A major trend in experimental medicine at present is ‘immunotherapy‘ – stimulating or reprogramming the immune system to help fight particular diseases.
A research group in Nebraska have attempted to use this approach for Parkinson’s disease, and recently they have published some very interesting clinical trial results.
In today’s post, we will discuss the science and review the results of their research.

Nebraska. Source: The Toast
Here at the SoPD HQ, we like surprises.
And when several readers contacted us about some interesting results from a new clinical trial for Parkinson’s disease that we knew nothing about, we were rather ‘OMG! What a fantastic surprise!’ about it.
The results stem from a clinical trial that has taken a rather different approach to tackling Parkinson’s disease: boosting the immune system to help fight off the condition. And rather than simply covering up the symptoms, the drug being tested may actually slow down the condition.
You may have heard about this trial as the results of this clinical study have attracted the attention of the media:
So what was the new clinical trial all about?
Let’s start with the context of the study. You see, it took place in the great US state of Nebraska.
Interesting place Nebraska.
Nebraska (in red). Source: Wikipedia
The birth place of actors Fred Astaire and Marlon Brando.
And home to the largest porch swing in the world (holds 18 adults or 24 children – amazing).

The world’s largest swing chair. Source: Pinterest
Nebraska is also one of the top agricultural states in the USA, with about 93% of the land being used for farming. And approximately 40% of the state’s population (750,000 out of 1.8 million) lives in those rural areas. As a result of this largely rural population, there are probably a lot of people in Nebraska being exposed to pesticide and insecticides (in the air they breath and the water they drink).
This exposure is believed to be one of the reasons why Nebraska has one of the highest rates of Parkinson’s disease in the USA.
There are approximately 330 people per 100,000 of the general population living with Parkinson’s Disease in Nebraska (Click here for more on this). Compare that with just 180 people per 100,000 of the UK general population having Parkinson’s Disease (Click here for more on this).
As a result of this statistic, Parkinson’s disease is taken very seriously in Nebraska.
Back in 1996, Nebraska became the first state to create a Parkinson’s disease registry. They also have tremendous support groups for the Parkinson’s community (such as Parkinson’s Nebraska).

There is also a lot of Parkinson’s disease research being conducted there.
And this brings us to the clinical study results we are going to discuss:

Title:Evaluation of the safety and immunomodulatory effects of sargramostim in a randomized, double-blind phase 1 clinical Parkinson’s disease trial
Authors: Gendelman HE, Zhang Y, Santamaria P, Olson KE, Schutt CR, Bhatti D, Shetty BLD, Lu Y, Estes KA, Standaert DG, Heinrichs-Graham E, Larson L, Meza JL, Follett M, Forsberg E, Siuzdak G, Wilson TW, Peterson C, & Mosley RL
Journal: npj Parkinson’s Disease (2017) 3, 10.
PMID: N/A (This article is OPEN ACCESS if you would like to read it)
For this randomised, double-blind phase 1 clinical trial, the researchers enrolled 20 people with Parkinson’s disease and 17 age-matched non-Parkinsonian control subjects. The people with Parkinson’s disease ranged in age from 53 to 76 years (mean age of 64) and they had had symptoms for 3–14 years (the mean was 7 years). Both the Parkinson’s disease group and control group were monitored for 2 months before the trial started in order to establish baseline measurements and profiles.
The Parkinson’s disease group were then randomly assigned into two equal sized groups (10 subjects each) and they were then self-administered (by self-injection) either sargramostim (6 μg/kg/day) or a placebo control solution (saline) for 56 days (click here to see the details of the clinical trial).
Hang on a second, what is Sargramostim?
Sargramostim (marketed by the pharmaceutical company Genzyme under the tradename ‘Leukine’) is an Food and Drug Administration (FDA) -approved recombinant granulocyte macrophage colony-stimulating factor (GM-CSF) that functions as an immunostimulator.
What…on earth…..does any of that….actually mean?
Ok, so Food and Drug Administration (FDA) -approved means that this drug is safe to use in humans. Sargramostim is currently widely used in bone marrow transplantation procedures, to stimulate the production of new blood cells.
‘Recombinant‘ basically means that we are talking about an artificially produced protein.
‘Granulocyte macrophage colony-stimulating factor‘ is an actual protein that our bodies produce. GM-CSF is a small protein that is secreted by various types of cells in our body, and it functions as a cytokine. And yes, I know what you are going to ask:
What’s a cytokine?
Cytokines (from the Greek: kýtos meaning ‘container, body, cell’; and kī́nēsis meaning ‘movement’) are small proteins that are secreted by certain cells in the body and they have an effect on other cells. Cytokines are a method of communication for cells.
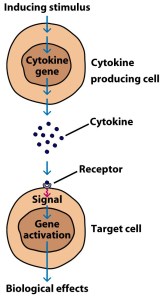
How cytokines work. Source: SBS
Granulocyte macrophage colony-stimulating factor is secreted by various cells around the body to communicate with the immune system that something is wrong. In it’s actually function, GM-CSF acts as a white blood cell growth factor, or a stimulant of white blood cell production.

