|
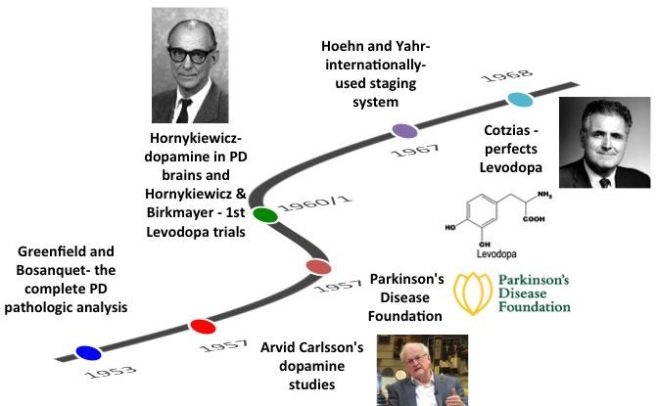
# # # # This week it was announced that Oleh Hornykiewicz had passed away. I appreciate that most readers will not know who he is, but understand that his contribution to Parkinson’s research was important. Not only was he instrumental in the discovery that dopamine is significantly reduced in the Parkinsonian brain, but he also demonstrated that levodopa treatment can help restore function. In today’s post, we remember Oleh Hornykiewicz. # # # # |
It was sad to hear of the passing away of Oleh Hornykiewicz this week.
Most readers will have little clue as to who he was, but he played a very important role in the development of the Parkinson’s treatment we know of as levodopa therapy.
Very early in the 20th century, a chemical called dopamine was discovered, but no one really knew anything about it until a young Swedish research named Arvid Carlsson started to play with it.

Prof Arvid Carlsson. Source: Alchetron
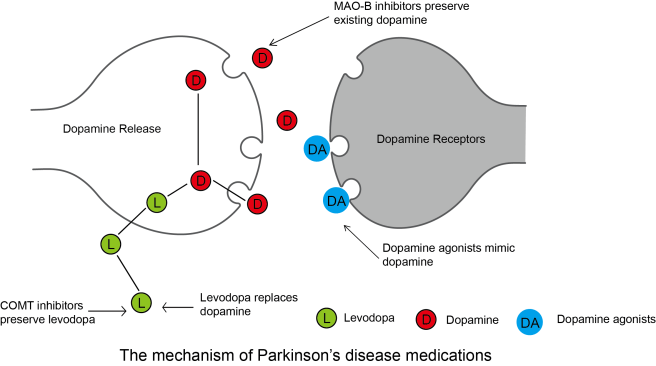
In 1957, Carlsson discovered that when he injected a drug called reserpine into the brains of rabbits, the animals exhibited limited ability to move. He found that reserpine depleted levels of dopamine in the brains of the rabbits. He also discovered that by injecting the dopamine precursor – levodopa (more on this below) – into those same animals, he was able to rescue their motor ability. Importantly, he found that the precursor (called 5-hydroxytryptophan) to another chemical called serotonin, it was not capable of reversing the reduction in motor ability, indicating that the effect was specific to levodopa and dopamine.
He published this amazing result in the prestigeous scientific journal ‘Nature’:

Title: 3,4-Dihydroxyphenylalanine and 5-hydroxytryptophan as reserpine antagonists.
Authors: Carlsson A, Lindqvist M, Magnusson T.
Journal: Nature. 1957 Nov 30;180(4596):1200. No abstract available.
PMID: 13483658 (the article on the Nature website – access required)
This was a fantastic discovery.
But what to do with it?
And that is where an Austrian researcher named Oleh Hornykiewicz becomes part of the story.