|
# # # # Dyskinesias are involuntary muscle movements associated with long-term use of levodopa therapy (use of levodopa is not a certainty for developing dyskinesias, but there is an association). A better understanding of the underlying biology of dyskinesias is required in order to alleviate this condition for those affected by it. This week researchers reported that a single protein – called RasGRP1 – plays a central role in the development of dykinesias, raising hope that agents targeting this protein could identified and provide better quality of life of sufferers. In today’s post, we will discuss what dyskinesias are and review the new research. # # # # |
Few people outside of the biomedical sciences may have heard of the Scripps Research Institute, but it is the largest private, not-for-profit medical research facility in the United States and among the largest in the world. It is headquartered in La Jolla, California but it has a sister facility in Jupiter, Florida.
 Nice spot to do research. Source: Scripps
Nice spot to do research. Source: Scripps
Collectively, “The Scripps” has 250 laboratories, which employs over 2,400 scientists, technicians, graduate students, and administrative staff.
It was founded in 1924 by journalist/philanthropist Ellen Browning Scripps.
 Ellen Browning Scripps. Source: Lajollalight
Ellen Browning Scripps. Source: Lajollalight
The Scripps covers a wide variety of area in biomedical research, but this week a group of researcher led by scientists at the Florida Scripps institute published an interesting report on Parkinson’s:
 Title: RasGRP1 is a causal factor in the development of l-DOPA–induced dyskinesia in Parkinson’s disease
Title: RasGRP1 is a causal factor in the development of l-DOPA–induced dyskinesia in Parkinson’s disease
Authors: Eshraghi M, Ramírez-Jarquín1 UM, Shahani1 N, Nuzzo T, De Rosa A, Swarnkar S, Galli N, Rivera O, Tsaprailis G, Scharager-Tapia C, Crynen G, Li Q, Thiolat ML, Bezard E, Usiello A, Subramaniam S
Journal: Science Advances, May 2020, 6, 18, eaaz7001
PMID: 32426479 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in proteins that could be playing a major role in the development of dyskinesias.
What are dyskinesias?
Dyskinesias (from Greek: dys – abnormal; and kinēsis – motion, movement) are a category of movement disorders that are characterised by involuntary muscle movements. And they are certainly not specific to Parkinson’s.
But in the case of Parkinson’s, dyskinesias have generally been believed to be associated with long-term use of levodopa (also known as Sinemet or Madopar – levodopa with carbidopa and levodopa with benserazide, respectively).

Sinemet is levodopa. Source: Drugs
NOTE: Long-term use of levodopa is not a certainty for developing dyskinesias, but there is an association. It will differ from person to person.
As for how they develop, there is a lot of debate over this topic, but there are some basic details that researchers generally tend to agree on.
Before being diagnosed and beginning a course of levodopa, the locomotion parts of the brain in a person with Parkinson’s gradually becomes more and more inhibited. This increasing level of inhibition results in the slowness and difficulty in initiating movement that characterises this condition.
A person with Parkinson’s may want to move, but they can’t – they are inhibited. In effect, they are akinetic (from Greek: a-, not, without; and kinēsis – motion).

Drawing of an akinetic individual with Parkinson’s, by Sir William Richard Gowers
Source: Wikipedia
Once inside the brain, levodopa is quickly converted into dopamine. It is changed into dopamine by an enzyme called DOPA decarboxylase, and this change rapidly increases the levels of dopamine in the brain, allowing the locomotion parts of the brain to function more normally.

The chemical conversion of L-DOPA to dopamine. Source: Nootrobox
In understanding this process, it is important to appreciate that when an levodopa tablet is consumed and levodopa enters the brain, there is a rapid increase in the levels of dopamine. This ‘spike’ in the supply of dopamine will last for the next few hours, before the dopamine is eventually used up.
As the effects of the levodopa tablet wear off, another tablet will be required. This use of multiple levodopa pills across the day gives rise to a wave-like shape to the dopamine levels in the brain over the course of the day (see the figure below). The first pill in the morning will quickly lift the levels of dopamine enough that the individual will no longer feel akinetic. This will allow them to be able to function with normal controlled movement for several hours before the levodopa begins to wear off. As the levodopa wears off, the dopamine levels in the brain drop back towards levels that will leave the person feeling akinetic and at this point another levodopa tablet is required.
