|
The biotech company Acorda Therapeutics Inc. yesterday announced that it was halting new recruitment for the phase III program of its drug Tozadenant (an oral adenosine A2a receptor antagonist).
In addition, participants currently enrolled in the trial will now have their blood monitoring conducted on a weekly basis.
The initial report looks really bad (tragically five people have died), but does this tragic news mean that the drug should be disregarded?
In todays post, we will look at what adenosine A2a receptor antagonists are, how they may help with Parkinson’s, and discuss what has happened with this particular trial.
|

Dr Ron Cohen, CEO of Acorda. Source: EndpointNews
Founded in 1995, Acorda Therapeutics Ltd is a biotechnology company that is focused on developing therapies that restore function and improve the lives of people with neurological disorders, particularly Parkinson’s disease.
Earlier this year, they had positive results in their phase III clinical trial of Inbrija (formerly known as CVT-301 – Click here to read a previous post about this). They have subsequently filed a New Drug Application with the US Food and Drug Administration (FDA) to make this inhalable form of L-dopa available in the clinic, but the application has been delayed due to manufacturing concerns from the FDA (Click here to read more about this). These issues should be solvable – the company and the FDA are working together on these matters – and the product will hopefully be available in the new year.
So what was the news yesterday?
Acorda Therapeutics has another experimental product going through the clinical trial process for Parkinson’s disease.
It’s called Tozadenant.
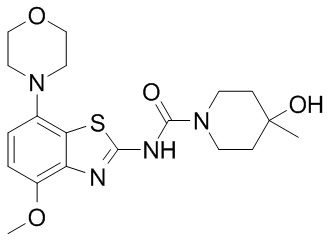
Source: Focusbio
Tozadenant is an oral adenosine A2a receptor antagonist (and yes, we’ll discuss what all that means in a moment).
Yesterday Acorda Therapeutics Inc announced that they have halted new recruitment for their phase III clinical program. In addition the company is increasing the frequency of blood cell count monitoring (from monthly to weekly) for participants already enrolled in the company’s Phase 3 program of Tozadenant for Parkinson’s disease.
The Company took this action due to reports of cases of agranulocytosis.
Continue reading “The Acorda’s Tozadenant Phase III clinical trials” →