|
This post is a game of two halves. The first half will explain the concept of a surgical procedure for Parkinson’s called ‘subthalamic deep brain stimulation‘, in which doctors permenantly implant electrodes into the brain to stimulate a region – the subthalamic nucleus. By stimulating this region with electrical impulses, doctors can provide a better quality of life (in most cases) to people with severe features of Parkinson’s. In the second half of this post, we will look at an approach to doing the same thing,… but without the electrodes. Rather, researchers are using gene therapy. In today’s post, we will discuss what deep brain stimulation is, what gene therapy is, and how the gene therapy approach is having a different kind of impact on the brain to that of deep brain stimulation.
|

Source: Youtube
Welcome to the first half of today’s post.
It begins with you asking the question:
What is deep brain stimulation?
Deep brain stimulation (or DBS) is a treatment method that involves embedding electrodes into the brain to help modulate the brain activity involved in movement.
It is a prodcedure that is usually offered to people with Parkinson’s who have excessive tremor or debilitating dyskinesias.
First introduced in 1987, deep brain stimulation consists of three components: the pulse generator, an extension wire, and the leads (which the electrodes are attached to). All of these components are implanted inside the body. The system is turned on, programmed and turned off remotely.
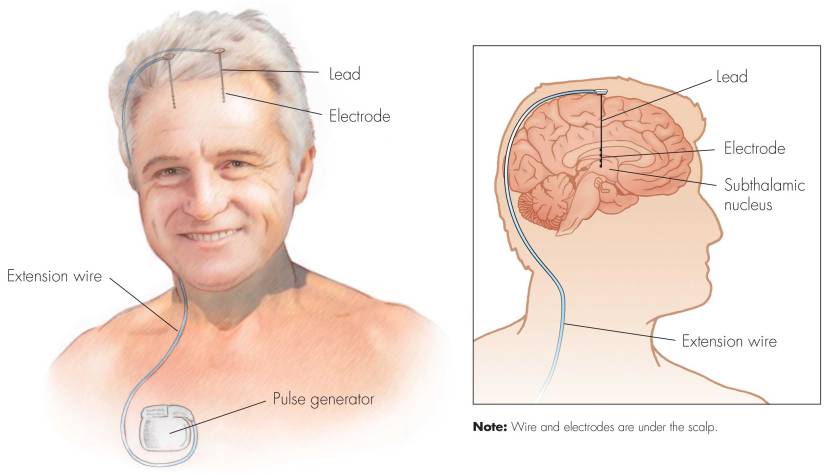
Source: Ucdmc
The electrodes that are implanted deep in the brain are tiny, and the very tip of them has small metal plates (each less than a mm in width) which provide the pulses that will help mediate the activity in the brain.

DBS electrode tip. Source: Oxford
The electrode extends up into the leads (or extension wire) which continue up and out of the brain, across the top of the skull, down the neck and to the pulse generator which is generally located on the chest.

Xray image demonstrating the leads. Source: Fineartamerica
There are many different brands/types of pulse generators, but they all largely do the same basic function. They are titanium packets containing the electronics and power supply for the leads and electrodes. They are implanted in a subcutaneous spot, usually located under the collar bone on the chest.

Pulse generators. Source: Cambridge (also a good read on DBS)
The pulse generator is programmable (which the doctor usually does several weeks post surgery). A small hand held device is used to turn on/off the generator, generally by holding the device over it.

Remote control. Source: Cambridge
For more information on the topic, click here to read about a case report of deep brain stimulation implantation.
Here is a video – kindly provided by fellow kiwi Andrew Johnson – that demonstrates the benefits of DBS:
How does deep brain stimulation actually work?
I have previously discussed how we initiate movement (Click here to read that post), and I will only briefly summarise the basics here.
Movement is largely controlled by the activity in a specific group of brain regions, collectively known as the ‘Basal ganglia‘.
