|
The biotech company Acorda Therapeutics Inc. yesterday announced that it was halting new recruitment for the phase III program of its drug Tozadenant (an oral adenosine A2a receptor antagonist). In addition, participants currently enrolled in the trial will now have their blood monitoring conducted on a weekly basis. The initial report looks really bad (tragically five people have died), but does this tragic news mean that the drug should be disregarded? In todays post, we will look at what adenosine A2a receptor antagonists are, how they may help with Parkinson’s, and discuss what has happened with this particular trial. |
Dr Ron Cohen, CEO of Acorda. Source: EndpointNews
Founded in 1995, Acorda Therapeutics Ltd is a biotechnology company that is focused on developing therapies that restore function and improve the lives of people with neurological disorders, particularly Parkinson’s disease.
Earlier this year, they had positive results in their phase III clinical trial of Inbrija (formerly known as CVT-301 – Click here to read a previous post about this). They have subsequently filed a New Drug Application with the US Food and Drug Administration (FDA) to make this inhalable form of L-dopa available in the clinic, but the application has been delayed due to manufacturing concerns from the FDA (Click here to read more about this). These issues should be solvable – the company and the FDA are working together on these matters – and the product will hopefully be available in the new year.
So what was the news yesterday?
Acorda Therapeutics has another experimental product going through the clinical trial process for Parkinson’s disease.
It’s called Tozadenant.
Source: Focusbio
Tozadenant is an oral adenosine A2a receptor antagonist (and yes, we’ll discuss what all that means in a moment).
Yesterday Acorda Therapeutics Inc announced that they have halted new recruitment for their phase III clinical program. In addition the company is increasing the frequency of blood cell count monitoring (from monthly to weekly) for participants already enrolled in the company’s Phase 3 program of Tozadenant for Parkinson’s disease.
The Company took this action due to reports of cases of agranulocytosis.
What is agranulocytosis?
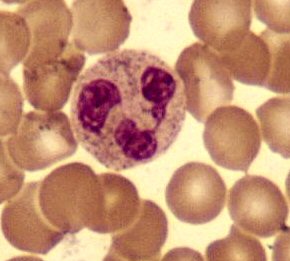
The big cell in the middle is a neutrophil granulocyte. Source: Wikipedia
Granulocytes are a type of white blood cell that help the body fight infection. ‘Agranulocytosis’ is a term used to describe a deficiency of these cells. A reduction in the body’s granulocytes can lead to Septicemia (also known simply as Sepsis). Acorda reported seven cases of sepsis within their clinical trial program (all in the Tozadenant treated groups), and five of those cases of sepsis were fatal.
What is Sepsis?
Your immune system does a pretty amazing job of keeping infections localised to one area of the body. White blood cells (like granulocytes) are produced and rush to the site of the infection to deal with the pathogen causing trouble. Upon arrival at the site of trouble, the white blood cells release chemicals that communicate the extent of the infection/damage to the rest of the immune system and body. This process is known as inflammation.
Now, if your immune system is weak or the infection is particularly severe, it can quickly spread to other parts of the body. Such a situation can cause the immune system to go completely bananas (highly technical term). The system goes into overdrive, and the inflammation can affect the entire body. This can cause organs to receive insufficient blood supply or fail completely.
This end state is what we call Sepsis – when the body’s response to infection causes injury to its own tissues and organs.
A disturbing fact: Sepsis is responsible for more deaths each year in the UK than bowel, breast and prostate cancer combined (Source).
What causes Agranulocytosis?
So if you read just the shock-horror media headlines about this news story (for examples, click here and here), you would be forgiven for thinking that a potentially useful drug like Tozadenant is heading for the trash can of clinical trial history. But there is actually a large number of widely available drugs that have been associated with agranulocytosis.
These include:
- Antiepileptics (such as carbamazepine and valproate)
- Antithyroid drugs (carbimazole, methimazole, and propylthiouracil)
- Antibiotics (penicillin, chloramphenicol and co-trimoxazole)
- ACE inhibitors (benazepril), cytotoxic drugs,
- NSAIDs (indomethacin, naproxen, phenylbutazone, metamizole), mebendazole, allopurinol)
- Antidepressants (mianserin and mirtazapine)
- Antipsychotics (particularly the atypical antipsychotic clozapine – Clozapine users in the United States, Australia, Canada, and the UK must be nationally registered for monitoring of low white blood cell counts) – (Source: Wikipedia)
So what is an oral adenosine A2a receptor antagonist?
