|
# # # # Drug repurposing represents a more rapid way of bringing new treatments to the public than the traditional novel drug development route. It involves clinically testing therapies in new conditions for which they are not currently approved. Researchers conduct large drug screening studies to identify agents that could be repurposed and basic science experiments can also provide data to support evaluating a particular class of drug in new medical conditions. Recently, accumulating evidence has pointed towards a class of drugs called sartans as potential candidates for repurposing for Parkinson’s. In today’s post, we will discuss what sartans are and review some of new studies suggesting the case for support. # # # # |
 The sartan family photo. Source: MDPI
The sartan family photo. Source: MDPI
The “Sartans” sound like an ancient Greek tribe or Scottish clan.
Like a formidable horde of Viking brigands bearing down on some hapless village that is unaware it is about to be pillaged. One can imagine someone in said village suddenly seeing something out of the ordinary, realizes the impending disaster, and screaming “the sartans are coming! The sartans are coming!”
 Source: Lifeinnorway
Source: Lifeinnorway
But the reality is something else entirely.
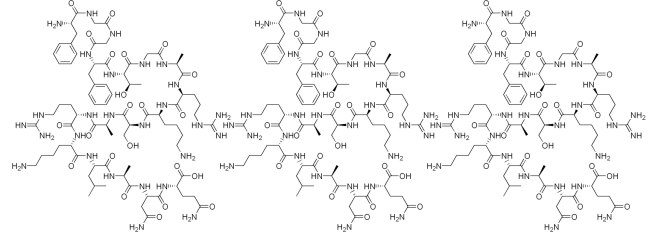
Sartans are in fact a class of drugs that are widely used for the treatment of hypertension (that is: high blood pressure). They are also administered to patients with certain heart or kidney diseases. The drugs work by blocking the function of a hormone.
That hormone is called angiotensin II.
Angiotensin II has a bad habit of causing your blood vessels (the tubes that transport blood around your body) to narrow. This action can increase your blood pressure and force your heart to work harder. Not a good situation in terms of longevity.
And before we go any further it has to be said, like any marauding tribe, the sartans need to be treated with respect – they are powerful drugs and can do worse than simply making people feel dizzy if they lower the blood pressure too much.
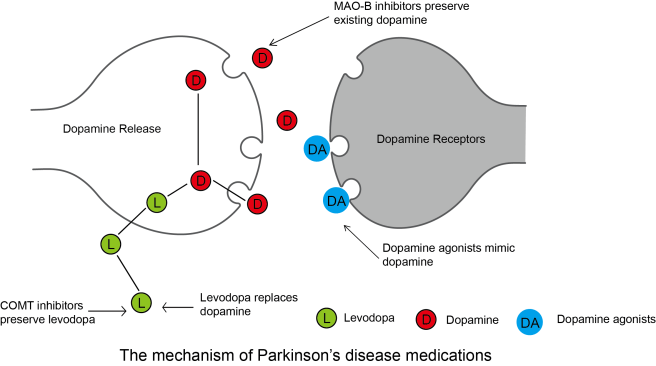
Good to know. But what do sartans have to do with Parkinson’s?