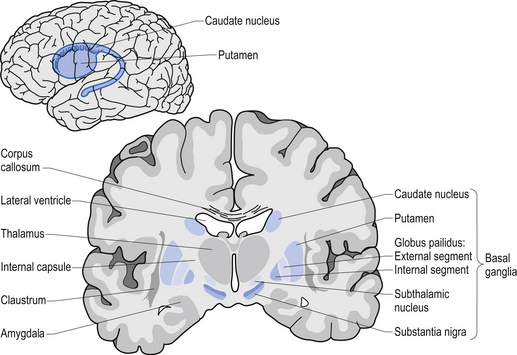
The basal ganglia structures (blue) in the human brain.
Source: iKnowledge
The basal ganglia receives signals from the overlying cerebral cortex regarding possible movements to make, and it processes that information before sending a signal on to another important participant in the regulation of movement: the thalamus.

A brain scan illustrating the location of the thalamus in the
human brain. Source: Wikipedia
The thalamus is a structure deep inside the brain that acts like the central control unit of the brain. Everything coming into the brain from the spinal cord, passes through the thalamus. And everything leaving the brain, passes through the thalamus. It is aware of most everything that is going on and it plays an important role in the regulation of movement. Importantly it is biased towards inhibiting movement – a signal from the basal ganglia has to be pretty strong, for the thalamus to give the green light and a particular movement can then be made.
Under normal circumstances, dopamine producing neurons release dopamine in the basal ganglia where it helps to mediate the local environment. It acts as a kind of lubricant for movement, the oil in the machine if you like. It helps to reduce the inhibitory bias of the basal ganglia.
Thus, with the loss of dopamine neurons in Parkinson’s, there is an increased amount of inhibitory activity. And as a result, the thalamus is kept in an overly inhibited state. And this is the reason why people with Parkinson’s have trouble initiating movement.

The workflow of the movement decision making process. Source: BJP
Now, as you can see from the image above, the globus pallidus is one of the main conduits of information into the thalamus. Given this pivotal position in the regulation of movement, the globus pallidus has been a region of major research focus for a long time.
It is also one of the sites targeted in deep brain stimulation therapy for Parkinson’s. Another target of deep brain stimulation therapy is the subthalamic nucleus which help to regulate the activity of the globus pallidus. By placing electrodes in one of these two brain regions (see image below), doctors can regulate the signal being passed from the basal ganglia to the thalamus and thus provide people with Parkinson’s relief from their motor-associated symptoms.

Two of the target sites for deep brain stimulation. Source: JAMA
But what do the electrodes do?
To keep things simple, in today’s post we are going to focus on the subthalamic nucleus.
The subthalamic nucleus is a very small region of the brain (140 cubic mm), consisting of approximately 300,000 neurons – making up about 0.02% of the total volume of the brain.
In Parkinson’s, the subthalamic nucleus becomes very excited, and the cells in the region start firing at an increased rate. This over activation of the region results in dysfunction in other parts of the basal ganglia – ultimately disrupting normal movement.
The electrodes that are placed in the subthalamic nucleus during deep brain stimulation will be programmed to release tiny electrical pulses that will help regulate the firing of the surrounding cells. By modulating the firing of those cells, deep brain stimulation reduces the inhibition in the basal ganglia circuit overall, which allows individuals with Parkinson’s to move more freely.
Sounds good. Right?
DBS is a good treatment option, and it is programmable/tunable (as opposed to high frequency focused ultrasound approaches – click here to read more about this). And while it must be remembered that this is not a curative therapy, it can make life a lot easier for those suffering from some of the more severe features of Parkinson’s.
HALF-TIME
Ok, welcome back to the second half of this post.
In this half, the conversation switches from deep brain stimulation to gene therapy.
And this half obviously begins with the question:
What is gene therapy?
Gene therapy is an experimental treatment approach that involves treating Parkinson’s with DNA rather than drugs.
The gene therapy involves inserting a piece (or multiple pieces) of DNA into a cell which will cause the cell to produce proteins that they usually do not. The introduction of the DNA is usually achieved using genetically modified viruses which have had all the disease causing component removed, allowing us to use the virus as an efficient delivery system. Viruses by their very nature are good at infecting cells so if we remove the disease causing components, what is left is a very efficient biological delivery system.
