|
A recent study published by French, British and Swiss researchers has grabbed the attention of some readers. The report suggests that the inert/noble gas, Xenon, has powerful anti-dyskinetic properties in both mouse and primate models of Parkinson’s with L-DOPA-induced dyskinesias. Dyskinesias are involuntary movements that can develop over time with prolonged used of L-DOPA treatments. In today’s post, we will discuss what Xenon is, how it may be reducing dyskinesias, and we will consider some of the issues associated with using Xenon. |
Dyskinesia. Source: JAMA Neurology
There is a normal course of events following a diagnosis of Parkinson’s.
Yes, I am grossly over-generalising, and no, I’m not talking from personal experience, but just go with me on this for the sake of discussion.
First comes the shock of the actual diagnosis. For many it is devastating news – an event that changes the course of their future. For others, however, the words ‘you have Parkinson’s‘ can provide a strange sense of relief that their current situation has a name and gives them something to focus on.
This initial phase is usually followed by the roller coaster of various emotions (including disbelief, sadness, anger, denial). It depends on each individual.
The emotional rollercoaster. Source: Asklatisha
And then comes the period during which many will try to familiarise themselves with their new situation. They will read books, search online for information, join Facebook groups (Click here for a good one), etc.
That search for information often leads to awareness of some of the realities of the condition.
And one potential reality that causes concern for many people (especially for people with early onset Parkinson’s) is dyskinesias.
What are dyskinesias?
Dyskinesias (from Greek: dys – abnormal; and kinēsis – motion, movement) are a category of movement disorders that are characterised by involuntary muscle movements. And they are certainly not specific to Parkinson’s.
But in the case of Parkinson’s, dyskinesias are generally associated with long-term use of Levodopa (also known as Sinemet or Madopar).

Sinemet is Levodopa. Source: Drugs
How do dyskinesias develop in Parkinson’s?
There is a lot of debate over this topic, but there are some basic details that researchers generally tend to agree on.
Before being diagnosed and beginning a course of Levodopa, the locomotion parts of the brain in a person with Parkinson’s gradually becomes more and more inhibited. This increasing level of inhibition results in the slowness and difficulty in initiating movement that characterises this condition.
A person with Parkinson’s may want to move, but they can’t – they are inhibited. In effect, they are akinetic (from Greek: a-, not, without; and kinēsis – motion).

Drawing of an akinetic individual with Parkinson’s, by Sir William Richard Gowers
Source: Wikipedia
Levodopa (or L-DOPA) tablets provide the brain with the precursor to the chemical dopamine. Dopamine producing cells are lost in Parkinson’s, so replacing the missing dopamine is one way to treat the motor features of the condition. Simply giving people pills of dopamine is a non-starter though: dopamine is unstable, breaks down too quickly, and (strangely) has a very hard time getting into the brain (it is blocked by the protective layer called the blood-brain-barrier). Levodopa, on the other hand, is very robust and has no problem entering the brain.
Once inside the brain, Levodopa is quickly converted into dopamine. It is changed into dopamine by an enzyme called DOPA decarboxylase, and this change rapidly increases the levels of dopamine in the brain, allowing the locomotion parts of the brain to function more normally.

The chemical conversion of L-DOPA to dopamine. Source: Nootrobox
In understanding this process, it is important to appreciate that when an Levodopa tablet is consumed and Levodopa enters the brain, there is a rapid increase in the levels of dopamine. This ‘spike’ in the supply of dopamine will last for the next few hours, before the dopamine is eventually used up.
As the effects of the Levodopa tablet wear off, another tablet will be required. This use of multiple Levodopa pills across the day gives rise to a wave-like shape to the dopamine levels in the brain over the course of the day (see the figure below). The first pill in the morning will quickly lift the levels of dopamine enough that the individual will no longer feel akinetic. This will allow them to be able to function with normal controlled movement for several hours before the Levodopa begins to wear off. As the Levodopa wears off, the dopamine levels in the brain drop back towards levels that will leave the person feeling akinetic and at this point another Levodopa tablet is required.
