|
This post is a game of two halves. The first half will explain the concept of a surgical procedure for Parkinson’s called ‘subthalamic deep brain stimulation‘, in which doctors permenantly implant electrodes into the brain to stimulate a region – the subthalamic nucleus. By stimulating this region with electrical impulses, doctors can provide a better quality of life (in most cases) to people with severe features of Parkinson’s. In the second half of this post, we will look at an approach to doing the same thing,… but without the electrodes. Rather, researchers are using gene therapy. In today’s post, we will discuss what deep brain stimulation is, what gene therapy is, and how the gene therapy approach is having a different kind of impact on the brain to that of deep brain stimulation.
|

Source: Youtube
Welcome to the first half of today’s post.
It begins with you asking the question:
What is deep brain stimulation?
Deep brain stimulation (or DBS) is a treatment method that involves embedding electrodes into the brain to help modulate the brain activity involved in movement.
It is a prodcedure that is usually offered to people with Parkinson’s who have excessive tremor or debilitating dyskinesias.
First introduced in 1987, deep brain stimulation consists of three components: the pulse generator, an extension wire, and the leads (which the electrodes are attached to). All of these components are implanted inside the body. The system is turned on, programmed and turned off remotely.
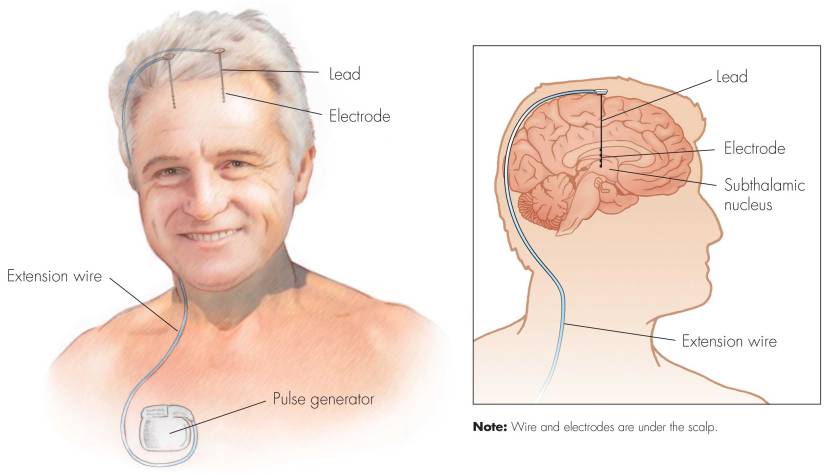
Source: Ucdmc