Exciting new last week from a small biotech company called Voyager Therapeutics which is using gene therapy to treat neurodegenerative disease. Their primary product (VY-AADC01) is focused on Parkinson’s disease and the initial results look very positive.
The press release has indicates that the treatment is well tolerated and has beneficial effects on the subject’s motor functions. This last part is very interesting as the trial is being conducted on people with advanced Parkinson’s disease.
In today’s post, we’ll review the technology and what the results mean.

Gene therapy. Source: HuffingtonPost
In Parkinson’s disease, we often talk about the loss of the dopamine neurons in the midbrain as a cardinal feature of the disease. When people are diagnosed with Parkinson’s disease, they have usually lost approximately 50-60% of the dopamine neurons in an area of the brain called the substantia nigra.

The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinson’s disease brain (right). Source: Memorangapp
The midbrain is – as the label suggests – in the middle of the brain, just above the brainstem (see image below). The substantia nigra dopamine neurons reside there.

Location of the substantia nigra in the midbrain. Source: Memorylossonline
The dopamine neurons of the substantia nigra generate dopamine and release that chemical in different areas of the brain. The primary regions of that release are areas of the brain called the putamen and the Caudate nucleus. The dopamine neurons of the substantia nigra have long projections (or axons) that extend a long way across the brain to the putamen and caudate nucleus, so that dopamine can be released there.

The projections of the substantia nigra dopamine neurons. Source: MyBrainNotes
In Parkinson’s disease, these ‘axon’ extensions that project to the putamen and caudate nucleus gradually disappear as the dopamine neurons of the substantia nigra are lost. When one looks at brain sections of the putamen after the axons have been labelled with a dark staining technique, this reduction in axons is very apparent over time, especially when compared to a healthy control brain.
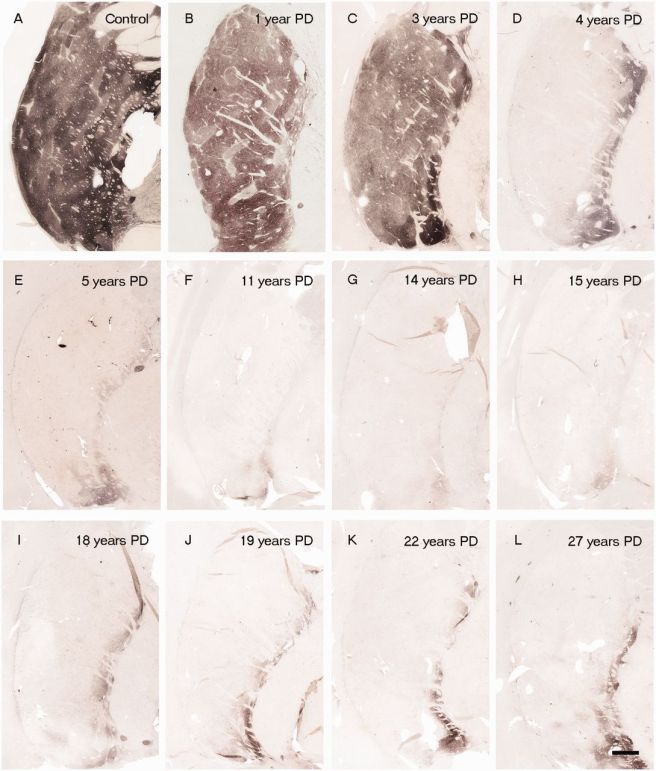
The putamen in Parkinson’s disease (across time). Source: Brain
Previously we have discussed replacing the loss dopamine by transplanting dopamine producing cells into the putamen (click here to read that post), but some researchers now believe that this is not necessary. Instead they have proposed using gene therapy for Parkinson’s disease.
What is gene therapy?
The gene therapy involves inducing cells to produce proteins that they usually do not. This is usually done using genetically modified viruses which have had all the disease causing component removed, allowing us to use the virus as an efficient delivery system. Viruses by their very nature are very good at infecting cells, so if we remove the disease causing components, what is left is a very effective delivery system. Taking this approach one step further, we could next take genes involved with dopamine synthesis and insert them into our empty virus. By then injecting this virus into the brain, we could produce dopamine in any infected cells (it’s slightly more complicated than that, but you get the basic idea).
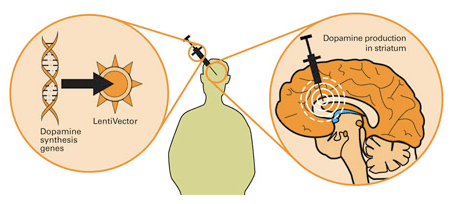
Gene therapy for Parkinson’s disease. Source: Wiki.Epfl
This approach demonstrated amazing results in preclinical studies in the lab, but the transition to the clinic has not been easy (click here for a good review of the field).
What has been done in the clinic for gene therapy and Parkinson’s disease?
The first clinical attempt at gene therapy for Parkinson’s disease involved injecting a virus containing a gene called glutamic acid decarboxylase (GAD), which is an enzyme involved in the production of a chemical called GABA. The virus was injected into an area of the brain called the subthalamic nucleus, which becomes over-active in Parkinson’s disease. By ectopically producing GAD in the subthalamic nucleus, researchers were able to reduce the level of activity (this is similar to deep brain stimulation in Parkinson’s disease which modulates the activity of the subthalamic nucleus). The clinical trials for GAD produced modest results. The virus was well tolerated, but the clinical effect was limited.
Another clinical trial attempted to cause cells in the putamen to produce a chemical called neurturin (which is very similar to GDNF – we have previously written about GDNF, click here to read that post). The goal of the study was to prove neuroprotection and regeneration to the remaining dopamine neurons, by releasing neurturin in the putamen. Subjects were injected in the putamen with the virus and then the participants were followed for 15 months. Unfortunately, this study failed to demonstrate any meaningful improvement in subjects with Parkinson’s disease.
So what were the results of the trial?
Voyager Therapeutics‘s gene therapy product, VY-AADC01 is an adeno associated virus that carries a gene called Aromatic L-amino acid decarboxylase (or AADC).
AAV Viruses. Source: HuffingtonPost
Yeah, I know: what is AADC?
AADC is the enzyme that converts L-dopa into dopamine. L-dopa can be naturally produced in the brain from Tyrosine that is absorbed from the blood. It is also the basic component of many treatments for Parkinson’s disease.