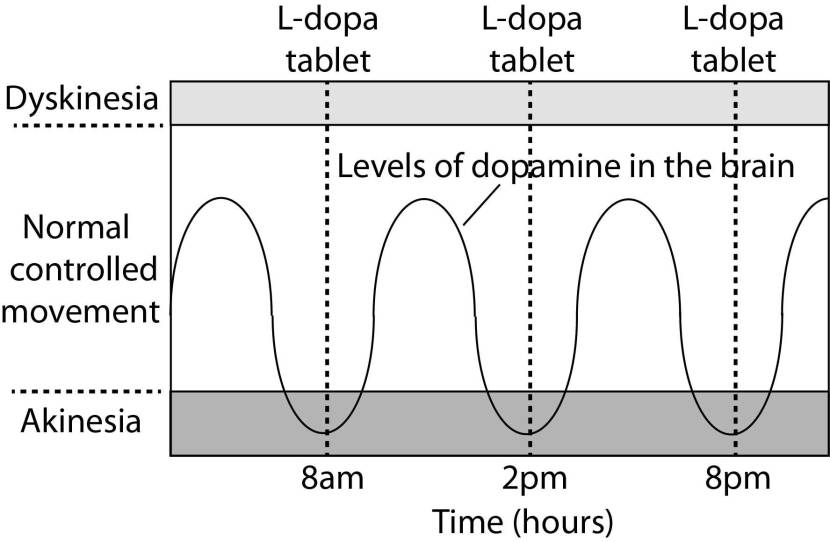
A hypothetical illustration of dopamine levels over a day
After several years of Levodopa use, many people with Parkinson’s will experience a weaker response to each tablet. They will also find that they have more time during which they will be unable to move (exhibiting akinesia). This is simply the result of the progression of Parkinson’s – Levodopa treats the motor features of the condition but only hides/masks the fact that the disease is still progressing.
To combat this shorter response time, the dose of Levodopa is usually increased. This will result in increasing levels of dopamine in the brain (as illustrated by the higher wave form over time in the image below). Gradually it will take more Levodopa medication-induced dopamine to lift the individual out of the akinetic state.

Again this illustration is hypothetical (situation differs between individuals)
This increasing of Levodopa dosage, however, results in too much dopamine being present in the brain at times. And this situation is often associated with the gradual development of abnormal involuntary movements that appear when the levels of Levodopa induced dopamine are the highest.
These are the dyskinesias.
Are there different types of dyskinesias?
Yes there are.
Dyskinesias have been broken down into many different subtypes, but the two main types of dyskinesia are:
- Chorea – these are involuntary, irregular, purposeless, and unsustained movements. To an observer, Chorea will look like a very disorganised/uncoordinated attempt at dancing (hence the name, from the Greek word ‘χορεία’ which means ‘dance’). While the overall activity of the body can appear continuous, the individual movements are brief, infrequent and isolated. Chorea can cause problems with maintaining a sustained muscle contraction, which may result in affected people dropping things or even falling over.
- Dystonia – these are sustained muscle contractions. They often occur at rest and can be either focal or generalised. Focal dystonias are involuntary contractions in a single body part, for example the upper facial area. Generalised dystonia, as the name suggests, are contraction affecting multiple body regions at the same time, typically the trunk, one or both legs, and another body part. The intensity of muscular movements in sufferers can fluctuate, and symptoms usually worsen during periods of fatigue or stress.
I have previously discussed the current treatment options for dyskinesias (click here to see that post). But in today’s post we are going to have a look at some new research that could potentially help in the treatment of dyskinesias.
Here is the report of the research:
Title: Inhaling xenon ameliorates l-dopa-induced dyskinesia in experimental parkinsonism.
Authors: Baufreton J, Milekovic T, Li Q, McGuire S, Moraud EM, Porras G, Sun S, Ko WKD, Chazalon M, Morin S, Normand E, Farjot G, Milet A, Pype J, Pioli E, Courtine G, Bessière B, Bezard E.
Journal: Mov Disord. 2018 May 14. [Epub ahead of print]
PMID: 29756234 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers wanted to explore the effect that the noble gas xenon has on L-DOPA-induced dyskinesias.
What is Xenon?