GM-CSF stimulates blood stem cells into production. Source: Oxymed
Why are white blood cells important?
While red blood cells are principally involved with the delivery of oxygen to the various parts of the body, the white blood cells (also referred to as leukocytes or leucocytes), are the cells of your immune system that protect your body against both infectious disease and foreign invaders.
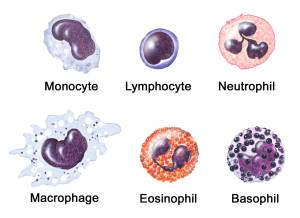
6 types of white blood cells. Source: Stfranciscare
GM-CSF stimulates blood stem cells to produce more neutrophils, eosinophils, basophils, and monocytes (all types of white blood cells – see image above). Monocytes then migrate towards the tissue affected by the injury or disease, where they then mature into macrophages and dendritic cells (Macrophages are large, specialised cells that are responsible for removing damaged target cells).
Once at the site of trouble, macrophages produce pro-inflammatory neurotoxins that help to destroy unhealthy or damaged cells, making them easier to engulf and dispose of. The problem is that those released neurotoxins can also damage surrounding healthy cells.
Given that GM-CSF stimulates this kind of activity, you are probably wondering why researchers would be giving Sargramostim to folks with Parkinson’s disease.
But GM-CSF also does something else that is really interesting:
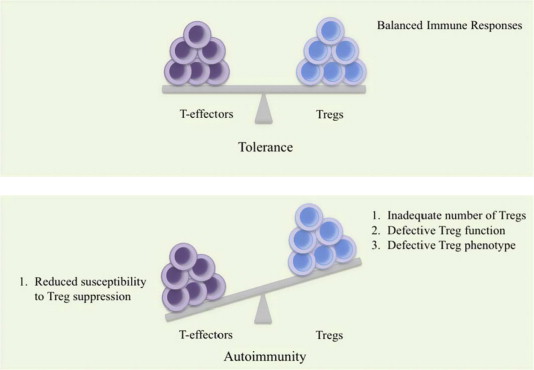
GM-CSF stimulates regulatory T (Treg) cells.
What are regulatory T cells?
Regulatory T (Treg) cells maintain order in the immune system. They do this by enforcing a dominant negative regulation on other immune cells, particularly other T-cells.
T-cells are a type of white blood cell that circulate around our bodies, scanning for cellular abnormalities and infections.
Think of T-cells as the inquisitive neighbours curious about and snooping around a local crime scene, and then imagine that Treg cells are the police telling them “nothing to see here, move along”.