The production of dopamine. Source: Slideplayer
By injecting VY-AADC01 into the putamen of people with advanced Parkinson’s disease, Voyager is hoping to alleviate the motor features of the condition by allowing the brain to produce a constant supply of dopamine in the exact location that is missing the dopamine (remember, the putamen is where dopamine is released). This approach will not cure the disease, but it may make life a lot easier for those affected by it.
The phase 1b clinical trial was designed to assess whether the virus had any negative side effects in humans. After the subjects were injected in the brain with VY-AADC01, they were assessed at six and twelve months after the surgery. The results suggest that the virus was well tolerated and resulted in increased AADC enzyme activity, enhanced response to L-dopa treatment, and clinically meaningful improvements in various measures of patients’ motor function (44% improvement in ‘off medication’ measures and 55% improvement in ‘on medication’ measures).
The company currently has 2 groups of subjects injected with the virus (two different concentrations) and they are looking to have a third group injected in early 2017. Phase 2 trials are planned to begin in late 2017.
What does it all mean?
Well, this is a very interesting result and bodes well for other companies taking similar gene therapy approaches (these include Oxford BioMedica and Genepod Therapeutics).
They are also interesting results because the subjects involved in the study all have advanced Parkinson’s disease (the average time since diagnosis in the subject was 10 years). So it is very positive news to see beneficial effects in later stage subjects.
Our ability to delivery of genes to different locations is a symbol of how far we have come with our understanding of biology. The fact that this knowledge is now having a positive impact in the medical world is very exciting. Gene therapy is one treatment approach that we here at SoPD are very excited about and watching very closely.
The banner for today’s post was sourced from Voyager Therapeutics
 The biotech firm presented data at the meeting regarding their gene therapy approach for Parkinson’s.
The biotech firm presented data at the meeting regarding their gene therapy approach for Parkinson’s.