Xenon is a chemical element (symbol Xe; atomic number 54 – due to the number of protons found in the nucleus of an atom – see image below). It is a colorless, odorless but very dense noble gas found in the Earth’s atmosphere in trace amounts.
Xenon. Source: Adopt-a-element
First discovered in the UK by William Ramsay and Morris Travers in 1898 (Ramsay suggested the name xenon from the Greek word ξένον [xenon] for ‘stranger’ or ‘foreigner’), it was in the 1930s that xenon became a really useful element when American engineer Harold Edgerton invented of the camera flash bulb by passing brief electric current through a tube filled with xenon gas (earning him the nickname in photographic circles of “Papa Flash”).
Harold “Papa Flash” Edgerton. Source: Wikipedia
There are now many uses for xenon, for example it is used as an anesthesia (Click here and here to read more about this), it is used to image the heart, lungs, and brain, it is with lasers, and on the 31st August 2014, the World Anti Doping Agency (WADA) added inhaled xenon to the list of prohibited substances and methods (yes, a performance enhancing drug via increased production of erythropoietin).
Xenon Imaging. Source: Siemens
So what did the researchers find with Xenon and dyskinesias?
The researchers began their study with the knowledge that xenon was capable of blocking the binding of the chemical glutamate to the N-methyl-D-aspartate (or NMDA) receptor.
And before you ask: NMDA receptors (or NMDAR) are receptors for the chemical glutamate (which is a neurotransmitter like dopamine). They are proteins that sit on the surface of a cell, waiting for glutamate to come floating along and bind to them. By binding to the receptor, glutamate activates the receptor which opens and lets calcium flow into the cell.
An NMDA receptor. Source: Wikipedia
NMDARs are found widely distributed in the brain, but with the development of dyskinesias they appear to become more abundant. And as a result, researchers have been experimenting with drugs that block NMDA receptors (called NMDA receptor antagonists) as potential treatment for dyskinesias.
And they appear to help in many cases.
Amantadine is an example of a (weak) NMDAR antagonist, and in 2017 the U.S. Food and Drug Administration (FDA) approved the use of amantadine in an extended release formulation (developed by Adamas Pharma) for the treatment of dyskinesias.
Amantadine. Source: Dailymed
So Xenon acts like Amantadine? By blocking the NMDA receptors?
Yes.
This was previously demonstrated in this study:
Title: The xenon-mediated antagonism against the NMDA receptor is non-selective for receptors containing either NR2A or NR2B subunits in the mouse amygdala
Authors: Haseneder R, Kratzer S, Kochs E, Höfelmann D, Auberson Y, Eder M, Rammes G.
Journal: Eur J Pharmacol. 2009 Oct 1;619(1-3):33-7.
PMID: 19686722
In this study, the researchers demonstrated that treatment with Xenon robustly blocks NMDARs, and this effect of Xenon on NMDA receptors has been replicated in other regions of the brain (Click here to see that research).
And as I suggested above, there is ample evidence that targeting NMDA receptors can help to alleviate dyskinesias. For example:
Title: Targeting NR2A-containing NMDA receptors reduces L-DOPA-induced dyskinesias.
Authors: Gardoni F, Sgobio C, Pendolino V, Calabresi P, Di Luca M, Picconi B.
Journal: Neurobiol Aging. 2012 Sep;33(9):2138-44.
PMID: 21821315
In this particular study, the researchers used a small peptide to target the NR2A subunit of NMDA receptors during the development of dyskinesias in a rat model of Parkinson’s, and they found that this treatment led to a reduction in the percentage of rats that went on to develop dyskinetic movements.
OK, so targeting NMDA receptors help reduce dyskinesias, and xenon targets NMDA receptors. How well did xenon work compared to normal NMDA receptor targeting medication?
It actually worked as well as amantadine (the anti-dyskinetic NMDAR targeting drug we mentioned above). First the researchers took sections of brain from dyskinetic and non-dyskinetic rodents and they looked at the electrical activity of the cells in those section following xenon treatment, and they found that xenon normalised the electrical output of the cells – via manipulation of the NMDARs.