An antagonist is a drug that binds to and blocks the activation of a particular receptor.
On the surface of a cell, there are lots of receptors which act as switches for certain biological processes to be initiated. Receptors will wait for a protein to come along and activate them or alternatively block them. The activators are called agonists, while the blockers are antagonists.
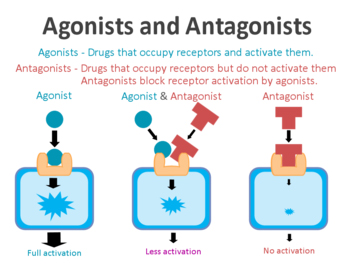
Agonist vs antagonist. Source: Psychonautwiki
Adenosine A2a receptor antagonist are drugs that bind to and block (as the label on the can suggests) the Adenosine A2a receptor:
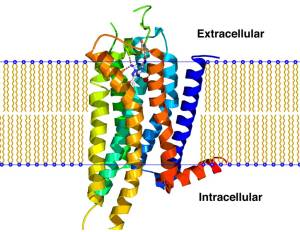
The Adenosine A2a receptor. Source: Wikipedia
Adenosine binds to the Adenosine A2a receptor. Adenosine is an important chemical in the body as it helps in cellular energy transfer. It does this by forming molecules like adenosine triphosphate (ATP) and adenosine diphosphate (ADP). Adenosine also plays a role in signalling various biological pathways which it does by binding to one of 4 different subtypes of adenosine receptor. All adenosine receptor subtypes (A1, A2a, A2b, and A3) are G protein-coupled receptors.
What are G protein–coupled receptors?
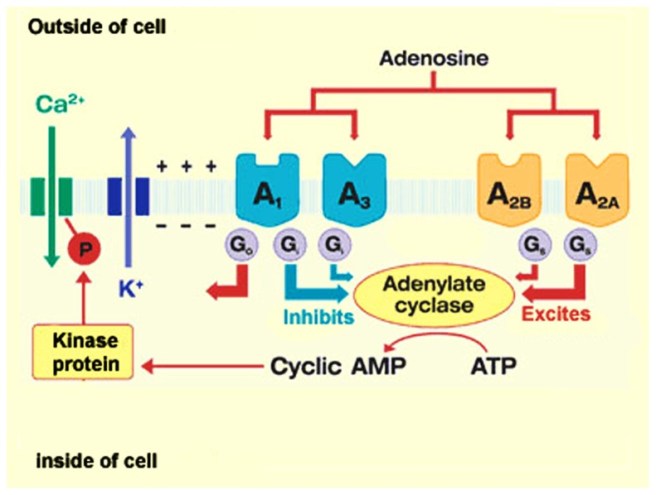
When adenosine binds to an adenosine receptor, this action activates a process (or multiple processes) associated with that particular receptor. Some of these processes increase the activity of a protein called adenylate cyclase, while binding to other adenosine receptors can reduce it. This activity is connected to G proteins which are associated with that particular receptor:
The adenosine receptor pathway. Source: Researchgate
You have probably never heard of G protein–coupled receptors, but they involve about 4% of the protein-producing DNA in each cell of your body. They are a very common mechanism for getting things done inside of cells. When adenosine binds to one of the four receptors and they becomes activated, the part of the receptor structure inside the cell will send a signal via a messenger (called a G-protein).
There are two primary messaging pathways involving the G protein in G protein–coupled receptors:
- the cyclic adenosine monophosphate (cAMP) signal pathway
- the phosphatidylinositol signal pathway
Adenosine activation of one of its G protein–coupled receptors affects the level of cAMP in target cells. cAMP then goes on to activate other pathways such as calcium channels which increase the amount of calcium entering a cell.