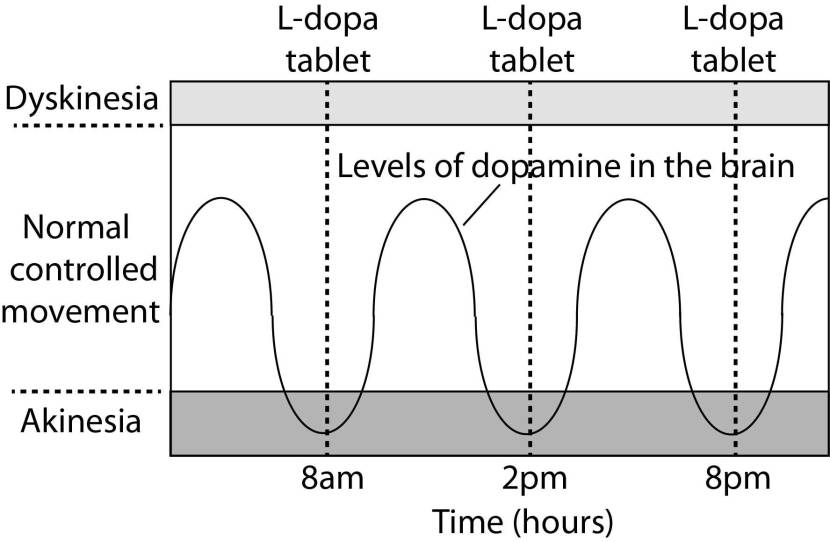
A hypothetical illustration of dopamine levels over a day
After several years of levodopa use, many people with Parkinson’s will experience a weaker response to each tablet. They will also find that they have more time during which they will be unable to move (exhibiting akinesia). This is simply the result of the slow progression of Parkinson’s – levodopa treats the motor features of the condition but only hides/masks the fact that the disease is still progressing.
To combat this shorter response time, the dose of levodopa is usually increased. This will result in increasing levels of dopamine in the brain (as illustrated by the higher wave form over time in the image below). Gradually it will take more levodopa medication-induced dopamine to lift the individual out of the akinetic state.

Again this illustration is hypothetical (situation differs between individuals)
This increasing of levodopa dosage, however, results in too much dopamine being present in the brain at times. And this situation is often associated with the gradual development of abnormal involuntary movements that appear when the levels of levodopa induced dopamine are the highest.
These are the involuntary muscle movements that we refer to as dyskinesias.
|
# RECAP #1: Dyskinesias are involuntary muscle movements associated with the long-term use of levodopa therapy. They represent a very troublesome aspect of life for some individuals who have been living with Parkinson’s for some time. The biology of dyskinesias is not entirely clear, but researchers around the world are trying to determine underlying pathways that can be therapeutically targetted to improve quality of life for sufferers. # |
Ok, so what does the new research report suggest?
In their study, the researchers were interested in a protein called RasGRP1.
What is RasGRP1?
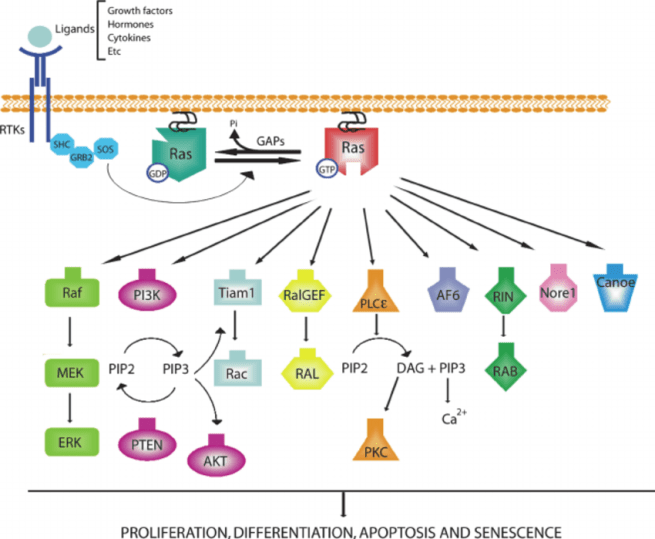
RAS guanyl-releasing protein 1 (or RasGRP1) is a protein that functions as an activator of another protein called RAS.
RAS proteins are tiny molecular switches that cycle between an active (or guanosine triphosphate (GTP)-bound) state and an inactive (guanosine diphosphate (GDP)-bound) state – see the image below.
