|
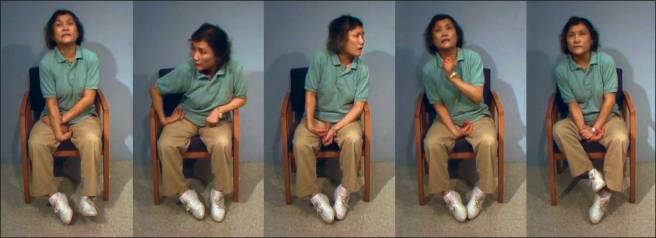
# # # # Dyskinesias are involuntary muscle movements associated with long-term use of levodopa therapy (use of levodopa is not a certainty for developing dyskinesias, but there is an association). A better understanding of the underlying biology of dyskinesias is required in order to alleviate this condition for those affected by it. This week researchers reported that a single protein – called RasGRP1 – plays a central role in the development of dykinesias, raising hope that agents targeting this protein could identified and provide better quality of life of sufferers. In today’s post, we will discuss what dyskinesias are and review the new research. # # # # |
Few people outside of the biomedical sciences may have heard of the Scripps Research Institute, but it is the largest private, not-for-profit medical research facility in the United States and among the largest in the world. It is headquartered in La Jolla, California but it has a sister facility in Jupiter, Florida.
 Nice spot to do research. Source: Scripps
Nice spot to do research. Source: Scripps
Collectively, “The Scripps” has 250 laboratories, which employs over 2,400 scientists, technicians, graduate students, and administrative staff.
It was founded in 1924 by journalist/philanthropist Ellen Browning Scripps.
 Ellen Browning Scripps. Source: Lajollalight
Ellen Browning Scripps. Source: Lajollalight
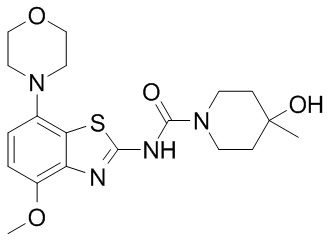
The Scripps covers a wide variety of area in biomedical research, but this week a group of researcher led by scientists at the Florida Scripps institute published an interesting report on Parkinson’s:
 Title: RasGRP1 is a causal factor in the development of l-DOPA–induced dyskinesia in Parkinson’s disease
Title: RasGRP1 is a causal factor in the development of l-DOPA–induced dyskinesia in Parkinson’s disease
Authors: Eshraghi M, Ramírez-Jarquín1 UM, Shahani1 N, Nuzzo T, De Rosa A, Swarnkar S, Galli N, Rivera O, Tsaprailis G, Scharager-Tapia C, Crynen G, Li Q, Thiolat ML, Bezard E, Usiello A, Subramaniam S
Journal: Science Advances, May 2020, 6, 18, eaaz7001
PMID: 32426479 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in proteins that could be playing a major role in the development of dyskinesias.
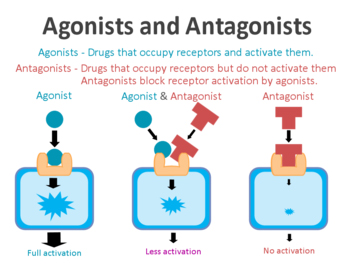
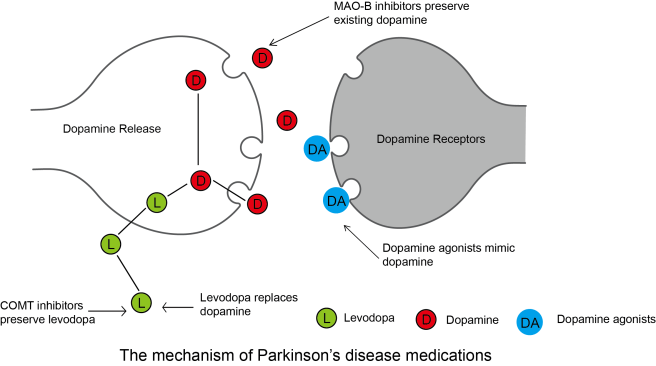
What are dyskinesias?