Next, they treated dyskinetic and non-dyskinetic rodents with xenon (or amantadine) and they found that xenon reduced the dyskinesias in the majority of the animals to a comparable level to that of amantadine (see top graph in the image below). Importantly, this xenon treatment did not affect the overall level of activity in the animals compared to the control-treated animals (bottom graph in the image below) – indicating that the animals were not becoming akinetic or anesthetised:
Source: Wiley
After demonstrating the effect in rats, the researchers next shifted their attention to investigating this phenomenon in a non-human primate model of Parkinson’s with L-DOPA-induced dyskinesias. And again, they observed a reduction in dyskinesias (comparative to amantadine – top graph in the image below), without any deterioration in the anti-Parkinsonian effect of L-DOPA (the Disability score in the bottom graph in the image below):
Source: Wiley
The researchers also observed a trend toward an increase in ‘good ON‐time’, that is, more time spent without dyskinesias interfering with normal daily activities. They concluded their report by suggesting that their “results in the gold‐standard mouse, rat, and non-human primate models of L-DOPA-induced dyskinesias provide a strong rationale to expedite clinical testing of this safe yet unconventional approach”.
And they suggest that “Patients at an advanced stage with severe dyskinesia uncontrolled by treatment or severe gait disorders (skewness, freezing) who may need a “rescue therapy” could take advantage of such an inhaled therapy”.
Has anyone looked to see if xenon treatment is neuroprotective?
Yes, they have:
Title: The noble gas xenon provides protection and trophic stimulation to midbrain dopamine neurons
Authors: Lavaur J, Le Nogue D, Lemaire M, Pype J, Farjot G, Hirsch EC, Michel PP.
Journal: J Neurochem. 2017 Jul;142(1):14-28.
PMID: 28398653 (This report is OPEN ACCESS if you would like to read it)
In this study, the investigators found that xenon provided partial but sustained protection to midbrain DA neurons in a cell culture-based progressive loss of dopamine neurons model of Parkinson’s. It should be noted, however, that the ‘progressive model’ of Parkinson’s was induced by over-activation of the NMDARs, which may not be a disease-relevant model of PD. But even when the researchers looked at spontaneous cell death in their cultures, they found that xenon treatment was having a beneficial effect.
Wow. That’s really interesting. Where can I get me some of that Xenon gas?
So here comes the health warning:
|
Xenon is a very dense gas, which can cause asphyxiation. It can be strongly oxidative when mixed with oxygen, and (as the camera flash bulb use of it should indicate) xenon is also explosive. Thus, great caution must be taken when dealing with it and it should not be taken without medical supervision. In addition, while these new research results are very interesting, xenon treatment must be considered extremely experimental – we have no idea of how it will affect an individual with Parkinson’s with L-DOPA-induced dyskinesias, let alone what affect it may have on a particular treatment regime (such as Amantadine). While xenon is available for medical purposes, people with Parkinson’s should speak to their clinician before even contemplating any kind of usage (for example, anesthesia). The information being provided here on this website is for educational purposes only. |
All of that said, it will be interesting to see how the researchers take this work forward for clinical testing, which is apparently what they plan to do.
Xenon gas can be safely kept in sealed glass or metal containers, but it readily dissolves in most plastics and rubbers – it will eventually escape from any container sealed with these materials – thus packaging of it will need to be handled carefully.
At low levels, Xenon is non-toxic, plus it readily dissolves in blood and penetrates the blood–brain barrier – the protective membrane surrounding the brain – which is very convenient going forward with clinical testing.
But xenon does have one curious side effect that may make any clinical trial of this gas rather amusing to be part of.
You see, xenon inhalation makes your voice go really low.
And when I say really low, I mean REALLY low – literally the opposite of helium:
But (AGAIN) it should be noted that Xenon is not a gas that should be played with at home in any kind of unsupervised manner.
At high doses it can be very dangerous as this TV presenter found out when he breathed Xenon simply to make his voice sound like James Earl Jones (watch from 1:55 onwards):
What does it all mean?
L-DOPA-induced dyskinesias are one of the most debilitating aspects of Parkinson’s for a lot of sufferers, and there are a limited number of treatments to help alleviate these involuntary movements. Recently French, Swiss & UK researchers have proposed a new experimental method for reducing dyskinesias that involves the inhalation of the noble gas xenon.