RasGRP1 works by activating RAS.
When RAS is ‘switched on’ by RasGRP1, it subsequently activates other proteins, which in turn initiate all kinds of important biological activities.
Lots of things happen when RAS is activated. Source: Researchgate
Why were they interested in RasGRP1?
So this story really starts back in 2011, when this report was published:
 Title: Rhes, a striatal-enriched small G protein, mediates mTOR signaling and L-DOPA-induced dyskinesia.
Title: Rhes, a striatal-enriched small G protein, mediates mTOR signaling and L-DOPA-induced dyskinesia.
Authors: Subramaniam S, Napolitano F, Mealer RG, Kim S, Errico F, Barrow R, Shahani N, Tyagi R, Snyder SH, Usiello A.
Journal: Nat Neurosci. 2011 Dec 18;15(2):191-3. doi: 10.1038/nn.2994.
PMID: 22179112 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that another protein called Rhes influenced levels of dyskinesia. Like RasGRP1, Rhes also affects RAS activity. Importantly, however, when Rhes was lowered in mice, their levodopa-induced dyskinesias were also reduced. And critically, this effect did not change the motor improvement to levodopa treatment, suggesting to the researchers that Rhes could be a therapeutic target for treating dyskinesia.
Interesting side note here: the investigators also reported that Rhes binds to and activates mTOR (Click here to read a previous SoPD post on this topic). Their results suggest that agents that block Rhes from binding to mTOR, can reduce dyskinesias – thus is will be interesting to see if the TORC1 inhibitor, RTB101 being clinically tested in Parkinson’s in New Zealand by the biotech company resTORbio Inc has any affect on dyskinesias (or even delaying the onset of them).
The researchers explored this Rhes research further and in 2016 they published this report:
 Title: RasGRP1 promotes amphetamine-induced motor behavior through a Rhes interaction network (“Rhesactome”) in the striatum.
Title: RasGRP1 promotes amphetamine-induced motor behavior through a Rhes interaction network (“Rhesactome”) in the striatum.
Authors: Shahani N, Swarnkar S, Giovinazzo V, Morgenweck J, Bohn LM, Scharager-Tapia C, Pascal B, Martinez-Acedo P, Khare K, Subramaniam S.
Journal: Sci Signal. 2016 Nov 15;9(454):ra111.
PMID: 27902448 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers highlighted a protein called RasGRP1 (yes, that RAS guanyl-releasing protein 1 – the same protein we were discussing above). They reported that RasGRP1 interacts with and stabilises Rhes, in effect promoting an abundance of the protein and enhancing its effect.
|
# # RECAP #2: Researchers have reported that reducing levodopa-induced dyskinesias can be achieved by lowering levels of a protein called Rhes. Additional research found that another protein called RasGRP1 binds to Rhes and regulates its function. # # |
And this brings us back to the new research report.
So what did the discover in the new research report?
In the recently published report, the investigators took a good look at RasGRP1 in the context of levodopa-induced dyskinesias:
 Title: RasGRP1 is a causal factor in the development of l-DOPA–induced dyskinesia in Parkinson’s disease
Title: RasGRP1 is a causal factor in the development of l-DOPA–induced dyskinesia in Parkinson’s disease
Authors: Eshraghi M, Ramírez-Jarquín1 UM, Shahani1 N, Nuzzo T, De Rosa A, Swarnkar S, Galli N, Rivera O, Tsaprailis G, Scharager-Tapia C, Crynen G, Li Q, Thiolat ML, Bezard E, Usiello A, Subramaniam S
Journal: Science Advances, May 2020, 6, 18, eaaz7001
PMID: N/A (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that levodopa treatment caused an increase in RasGRP1 levels in the striatum (the region of the brain where the chemical dopamine is released) of both mouse and primate models of Parkinson’s.
The investigators next tested if levodopa-induced dyskinesias could be caused in mice with no RasGRP1. They bred some mice that had been genetically engineered to not produce the RasGRP1 protein, and they then induced a model of Parkinson’s and started treating the mice with high levels of levodopa.
Remarkably, the researchers found that the absence of RasGRP1 protein dramatically reduced levodopa-induced dyskinesias, without interfering with the therapeutic effects of the levodopa treatment. As you can see in the image below, while normal/wild-type (WT) mice gradually developed higher abnormal involuntary movements (or AIMs) scores as the days of high dose levodopa treatment continued. But in the mice with no RasGRP1, this effect was significantly reduced.
