|
# # # # Neurotrophic factors – like Glial cell line-derived neurotrophic factor (or GDNF) – hold great hope for regenerative therapy in Parkinson’s research. New research, however, indicates that simply injecting the protein into the brain may not be enough. Scientists at Rush University Medical Center (in Chicago) conducted a postmortem analysis of brains from people who passed away with Parkinson’s and made an intriguing discovery. They found that many of the remaining dopamine neurons appear to not be producing a protein called Ret, which is required for GDNF signaling. In addition, other components of GDNF signaling pathway were missing. In today’s post, we will review the background of this new study, outline what the study found, and discuss the implications of the research. # # # # |
GDNF. Source: Wikipedia
Glial cell line-derived neurotrophic factor (or GDNF) is a topic that gets a lot of reader attention on the SoPD. It is a tiny protein that holds great hope for the Parkinson’s community in terms of providing a potential neuroprotective and regenerative therapy.
GDNF is a type of neurotrophic factor, which are small naturally-occurring proteins that nurture neurons and support their growth. There are different kinds of neurotrophic factors, and the testing of some of them in preclinical models of Parkinson’s has generated encouraging results (particularly in the case of GDNF – click here to read a previous SoPD post on this topic).
But the translation of those initial results in cell culture and animal models of Parkinson’s has been difficult in clinical trials of neurotrophic factors.
This has led to many questions being asked within the research community about the nature of biological signaling pathways involved with neurotrophic factors and whether they might be affected in Parkinson’s.
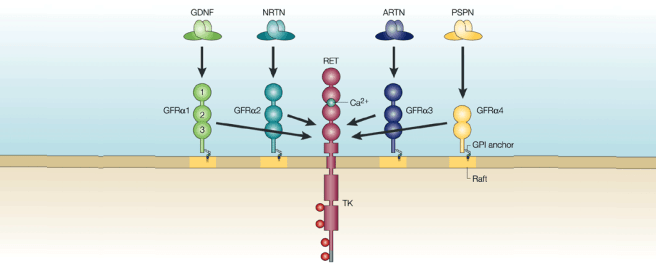
The majority of the neurotrophic factors that have been tested in models of Parkinson’s and in clinical trials for Parkinson’s belong to a branch that requires the RET signaling pathway to be available to have their neuroprotective effect.
What is the RET signaling pathway?