Taking this approach one step further, we can take sections of DNA that contain the genes (these are the instructions for making proteins) involved with the production of a chemical called dopamine and insert them into our empty virus. Dopamine is a chemical in the brain whose levels are badly affected by Parkinson’s disease. By then injecting a virus with dopamine-production associated DNA into the brain, we can produce dopamine in any infected cells (it’s slightly more complicated than that, but you get the basic idea).
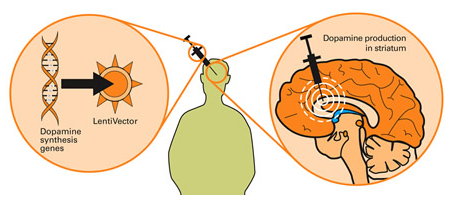
Gene therapy for Parkinson’s disease. Source: Wiki.Epfl
Generally speaking, the gene therapy approach has demonstrated amazing results in preclinical studies in the lab, but the transition to the clinic has not been easy (click here for a good review of the field).
One of the first clinical attempts at gene therapy for Parkinson’s was attempted by a company called Neurologix ltd.
 The company was developing a gene therapy product called NLX-P101, which involved a virus containing a gene called glutamic acid decarboxylase (GAD) – which is an enzyme involved in the production of a chemical called GABA – being injected into the subthalamic nucleus.
The company was developing a gene therapy product called NLX-P101, which involved a virus containing a gene called glutamic acid decarboxylase (GAD) – which is an enzyme involved in the production of a chemical called GABA – being injected into the subthalamic nucleus.
What is GABA?
Gamma-Aminobutyric acid (or GABA) is a chemical in the brain that is used for transmitting a signal between neurons. Hence, it is called a ‘neurotransmitter’ – similar to dopamine. The signal GABA passes on is usuallly inhibitory, reducing the chance that the recieving cell will be activated.
Ok, but why were the researchers injecting this ‘GAD’ virus into the subthalamic nucleus?
The virus containing GAD DNA was injected into the subthalamic nucleus, which becomes over-active in Parkinson’s disease. By introducing GAD (and subsequently GABA production) in the subthalamic nucleus, researchers were hoping to reduce the level of activity by inhibiting the levels of firing. By lowering the level of activity in the subthalamic nucleus, the researchers hypothesised that there would be a reduction in the level of dysfunction caused by over-active subthalamic output in other parts of the basal ganglia – ultimately improving normal movement.
Does it work?
Well, preclinical research provided proof of principle data to suggest that it worked. In 2002, this study was published:
 Title: Subthalamic GAD gene therapy in a Parkinson’s disease rat model.
Title: Subthalamic GAD gene therapy in a Parkinson’s disease rat model.
Authors: Luo J, Kaplitt MG, Fitzsimons HL, Zuzga DS, Liu Y, Oshinsky ML, During MJ.
Journal: Science. 2002 Oct 11;298(5592):425-9.
PMID: 12376704 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers the researchers used viruses (adeno-associated viruses – AAVs) to transfer the glutamic acid decarboxylase (GAD) gene into neurons of the subthalamic nucleus of rats. They noted changes in the activity of the subthalamic nucleus neurons in these animals. The investigators next modelled Parkinson’s in these animals and they found that this shift in activity actually resulted in a strong neuroprotective effect on the dopamine neurons (which are a population of neurons badly affected by Parkinson’s), and rescued the Parkinsonian behaviour of the rodents.
This research led to a series of clinical trials, that were started by Neurologix in 2003.
A Phase I study of GAD-gene therapy in 12 participants was conducted to assess safety of the treatment and the results were announced in late Sept 2005 (Click here to read the press release). The results of that study were published in 2007:
Title: Safety and tolerability of gene therapy with an adeno-associated virus (AAV) borne GAD gene for Parkinson’s disease: an open label, phase I trial.