Via this process adenosine is known to reduce levels of inflammation (Click here to read more about this). So much so, in fact, that adenosine has been proposed as a treatment for septic patients (Click here to read more about this). Thus by blocking the adenosine receptors with an antagonist, one could be increasing the chance of sepsis.
But how does Tozadenant have an effect in Parkinson’s?
Adenosine A2a receptors are very abundant in the striatum of both rodent and human brains. The striatum is the region of the brain where most of the dopamine is released. Adenosine A2a receptors sit right next to Dopamine receptors and researchers noticed that Adenosine A2a receptors functionally oppose the actions of Dopamine receptors. That is to say, binding of adenosine to the Adenosine A2a receptor reduces the activity of the dopamine receptor in the striatum. This observation led to the hypothesis that an antagonist for the Adenosine A2a receptor could actually enhance the activity of dopaminergic therapies (like L-dopa), by blocking the inhibitory activity of Adenosine A2a receptor. Thus a Adenosine A2a receptor antagonist in combination with L-dopa could help in alleviating Parkinsonian motor symptoms.
And this idea seems to actually work: in rodent and primate models of Parkinson’s, when Adenosine A2a receptor antagonists were given alone (as monotherapy) they demonstrated only marginal activity (Click here and here to read more on this), but when given with L-dopa, they were able to significantly potentiate the effect of the dopaminergic treatment (Click here and here to see more on this).
And these findings led to clinical trials of Adenosine A2a receptor antagonists for Parkinson’s.
What clinical research has been done in Parkinson’s with Tozadenant?
There have been a series of clinical trials looking at various Adenosine A2a receptor antagonists, such as Istradefylline. Here were are going to focus just on Tozadenant (Click here and here to read more about the other Adenosine A2a receptor antagonists in clinical trials of Parkinson’s).

Title: Tozadenant (SYN115) in patients with Parkinson’s disease who have motor fluctuations on levodopa: a phase 2b, double-blind, randomised trial.
Author: Hauser RA, Olanow CW, Kieburtz KD, Pourcher E, Docu-Axelerad A, Lew M, Kozyolkin O, Neale A, Resburg C, Meya U, Kenney C, Bandak S.
Journal: Lancet Neurol. 2014 Aug;13(8):767-76.
PMID: 25008546 (This article is OPEN ACCESS if you would like to read it)
In this clinical study, the researchers conducted an international, multi-centre, Phase IIb, randomised, double-blind, placebo-controlled, parallel-group, dose-finding clinical trial of Tozadenant (phew, say that lot three times really fast). The drug was tested in 420 levodopa-treated people with Parkinson’s (average age 63·3 years; average duration of PD: 8·7 years) who had motor fluctuations (at least 2·5 h off-time per day). Participants were randomly assigned to receive either Tozadenant 60, 120, 180, or 240 mg (or a matching placebo) twice daily for 12 weeks.
A total of 337 people completed study treatment and the results suggested that Tozadenant (at 120 or 180 mg twice daily) was generally well tolerated and was effective at reducing off-time.
It is now interesting to note that 6 participants died during this study (all of whom were receiving Tozadenant). Two of the deaths were regarded as possibly related to the study drug, two were unlikely to be related, and two not related to the drug. Two of the deaths (one each in the 60 and 180 mg twice-daily groups) were associated with Sepsis. Given the size of the study (400+ people), however, neither the independent data monitoring committee nor an independent panel of experts who reviewed the data at the end of the study found a relation between Tozadenant and the deaths.
And what does the current clinical trial involve?
Acorda actually has two Phase III clinical trials for Tozadenant.
Following the interesting results of the Phase IIb clinical trial, Acorda set up a Phase III trial in which Tozadenant is being self administered for 24 weeks (alongside participant’s other Parkinson’s medications). The trial is comparing two of doses of Tozadenant (60 mg and 120 mg) and comparing it to placebo, and it is assessing improvements in motor function and daily living in people with Parkinson’s. The results of the trial are expected in the first quarter of 2018.
And in addition to the first Phase III trial, Acorda is running a separate open-label, long-term safety study which started in April 2017. This trial is to determine the safety of people with Parkinson’s using Tozadenant for long periods of time.
So what does it all mean?