Tregs maintaining order. Source: Keywordsuggestions
Treg cells are particularly important for calming down effector T cells (or T-eff cells). These are several different types of T cell types that ‘actively’ respond to a stimulus. They include:
- Helper T cells (TH cells) which assist other white blood cells in the immunological process
- Killer T cells which destroy virus-infected cells, tumor cells, and are involve in transplant rejection.
The normal situation in the body is to have a balance between T-eff cells and Treg cells. If there are too many T-eff cells, there is increased chances of autoimmunity – or the immune system attacking healthy cells.
A delicate balance between healthy and autoimmune disease. Source: Researchgate
Too many Treg cells is not a good situation either, however, as they would leave the immune system suppressed and individuals vulnerable to disease.
How are Treg cells involved with Parkinson’s disease?
So, in Parkinson’s disease, researchers believe that the build up of the Parkinson’s associated protein, alpha synuclein may be toxic and killing certain cells in the brain (such as the dopamine neurons). When the cell dies and the alpha synuclein is released into the surrounding environment of the brain, it most likely does two things:
- irritates and activates the resident immune cells, called microglia
- activates the wider immune system, resulting in T-cell infiltration of the brain
The T-cells snoop around, detect that something isn’t quite right and then release their own cytokines which further activates the microglia. The microglia then release pro-inflammatory toxic chemicals which indiscriminately damage the unhealthy and healthy cells in the local area.

A.) The normal situation in PD; B.) the situation after GM-CSF treatment. Source: NCBI
Now the hypothesis is that GM-CSF may be able mediate this degenerative cycle by stimulating the induction of Treg cells, which can calm the activated microglia down, return it to a resting state and the healthy surrounding neurons survive intact.
Is there any research evidence for this effect in models of Parkinson’s disease?
Yes there is.
The group in Nebraska have actually been working ‘pre-clinically’ on this idea for some time:

Title: Neuroprotective activities of CD4+CD25+ regulatory T cells in an animal model ofParkinson’s disease.
Authors: Reynolds AD, Banerjee R, Liu J, Gendelman HE, Mosley RL.
Journal: J Leukoc Biol. 2007 Nov;82(5):1083-94.
PMID: 17675560
In this study, the researchers demonstrated that by increasing the number of activated Treg cells in neurotoxin (MPTP)-injected mice, they could produce a greater than 90% level of protection of the dopamine neurons when compared to mice that did not receive the increase of Treg cells.
The Treg cells were found to mediate this neuroprotection through suppression of the microglial response to the neurotoxin. The investigators concluded that their data strongly supported the use of immunomodulation as a strategy for treating Parkinson’s.
They next extended these findings by looking at whether GM-CSF could provide neuroprotection in the same model of Parkinson’s disease:

Title: GM-CSF induces neuroprotective and anti-inflammatory responses in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine intoxicated mice.
Authors: Kosloski LM, Kosmacek EA, Olson KE, Mosley RL, Gendelman HE.
Journal: J Neuroimmunol. 2013 Dec 15;265(1-2):1-10.
PMID: 24210793 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers gave GM-CSF prior to the neurotoxin (MPTP) which kills dopamine neurons. GM-CSF freely cross the blood-brain barrier which inhibits a lot of other drugs from entering the brain. This treatment protected the dopamine neurons and the investigators found increased Treg induction and reduced activation of the microglia cells.
This neuroprotective effect could also transferred between animals. Treg cells from GM-CSF treated mice were transferred to MPTP-treated mice and neuroprotection of the dopamine neurons was observed in those animals. The researchers concluded that the results provide evidence that GM-CSF modulation of the immune system could be of clinical benefit for people with Parkinson’s disease.
And they are not the only investigators who have demonstrated this. In addition to the work produced by the Nebraskan research team, other research groups have also observed beneficial effects of GM-CSF in models of Parkinson’s disease (Click here, here and here to read some of those reports).
In fact, for a very good OPEN ACCESS review on the topic of immunomodulation for Parkinson’s disease – click here.
And with all of this research backing them, the team in Nebraska decided to move GM-CSF towards the clinic with a small phase I clinical trial.