Authors: Kaplitt MG, Feigin A, Tang C, Fitzsimons HL, Mattis P, Lawlor PA, Bland RJ, Young D, Strybing K, Eidelberg D, During MJ.
Journal: Lancet. 2007 Jun 23;369(9579):2097-105.
PMID: 17586305
In this trial, all of the participants (11 men and 1 female) received the gene therapy treatment on on side of the brains. The researchers reported significant improvements in clinical motor scores (as measured by UPDRS) at both 3 months and persisting up to 12 months, generally on the one side of the body. Brain imaging (PET scans) also revealed a marked reduction in activity in the subthalamic region which was restricted to the treated side of the brain.
The researchers concluded that “AAV-GAD gene therapy of the subthalamic nucleus is safe and well tolerated by patients with advanced Parkinson’s disease, suggesting that in-vivo gene therapy in the adult brain might be safe for various neurodegenerative diseases”
Given these positive Phase I results, a Phase II trial was quickly organised and conducted.
While the Phase I study was “open label” (that is, all of the participants and doctors involved in the study knew who was receiving which treatments), the Phase II trial was double blind (meaning that no one knew who was receiving the treatment). The study involved 45 subjects, who were randomly split into two group (on a 50:50 basis), either treated or sham surgery.
The results of the study were announced in June 2010 (Click here for the press release), and the report was published in 2011:
 Title: AAV2-GAD gene therapy for advanced Parkinson’s disease: a double-blind, sham-surgery controlled, randomised trial.
Title: AAV2-GAD gene therapy for advanced Parkinson’s disease: a double-blind, sham-surgery controlled, randomised trial.
Authors: LeWitt PA, Rezai AR, Leehey MA, Ojemann SG, Flaherty AW, Eskandar EN, Kostyk SK, Thomas K, Sarkar A, Siddiqui MS, Tatter SB, Schwalb JM, Poston KL, Henderson JM, Kurlan RM, Richard IH, Van Meter L, Sapan CV, During MJ, Kaplitt MG, Feigin A.
Journal: Lancet Neurol. 2011 Apr;10(4):309-19
PMID: 21419704
In this Phase II study, 23 of the participants were randomly assigned to sham surgery (control) group and 22 were assigned to the GAD gene therapy (treatement) group. Of these, 21 participants in the control group and 16 in the treatment group were analysed. The treatment group in this study had the GAD DNA-containing virus injected into the subthalamic nucleus on both sides of the brain (as opposed to the Phase I study where it was only injected into one side of the brain).
At the end of the 6 month study, the UPDRS clinical scores GAD-treatment group had decreased by 8·1 points (indicating a slight improvement), but the control group also demonstrated a 4·7 points decrease (suggesting that a placebo effect might have occurred). Overall, the clinical effect observed in this blind test of the new treatment was considered rather mild.
The researchers persevered though, and conducted another assessment at 12 months post treatment. This report provides the results of that analysis:
 Title: Long-term follow-up of a randomized AAV2-GAD gene therapy trial for Parkinson’s disease.
Title: Long-term follow-up of a randomized AAV2-GAD gene therapy trial for Parkinson’s disease.
Authors: Niethammer M, Tang CC, LeWitt PA, Rezai AR, Leehey MA, Ojemann SG, Flaherty AW, Eskandar EN, Kostyk SK, Sarkar A, Siddiqui MS, Tatter SB, Schwalb JM, Poston KL, Henderson JM, Kurlan RM, Richard IH, Sapan CV, Eidelberg D, During MJ, Kaplitt MG, Feigin A.
Journal: JCI Insight. 2017 Apr 6;2(7):e90133.
PMID: 28405611 (This report is OPEN ACCESS if you would like to read it)
This study represented a follow on to the Phase II study which was only 6 months long. In this additional study, the Phase II participants were followed to 12 months post treatment, and the researchers reported that the benefits observed in the treatment group persisted. In addition, the duration of levodopa-induced dyskinesias (one of the side effects of long term Levodopa treatment – click here to read more about dyskinesias) had also declined at the 12 month timepoint in the treatment group compared to the control group.
