustment in advance of each step. This is anticipatory postural adjustment. During an episode of FOG, the intention to walk becomes uncoupled from the anticipatory postural adjustment, which results in a failure to move forward. This often shows itself in knee trembling and difficulty initiating a step.
Reactive postural control is simply the ability to recover from a a disruption of stability, and it ultimately determines whether an individual will fall following the freezing of gait or a loss of balance.
The results of the analysis in these four individuals implanted with spinal cord stimulators suggested that spinal cord stimulation improved both freezing of gait and anticipatory postural adjustment, but it failed to improve reactive postural responses. The researchers speculated about whether this difference could tell us something about the biology of movement and the nature of spinal cord stimulation, specifically that freezing of gait may be a cortical-based phenomenon (which benefits from spinal cord stimulation), while reactive posture control relies more on cell populations in the brain stem (which is less affected by spinal cord stimulation). This needs to be further analysed though.
So what does it all mean?
Researchers have been investigating the use of spinal cord stimulation for people with Parkinson’s and they have found that this technology may have benefits for certain aspects of the motor features of the condition.
While there is a mad dash for ‘curative’ treatments for Parkinson’s, it is also very encouraging to see new methods coming through that can provide benefits for very specific aspects of PD. Spinal cord stimulation has previously been used for pain relief, but if this technology can help people suffering from freezing of gait then it should definitely be investigated further.
The banner for today’s post was sourced from Hawthorncenter




Simon
Would external stimlation by a TENS machine help with FOG?
regards
Keith
LikeLike
Hi Keith,
It’s a great question, and one that has sort-of been looked into. There were some Swedish researchers several years back that looked at whether a device similar to the TENS could help with balance (https://www.reuters.com/article/us-sweden-parkinsons-device-idUSKBN0M512020150309). They published their results in 2015 (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5386963/) but I’m not sure if this has been followed up, or applied to FOG. I will go away and do some investigative work.
Thanks for the interesting question.
Kind regards,
Simon
LikeLike
Simon,
Thanks for writing this article. To be honest, very little progress appears to have been made over the last 10 years, or so.
In fact, the situation is probably worse than that. Back in 1892 Charcot wrote a paper (reported in [1]): “Charcot advocated vibratory therapy for the management of Parkinson’s disease. Charcot had observed that after long carriage, train, or horseback rides, patients with Parkinson’s disease experienced marked symptom amelioration. … Electrical stimulation … therapy was used to stimulate weakened muscles”
The slow progress so far leaves me considering DIY. And here Keith’s question is very important: can you get some benefit from using external electrical stimulation? I would probably start with haptic stimulation using small vibrating motors, linked to an Arduino to generate the pulses. You could probably build a prototype for about £20.
An even simpler approach is to stand on a vibrating board or, even, to sit on a washing machine in its spin stage.
Reference
[1] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3234454/
John
LikeLike
Hi John,
I like the washing machine spin cycle idea (dare I ask you have tried?). See the ‘stochastic vestibular stimulation’ research in my response to Keith. Having just had a quick search, there appears to have been quite a bit of this sort of research (for example, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4313776/ and more recently https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5835530/). Professor Bergquist and his team have gone quiet of late, but others are also looking into it. This will require some time to look into, which at present I have very little free. But once I have a better handle on it, there may be a post on this topic. Interesting stuff.
Charcot’s observations and ideas are still brilliant.
Kind regards,
Simon
LikeLike
Simon,
I read some place recently that Parkinson disease that is induced in animal models by neurotoxines or by genetic manipulation was cured hundreds of times. Yet these treatments failed to produce even a single disease modifying solution in humans. I can’t help it but wonder if what you described here is yet another illustration of the same issue. Which brings us to the quote commonly attributed to Einstein: “The definition of insanity is doing the same thing over and over again, but expecting different results”.
On a different note, if it is not too much trouble could you please point out a good reference that explains how Levodopa actually work in the brain to temporarily improve PD symptoms? L-dopa that is absorbed and ends up in the brain is not confined to the synaptic connections of motor neurons where one would think it is needed. So how does this “flood” of LDopa from Parkinson medication help?
Thanks,
Felix
LikeLike
Hi Felix,
Thanks for your comment and the interesting questions.
Yes, life would be great if all these remedies translated to humans much easier. Part of the problem is that we don’t have a good model of Parkinson’s (the neurotoxins etc just don’t replicate the condition properly). And the other part of the problem is that the experimental therapies are being administered in tightly controlled settings of a lab experiment, before or just after the neurotoxin is being delivered – thus there is the opportunity for dying dopamine cells (for example) to survive. In the reality of the clinic, when a person is diagnosed with PD they have already lost maybe 50% of those dopamine neurons. There needs to be a shift in the research towards remedies that deal with this issue.
As for an explanation of what L-dopa is, I wrote a post back in 2016 about the new inhalable L-dopa treatment being rolled out at the moment. That post explains what L-dopa is (with a bit of history): https://scienceofparkinsons.com/2016/10/26/inhaling-l-dopa/. This video explains the biology/pharmacology of L-dopa quite well.
I hope this helps.
Kind regards,
Simon
LikeLike
Simon, thank you for your response. The point you make about the time constraints of the neurotoxin approach sounds like a very logical one. In my (layman’s) view Parkinson can be divided into two parts: symptomatic (related to the lack of dopamine that is the result of cell death and dysfunction), and causal (related to that mysterious reason or reasons that is causing these cells to malfunction and die). The neurotoxin model would be great for the first part but it doesn’t appear to do much for the second. As you mentioned in your numerous posts there are many theories of what is causing Parkinson yet none was proven as true. If I understand it right there are things that are PROVEN to be true that somehow relate to that cause. It is clear that there is some inflammation process in the brain going on and there is prion like process that is propagated by misfolded ASNs. Please correct me if I am mistaken but from what I’ve read the inflammation in the brain is not fully understood. For obvious reasons immune response in the brain is very different from the same in other organs. I am surprised that no numerical bio-marker was found to indicate the direction and severity of this response in PD. My neurologist always makes fun of my fondness for numbers saying that it is an engineer in me talking. Well in our part of the world we say that numbers don’t lie (which we all know is not true 🙂 and it is always easier to reach your goal if you have numerical compass. I also understand (and it makes me cautiously optimistic) that we can modify and even cure PD even if we never find out the true cause of it.
Also thank you for the the L-Dopa post reference. It has some very interesting historical data. If I am not mistaken the inhaler is 2-3 months away from FDA approval and it would be good to understand how it works. I am still at a loss HOW l-dopa is actually helping with the symptoms. This is not just idle curiosity: when I take my l-dopa medication in the morning within 15 minutes I fill nauseous which tells me that it reached my brain. Yet it takes another 40 minutes before I can move. Also if I have any animal protein after I take my medication it stops working. If I knew how l-dopa actually helps with the PD symptoms may be I could make it work better for myself.
Thank you again for maintaining this very,very valuable resource.
Felix
LikeLike