The Nebraska team: Dr Howard Gendelman, Dr Pamela Santamaria & Prof R. Lee Mosley. Source: Omaha
What did they find in the clinical trial?
In their randomized, double-blind, phase 1 clinical trial of 20 people with Parkinson’s disease taking either sargramostim (10 subjects) or a placebo control solution (10 subjects) for 56 days, the researchers found that Sargramostim firstly increases the the induction of Treg cells, and mediated suppression of the immune cells
More importantly, the sargramostim treated group demonstrated a modest improvement in their motor performance scores after 6 and 8 weeks of treatment when compared with the placebo group. The study was not large enough in size or duration for robust conclusions to be made, but the deviation between the two groups in motor scores in intriguing. This is particularly curious given that the sargramostim treatment group returned to a similar level of performance as the control (placebo) group at the 8 week assessment when they were no longer on sargramostim:
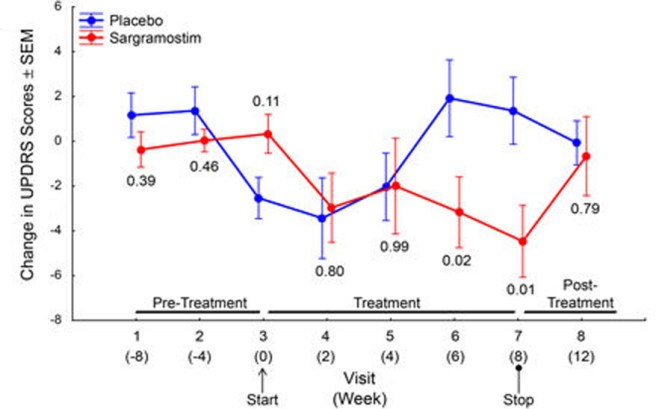
Change in motor scores of the participants. Source: Nature
One of the interesting features of this study was that the participants were a mixed bunch with regards to their Parkinson’s disease. The participants ranged in age from 53 to 76 years (mean age of 64) and they had had symptoms for 3–14 years (the mean was 7 years). It would be interesting to know (in a larger study) if there is any difference in the effect of this treatment based on length of time since diagnosis.
Another interesting aspect of the study is that it was double-blind. It is rather rare for a phase I clinical study to be double-blind, as they are usually just testing safety and tolerance. But given that sargramostim is used in the clinic already, the investigators had more flexibility with the study design. The double blind nature of the results only makes the findings more intriguing though.
The next step in this research is to plan a larger clinical study in 1-2 years time. The delay is caused by the desire for that trial to focus on an oral tablet (currently Sargramostim is only administered via an injection – not a popular route!). Those follow up studies will require groups taking different doses of the drug to get a better idea of effective dosages.
So what does it all mean?
Artificial modulation of the immune system represents tremendous opportunities for not only Parkinson’s disease, but also other conditions such as Alzheimer’s disease and amyotrophic lateral sclerosis. Recently, some researchers have concluded a clinical study of immunomodulation for Parkinson’s disease after almost 20 years of preclinical experimentation. The results are very interesting and may provide us with a novel method of treating the condition.
We here at the SoPD will be interested to see if Sargramostim makes it through the clinical testing process alone (as a “mono-therapy”) for Parkinson’s disease, or whether it will be used in combination with other drugs. One potential issue for this approach is that it leaves the individual with a suppressed immune system to defend them against other infectious agents.
Having said that, the fact that this approach may work could also tell us a great deal about the nature of Parkinson’s disease itself, and raising the idea that the body’s immune response could be involved with the progression of this neurodegenerative condition. We already know from several studies that certain anti-inflammation drugs (particularly Ibuprofen) can help to lower the risk of developing Parkinson’s disease (Click here for more on Ibuprofen).
Perhaps while we wait for the pill version of Sargramostim, a separate Ibuprofen study could be conducted to determine if this drug could slow down the progression of the disease.
The banner for today’s post was sourced from Diamond