|
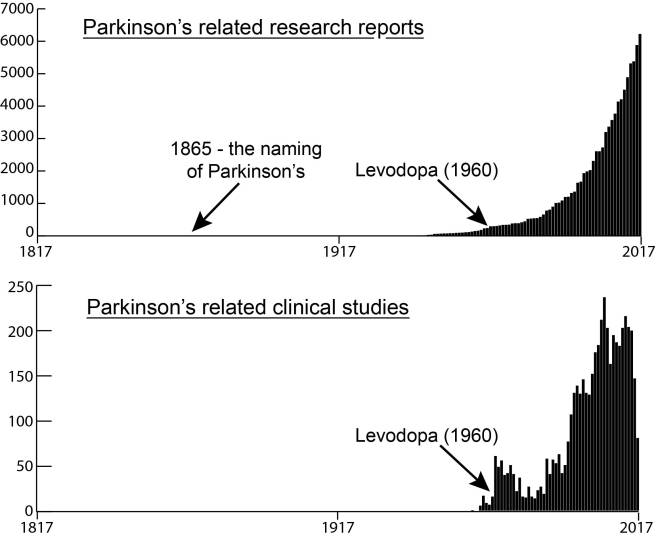
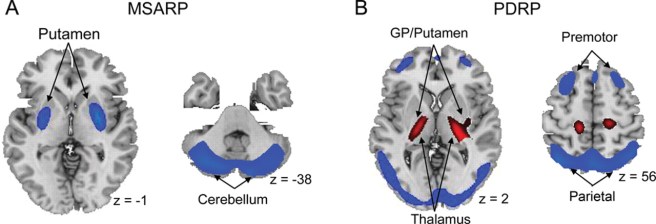
There is a lot of clinical and biological similarities between the neurodegenerative conditions of Parkinson’s and multiple systems atrophy (or MSA). Recently, however, researchers have published a report suggesting that these two conditions may be differentiated from each other using a technique analysing protein in the cerebrospinal fluid – the liquid surrounding the brain, that can be accessed via a lumbar puncture. Specifically, the method differentiates between different forms of a protein called alpha synuclein, which is associated with both conditions. In today’s post, we will look at what multiple systems atrophy (MSA) is, discuss how this differentiating technique works, and explore what it could mean for people with either of these conditions.
|
 Source: Assessment
Source: Assessment
Getting a diagnosis of Parkinson’s can be a tricky thing.
For many members of the affected community, it is a long and protracted process.
Firstly, there will be multiple visits with doctors and neurologists (and perhaps some brain imaging) until one is finally given a diagnosis of PD. There are a number of conditions that look very similar to Parkinson’s, which must be ruled out before a definitive diagnosis can be proposed.
But even after being diagnosed, there are a group of conditions that look almost identical to Parkinson’s. And many people will be given a diagnosis of Parkinson’s before they are then given a corrected diagnosis of one of these other conditions.
Can you give me an example of one of these other conditions?
Sure. A good example is multiple systems atrophy.
What is Multiple System Atrophy?