 Source: Science
Source: Science
Another side note: curiously, these mice with no RasGRP1 were fertile and had no significant changes in their levels of motor activity, or any undesirable issues/side effects elsewhere in the body – suggesting that RasGRP1 could be a good target for therapeutic intervention.
The researchers concluded their study by suggesting that drugs reducing “RasGRP1 in adults may produce beneficial effects against levodopa-induced dyskinesias without inducing serious adverse effects“.
|
# # # RECAP #3: Researchers reported that levodopa treatment causes an increase in RasGRP1 levels in the brain, in both mouse and primate models of Parkinson’s. They also found that reducing RasGRP1 levels significantly reduced levodopa-induced dyskinesias in mice. # # # |
Has anyone else ever reported these findings before?
Yes, independent research groups have previously reported increases in RasGRP1 (also known as CalDAG-GEFII) levels in the case of levodopa-induced dyskinesias:
 Title: Dysregulation of CalDAG-GEFI and CalDAG-GEFII predicts the severity of motor side-effects induced by anti-parkinsonian therapy.
Title: Dysregulation of CalDAG-GEFI and CalDAG-GEFII predicts the severity of motor side-effects induced by anti-parkinsonian therapy.
Authors: Crittenden JR, Cantuti-Castelvetri I, Saka E, Keller-McGandy CE, Hernandez LF, Kett LR, Young AB, Standaert DG, Graybiel AM.
Journal: Proc Natl Acad Sci U S A. 2009 Feb 24;106(8):2892-6.
PMID: 19171906 (This report is OPEN ACCESS if you would like to read it)
There have also been reports corroborating reducing Rhes levels also reduces the severity of dyskinesias (Click here to read more about this).
But this is the first time researchers have zeroed in on RasGRP1 as a potential causal factor in dyskinesias.
It is interesting to note, however, that inhibition of another RasGRF1 (Ras protein-specific guanine nucleotide-releasing factor 1 – different from RasGRP1) also reduces levodopa-induced dyskinesia:
 Title: Inhibition of Ras-guanine nucleotide-releasing factor 1 (Ras-GRF1) signaling in the striatum reverts motor symptoms associated with L-dopa-induced dyskinesia.
Title: Inhibition of Ras-guanine nucleotide-releasing factor 1 (Ras-GRF1) signaling in the striatum reverts motor symptoms associated with L-dopa-induced dyskinesia.
Authors: Fasano S, Bezard E, D’Antoni A, Francardo V, Indrigo M, Qin L, Doveró S, Cerovic M, Cenci MA, Brambilla R.
Journal: Proc Natl Acad Sci U S A. 2010 Dec 14;107(50):21824-9.
PMID: 21115823 (This report is OPEN ACCESS if you would like to read it)
Like RasGRP1, RasGRF1 is also involved in the conversion of Ras from an inactive (GDP-bound) form to an active (GTP-bound) form. And in this study, the researchers reported that genetically engineered mice (that produce no RasGRF1 protein) exhibited significantly less dyskinesias when exposed to high dose levodopa (compared to normal mice). And this did not affect the beneficial effects of levodopa on motor ability.
In addition, in a primate model of Parkinson’s, a gene therapy treatment that reduced RasGRF1 levels also reduced levodopa-induced dyskinesias (again without affecting the positive treatment effects of levopdopa).
The researchers concluded that a therapy combining Ras-GRF1 partial inhibition may provide an effective anti-dyskinetic approach.
Interesting, so what happens next?
I am not sure. But at a guess, two things.
- I am assuming that the Scripps researchers will be wanting to dig further into the biology of this research to better understand it, and
- I am also guessing that they will also be seeking to identify drugs that can target and reduce RasGRP1 levels, which could then be tested against dyskinesias (the researcher suggest this is a press release about their study – click here to read that).
How will they do that?
There are many different ways to screen for or develop therapeutic agents, but it is interesting to note that the Scripps Institute has a high throughput drug screening facility that is hopefully now attempting to identify inhibitors of RasGRP1.
 Source: Scripps
Source: Scripps
So what does it all mean?
Levodopa-induced dyskinesias are one of the most debilitating aspects of Parkinson’s for a lot of sufferers, and there are currently a limited number of treatments to help alleviate these involuntary movements. Recently, researchers in the USA have identified a new aspect of the underlying biology of dyskinesia, which provides new insights but may also point towards future potential therapies.