Brain scan analysis at 12 months revealed significant metabolic declines in multiple regions of the brain (from the thalamus and striatum to many areas of the cortes) in the treatment group compared with the control group – suggesting a reduction in the level of dysfunction caused by the over active subthalamic nucleus. This might all sound impressive, but the results were still considered mild for what the investigators had been hoping for. And although this Phase II study was not powered for efficacy (that is, it didn’t have enough participants to determine if the treatment was having a clinically relevant effect), the company Neurologix decided to discontinue the development of this treatment at this point. A planned Phase III trial was cancelled and the company went into bankrupcy (Click here to read more about this).
Sounds ugly right?
Yeah. So what happened next?
Well, not much… until November of 2018, when this report was published:
 Title: Gene therapy reduces Parkinson’s disease symptoms by reorganizing functional brain connectivity
Title: Gene therapy reduces Parkinson’s disease symptoms by reorganizing functional brain connectivity
Authors: Niethammer M, Tang CC, Vo A, Nguyen N, Spetsieris P, Dhawan V, Ma Y, Small M, Feigin A, During MJ, Kaplitt MG, Eidelberg D.
Journal: Sci Transl Med. 2018 Nov 28;10(469).
PMID: 30487248
In this study, some of the researchers who conducted the Phase II study were interested in understanding what were the precise mechanisms underlying the clinical improvements that they observed in the GAD-gene therapy treatment group. To do this, they turned to metabolic network analysis.
What is metabolic network analysis
A metabolic network shows all of the interactions between components of a specific biological system (such as a cell, or the brain).
 A metablic network. Source: Cell
A metablic network. Source: Cell
A metabolic network is a reconstruction of a biological system that collects all of the relevant metabolic information and compiles it in a mathematical model, which can be compared and analysed.
Throughout the Phase II trial, the participants were having their brains scanned. These multiple scans across the study allowed the researchers to observe changes over time. Using longitudinal FDG PET scans of brain glucose metabolism, the researchers found that the GAD-gene therapy treatment group had a new brain network, which was not observed in control/placebo group.
Hang on a second. What is an FDG PET scan?
PET is short for Positron Emission Tomography. It is a nuclear medicine technique in which a radioactive (unstable) substance is injected into the body, and can be used to visualise different parts/functions of the body.
 The priniciples of PET imaging. Source: Priyalearthproject
The priniciples of PET imaging. Source: Priyalearthproject
By attached radioactive substances to specific molecules, investigators can identify where in the body those molecules are and whether the amount/distribution differs from the norm.
Fluorodeoxyglucose (or FDG) is basically a radiolabelled sugar molecule. It acts in a very similar manner to glucose – being taken up by cells like glucose – and in this fashion it can be used to assess where glucose is being utilised in the body. Such measurements can be used to assess changes in the metabolic activity of the body. For example, given that cancer tumors are high consumers of glucose, FDG is often used to determine where in the body a tumor might be (as in the image below)
FDG imaging of a cancer tumor in the body. Source: Researchgate
Ok, so this imaging technique allowed the researchers to determine that GAD treatment group had a new brain network?
Yes.
Using FDG PET, researchers can generate what is called the “Parkinson’s specific disease-related spatial covariance pattern” (or PDRP) of regional brain glucose metabolism.
 PDRP. Source: Researchgate
PDRP. Source: Researchgate
There is evidence to suggest that the patten of the PDRP network can be used to distinguish PD patients from healthy controls (Click here to read more about this). The PDRP can also be used to differentiate between individuals with Parkinson’s and conditions similar to Parkinson’s (such as Multiple Systems Atrophy or MSA) – click here to read more about this.
When the researchers looked at the PDRP of the GAD-gene therapy treatment group and the control groups, they were curious to see what differences could account for the clinical improvements observed in the treatment group.
But here’s the thing: there was little if any difference.
The PDRP of both groups continued to get worse over time.
But the researchers did notice a new network of activated areas in the GAD-gene therapy group, which they called the GAD-related covariance pattern (GADRP), which appears in the 6 month data, but becomes strengthened in the 12 month data.