The results of their study are interesting, and deserve further investigation – particularly as the researchers appear to be very keen to take this approach forward for clinical testing.
One small caveat with the two xenon-focused research reports discussed in this post (the dyskinesia and the neuroprotective studies) is that both studies were supported by a French company called Air Liquide Santé International, which “aims to provide customers in the continuum of care from hospital to home with medical products, healthcare specialty ingredients and services that contribute to protecting vulnerable lives”.
I am not for a second suggesting that there is anything suspicious about this situation, I am only mentioning this minor detail for the purposes of transparency. It would be prudent, however, to see the results of these studies independently replicated by other research groups before we get too excited about the findings.
Once replicated, it will be really interesting to start the ‘creepy voice’ clinical trial of this new approach.
EDITOR’S NOTE: The information provided by the SoPD website is for educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. Please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Wikipedia














Very interesting and well written in my unprofessional opinion. The ‘splaining of dyskinesias was very well written and it got more interesting from there. Useful info and then hopeful info. Thanks
LikeLiked by 1 person
Thanks DKDC – glad you liked it.
Simon
LikeLike
Indeed, interesting. Thank you, once again, Simon.
In your hypothetical illustration showing the narrowing region between akinesia and dyskinesia, I think I understand the rising of the akinesia threshold as a desensitization to levodopa (or possibly continuing underlying disease progression?). But, I don’t have any idea what gives rise to the lowering of the dyskinesia threshold. Is this understood?
Thanks once more. You’ve been BUSY lately!
LikeLike
Hi Tom,
Thanks for your comment – glad you found the post interesting.
The rising akinesia is a result of the progressive nature of the condition. When folks get diagnosed with PD, they may have lost 40-60% of the dopamine producing neurons, but unfortunately this cell loss continues after diagnosis as well. This is why the dose of L-Dopa needs to slowly increase over time. There is more lost dopamine to replace.
As for the dyskinesia threshold, this is less clear and the source of a lot of academic debate (hence the reason I avoided this matter in the post). For years it was assumed that dyskinesias were solely an issue of dopamine dysregulation in the striatum (the region where the bulk of the dopamine is released), but more recently it has become clear that the situation is a lot more complicated than that. For example, a few years back a group of researchers reduced L-DOPA-induced dyskinesias simply by delivering a dopamine receptor 1 antagonist to the primary motor cortex (a region some distance from the striatum). Here is that research report: http://www.jneurosci.org/content/32/47/16541. That said, we are slowly chipping away at the mechanism of action and this will result in better treatments.
I hope this answers your question – happy to expand on it.
Kind regards,
Simon
LikeLiked by 1 person
Thanks, Simon. The breadth AND depth of your knowledge, and your passion for the research continues to impress me. Clearly, Billy didn’t do any damage.
🙂
Tom
LikeLiked by 1 person
Thank you so much for a brilliant panoramic 360-view of Parkinsonism and all innovations around it . God bless You , Thank You
LikeLike
Hi Radosla,
You’re welcome. Glad you like it!
Simon
LikeLike
Fascinating Simon! Sorta blows your mind – one permanent end to that God-awful ‘you don’t die from it, you die with it’ aphorism. Kidding folks 🙂
LikeLike
Hi Lisa,
Glad you found it interesting.
Kind regards,
Simon
LikeLike
Simon, thank you again. There is one aspect of this report that I find fascinating: in college they teach you that so called noble gases have very few chemical compounds and most of those with very strong oxidizing agents . The fact that xenon is active in a biological system contradicts this notion to some degree. It would be interesting to understand the chemistry and the energy behind this process.
LikeLike
Hi Felix,
Thanks for the comment and interesting note. Given these sorts of potential applications, I think there is still a bit of research left in the field of chemistry trying to work it all out. But the idea of an inert ‘noble’ gas is certainly something of the past. As for the explanation of the “chemistry and energy behind this process”, I am definitely not the man for that task – my knowledge of chemistry is terrible!
Kind regards,
Simon
LikeLike