While disease modification remains challenging for Parkinson’s, it is encouraging that significant efforts are being made to deal with some of the more troublesome aspects of living with the condition. And hopefully, these projects will bear fruit in the not too distant future.

All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from kindredproductions



Is there evidence that a lower dose of levodopa May have a reduced incidence of LIDs? Hopefully with the advent of restorative therapies like AADC/neuron replacement, and the consequent reduction in the amount of necessary medication, we should be able to reduce the LIDs. Thanks for working towards joblessness Simon. We all appreciate it
LikeLike
ras was one of the first oncogenes to be discovered, maybe 30 years ago. Mutated ras that remains GTP-liganded is associated with tumorigenesis. While Scripps’ intervention acts to diminuish GTP-ras, I do wonder what effects ras inhibition would have on normal cell replication. Ras’ normal functions are extensive: https://en.wikipedia.org/wiki/Ras_GTPase
LikeLiked by 1 person
While I have no specific knowledge of the normal function of Ras, philosophically I prefer treatments that act at a more fundamental level (i.e., earlier in the causal chain) to those which manipulate things downstream through means that may generate additional side-effects, and thus I share your concerns about the possible effects inhibiting Ras. My own approach has been to preserve the population of dopamine neurons for as long as possible (e.g., through exercise and anti-inflammatory, antioxidant and mitochondrial supporting foods and supplements). Although both approaches may have benefits.
LikeLike
I follow the same approach as you. One component is described here: https://www.mdpi.com/2076-3425/10/5/284
LikeLiked by 1 person
My own understanding of “levodopa-induced” dyskinesias is that they are fundamentally caused by the reduction of the population of dopamine neurons that occurs over the course of the illness.
I have seen a rodent study that indicates that, for rats at least, the axon terminals of dopamine neurons are capable of holding about 2.5 times the amount of dopamine that they hold under physiological conditions. When levodopa is converted by HT5 neurons into dopamine, that dopamine is then imported via the dopamine transporter (DAT) to the axon terminals of the patient’s remaining dopamine neurons, where it is packaged into vesicles and released under the direct control of the firings of those remaining dopamine neurons. This means that a reduced population of neurons can compensate for their damaged and destroyed peers by functioning at a higher level.
This buffering of dopamine from exogenous levodopa not only prevents ambient dopamine levels in the striatum from reaching the excessive levels associated with dyskinesias–it also allows dopamine to be released via the same mechanisms as release it in the non-Parkinsonian brain, which has other benefits, including the more normal functioning of dopamine-based reward mechanisms.
As the population of dopamine neurons declines over the course of the illness, this buffering is reduced, and more of the dopamine from levodopa builds up in the extra-cellular space, stimulating dopamine receptors on an ongoing and uncontrolled basis that fluctuates with the dosing cycle, as you have described in this article. This is what, in my own limited understanding, causes dyskinesias to result–not the treatment with levodopa itself.
And yes, there are higher doses of levodopa required in later stages of the illness, but I would attribute that to the lack of endogenous generating capacity (due to the reduction in the dopamine neuron population), and also to the inferior effects of ambient striatal dopamine, as compared to dopamine that is directly released into a synaptic juncture by the patient’s surviving dopamine neurons.
So when I read about this association of RasGRP1 with dyskinesias, my intuition is that this is an effect that occurs *downstream* of what I have described above. In other words, could it be that the specific *ways* that excess dopamine in the inter-cellular space of the striatum result in dyskinesias *requires* RasGRP1, and that is why suppressing RasGRP1 results in the reduction of dyskinesias?
Even if I am correct, this finding seems like good news for LID sufferers, for exactly the reasons that you have described. But I would question whether long-term levodopa use is what *causes* dyskinesias to increase over the course of the illness, even if levodopa, when used in later stages of PD, does have the acute effect of increasing RasGRP1, and RasGRP1 inhibition is able to suppress the dyskinesias.
I think this is an important question, because I hear patients still asking whether they should delay levodopa therapy in order to delay the development of dyskinesias, and what I presently tell them is, no, don’t delay if you need it to function more normally–instead, do everything you can to preserve your remaining dopamine neurons.
So, is that bad advice? Is there actual evidence that long-term levodopa therapy *causes* dyskinesias, independent of the decline of the dopamine neuron population?
LikeLike