As the image below illustrates, when the researchers looked at differences in glucose activity in the brain, and then formed a metabolic network of the data, they found significant differences between the networks in the brains of the GAD-gene therapy treatment group and the control group.
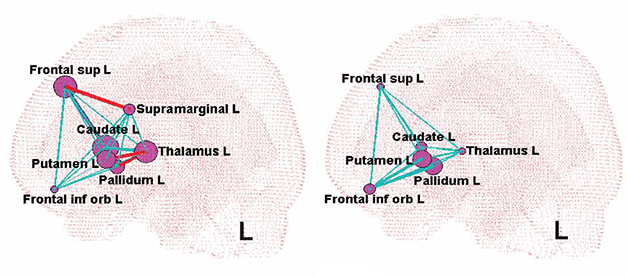 Differences in networks between treated participants (left) & controls (right). Source: Alzforum
Differences in networks between treated participants (left) & controls (right). Source: Alzforum
Specifically, by 12 months post treatment, the GAD-gene therapy group had five new discrete connections (including connections to the supramarginal gyrus in the parietal lobe). The expansion of the GADRP over the time frame of the study also correlated with the pattern of improvements observed in the clinical features.
Do we see this in deep brain stimulation?
So the researchers asked this very question: do individuals who have DBS have a similar pattern (or change) of metabolic activity in their brains following the implantation of the electrodes?
Longitudinal analysis of brain imaging data suggested that the GAD-gene therapy brain imaging data differed significantly to those from DBS patients. That is to say, the individuals with DBS did not have GADRP pattern of the GAD-gene therapy treated group.
Thus, in this clinical trial gene therapy appears to have activated a new network in the brain. Its increase in strength correlated with the improvements observed in clinical motor features, and this brain imaging approach could potentially serve as a measure of efficacy in future trials.
Sounds not-so-ugly, right?
Very interesting. So what happens now?
Given these new findings, the investigators are now planning a new clinical trial of GAD-gene therapy which will begin in late 2019. A company called MeiraGTx is apparently taking on this project (reading between the lines from a recent press release – click here to read the press release)
 There is currently limited information about this new clinical trial, but we will keep our ear to the ground here at the SoPD and let you know when we hear something.
There is currently limited information about this new clinical trial, but we will keep our ear to the ground here at the SoPD and let you know when we hear something.
So what does it all mean?
Researchers in the USA have provided evidence that an experimental gene therapy approach that was clinically tested for Parkinson’s may have had the unexpected consequences of forming new networks in the brain (“adaptive rewiring” as the investigators referred to the phenomenon). And the effect not only correlates with improvements in clinical features, but also appears to be specific to the gene therapy approach, as deep brain stimulation of the same targetted brain region does not have the same impact. The findings highlights both a novel effect and a potentially new method of evaluating outcomes in clinical trials.
While this new finding is tremendously exciting and I see a big future ahead for this technology, there is still a long way to go in the development of gene therapy. We currently have multiple gene therapy clinical trials being conducted for Parkinson’s – each exploring new innovations (from method of injection to the type of genomic material being transferred). And it will be interesting to see how the next clinical trial of this GAD-gene therapy approach is adapted/improved upon moving forward.
Stay tuned.
The banner for today’s post was sourced from positivelyaware


Simon said: “There is currently limited information about this new clinical trial …”.
Indeed. In fact, I couldn’t find any mention of it in the MeiraGTx press release. The only hint I could find of a future trial was the following sentence: “AAV-GAD has received Fast Track designation from the U.S. Food and Drug Administration (FDA).”
An article on the AlzForum website says that it will be a Phase 3 trial, but I guess that will need to be confirmed.
“The findings have sparked plans by a new company to take the therapy into a Phase 3 trial.”
“Based on these new imaging data, a British gene therapy company has acquired the rights to the AAV2-GAD therapy …”
https://www.alzforum.org/news/research-news/imaging-data-resurrects-abandoned-parkinsons-gene-therapy
LikeLike