Something going wrong in a clinical trial is a bad thing. And agranulocytosis and sepsis are very serious medical situations – in this particular case, unfortunately five people who volunteered to take part in these trials have died. This is definitely not a minor issue. And Acorda is now in discussions with the trials independent Data Safety Monitoring Board (DSMB) and officials from the US FDA.
But the point of this post is that we should not panic when reading the media headlines and rush to trash this drug.
Tozadenant could potentially be very useful for Parkinson’s and may just requires more careful participant selection and better monitoring. While it is extremely unfortunate that people have died in this clinical trial, it would also be regrettable if this episode causes people with Parkinson’s to be fearful of volunteering for a clinical trial based on mass-media headlines.
Acorda and the FDA are going to investigate and review the situation, and we shall see what they find. Hopefully they will be able to identify the risk factors associated with the individuals that passed away, and the overall trial will be able to continue.
Addendum – 20/11/2017: In a move that was widely predicted, Acorda Therapeutics today announced that it was discontinuing its clinical development program for the drug Tozadenant in Parkinson’s. This decision includes immediately discontinuing the dosing of all participants currently enrolled in the studies (Click here to read the press release).
There had been concerns expressed by participants and research centres alike about the cost and inconvenience of the increased blood monitoring, as well as some participant deciding to pull out of the study. Rather than continuing the program, the company has decided to stop the studies for now and analyse the results it already has from the phase III safety and efficacy trial – over 90% of the participants in that short, placebo-controlled study (entitled CL-05) have completed the study. It will be interesting to see those results. The Company expects to release them early in the new year.
EDITORIAL NOTE: Acorda Therapeutics Inc is a publicly traded company. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. Acorda Therapeutics Inc has not requested this material to be produced, nor has the author had any contact with the company or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from Acorda


Thanks
LikeLike
“….we should not panic when reading the media headlines and rush to trash this drug.
Tozadenant could potentially be very useful for Parkinson’s and may just requires more careful participant selection and better monitoring.
…(It will be) regrettable if this episode causes people with Parkinson’s to be fearful of volunteering for a clinical trial based on mass-media headlines.”
Keith
LikeLike
Doesn’t adenosine have an important role in sleep?
LikeLike
Hi Kevin,
Thanks for the interesting question.
Adenosine levels in the brain are known to rise over the course of the day and drop rapidly with periods of sleep. I don’t know enough about it to say whether it is important for sleep, but it certainly is affected by sleep. Why do you ask? Do you have a theory?
Kind regards,
Simon
LikeLike
Interesting–surprising there were such unexpected (and lethal) side effects so late in the process.
Agree the decision to participate (or not) in a trial should be rational–not based on unrealistic fears (or for that matter, unrealistic hopes of being ‘cured’).
Last month, I finished participating in a Phase 1 trial of an immunotherapy drug, targeting alpha synuclein. I was one of only 18 volunteers w/ PD–this was after initial safety testing in 40 people without PD. There were a few uncomfortable and inconvenient aspects to the trial, but I’d have little hesitation in doing something like it again.
LikeLike
Hi David,
Thanks for your comment and for taking the courageous step of being involved in a clinical trial. I have a lot of respect for that. While I appreciate that you can’t really say anything about the study, we will certainly be very interested to see the final results.
It is unfortunate that the Acorda situation has occurred, especially the loss of life. But it was recently pointed out by a senior academic here in the UK that in the current clinical trial environment L-dopa would never have been approved for clinical use. It displayed mixed results when it was first independently tested by different research groups, with many of those researchers failing to find any benefits at all. It wasn’t until George Cotzias tamed it that L-Dopa was eventually approved by the FDA.
In the current environment, it is so hard to get drugs to trial that any setback could halt a trial program and result in a potentially useful drug being canned. I hope that the initial results of the Tozadenant phase III trial will be interesting enough for further exploration.
I fear that this situation is not so much a failure of a drug, but rather an illustration of how poor how assessment tools are in these efforts. We really require more continuous monitoring (which may have saved lives here) of both PD features and overall wellbeing. Hopefully new tracking technology and biomarkers will aid us in efforts to improve this.
Thanks again for your comment.
Kind regards,
Simon
LikeLike