|
‘Parkinsonisms’ refer to a group of neurological conditions that cause movement features similar to those observed in Parkinson’s disease. They include multiple system atrophy (MSA) and Progressive supranuclear palsy (PSP) and idiopathic Parkinson’s. Newly published research now shines a light on a possible mechanism for differentiating between multiple system atrophy and idiopathic Parkinson’s. In today’s post we will look at what multiple system atrophy is, review the new research report, and discuss what these results could mean for the Parkinson’s community. |
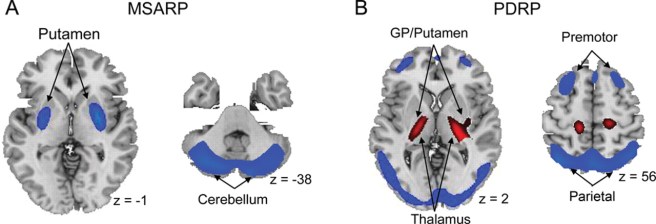
Brain immaging of multiple system atrophy–related spatial covariance pattern (MSARP) and Parkinson disease–related spatial covariance pattern (PDRP). Source: Neurology
For a long time I have been looking to write a piece of Multiple system atrophy.
I have been contacted by several readers asking for more information about it, and the only thing really delaying me – other than the tsunami of Parkinson’s related research that I am currently trying to write posts for – was the lack of a really interesting piece of research to base the post around.
Guess what came into my inbox yesterday:
Title: Familial Parkinson’s point mutation abolishes multiple system atrophy prion replication.
Authors: Woerman AL, Kazmi SA, Patel S, Aoyagi A, Oehler A, Widjaja K, Mordes DA, Olson SH, Prusiner SB.
Journal: Proc Natl Acad Sci U S A. 2017 Dec 26. pii: 201719369.
PMID: 29279394
This is a really interesting piece of research, that continues a line of other really interesting research.
And if it is independently replicated and verified, it will have massive implications for the Parkinson’s community, particularly those affected by Multiple System Atrophy.
But before we deal with that, let’s start with the obvious question:
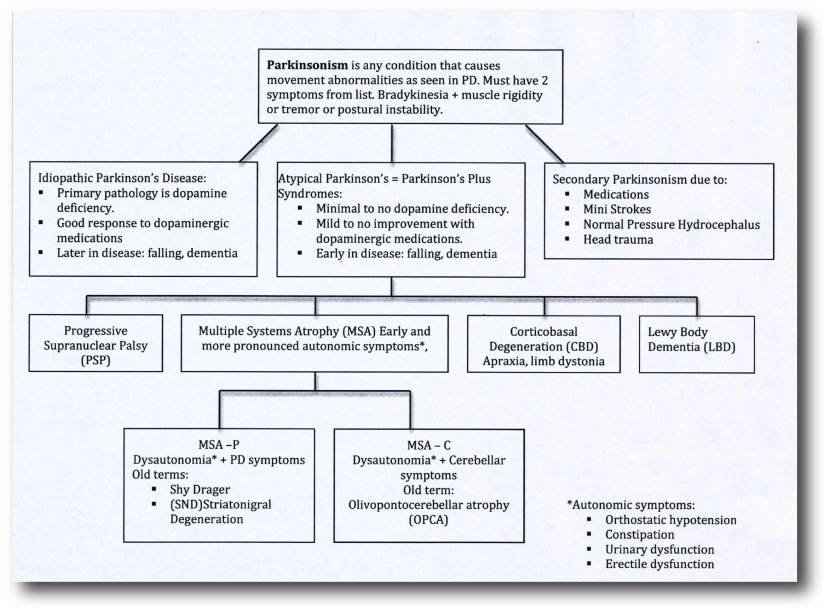
What is Multiple System Atrophy?
Multiple System Atrophy (MSA), also known as Shy-Drager syndrome, is a rare neurodegenerative condition. It is considered an Atypical Parkinsonism.
What is an ‘Atypical Parkinsonism’?
‘Parkinsonisms’ refer to a group of neurological conditions that cause movement features similar to those observed in Parkinson’s disease, such as tremors, slow movement and stiffness. The name ‘Parkinsonisms’ is often used as an umbrella term that covers Parkinson’s disease and all of the other ‘Parkinsonisms’.
Parkinsonisms are generally divided into three groups:
- Classical idiopathic Parkinson’s (the common spontaneous form of the condition)
- Atypical Parkinson’s or Parkinson-plus syndromes (such as multiple system atrophy and Progressive supranuclear palsy (PSP))
- Secondary Parkinson’s (which can be brought on by mini strokes (aka Vascular Parkinson’s), drugs, head trauma, etc)
Source: Parkinsonspt
Some forms of Parkinsonisms that at associated with genetic risk factors, such as juvenile onset Parkinson’s, are considered atypical. But as our understanding of the genetics risk factors increases, we may find that an increasing number of idiopathic Parkinson’s cases have an underlying genetic component (especially where there is a long family history of the condition) which could alter the structure of our list of Parkinsonisms.
So what is the difference between Multiple system atrophy and idiopathic Parkinson’s?
When a person first presents at the clinic with rigidity, slowness of movement and a resting tremor, it can be very difficult to differentiate between classical idiopathic Parkinson’s and other kinds of Parkinsonisms. But there are some telltale signs that can help differentiate the Multiple System Atrophy from idiopathic Parkinson’s:
- The disability progresses more rapidly in MSA
- People with MSA are poor responders to levodopa treatment over time (such as Sinemet)
- Urinary retention and orthostatic hypotension (blood pressure falls when suddenly standing up) are common
- Rigidity and bradykinesia are out of proportion to tremor
- Speech is severely affected
- Gasping and high pitched wheezing sounds when breathing are present
- In the vast majority of cases, there is an absence of dementia, though other cognitive functions can be affected
- Reduced blinking and dry eye, in addition to jerky or slower eye movements
And the Second Consensus Statement on the diagnosis of multiple system atrophy provides for two basic types of multiple system atrophy, based on the symptoms of the condition at the time of evaluation.
These two types are:
- MSA with predominant parkinsonism (MSA-P), which can resemble idiopathic Parkinson’s because of slow movement and stiff muscles. The terms striatonigral degeneration or parkinsonian variant are sometimes used for this category of MSA.
- MSA with cerebellar features (MSA-C). It is sometimes termed ‘sporadic olivopontocerebellar atrophy’ (or OPCA). MSA-C primarily affects balance, coordination, and speech.
Some researchers believe that there is also a third type of MSA which is a combination of the two.
How is Multiple system atrophy diagnosed?
Currently there is no test that can definitively make or confirm the diagnosis of MSA in a living person.
Having said that, regular use of brain imaging techniques such as MRI and CT scanning can show a decrease in the size of specific brain structures affected in MSA (such as the cerebellum and pons). For example, in the image below you can see serial MRI images of the brain of a person with MSA-C over a 2-year period. Note the subtle reduction in the size of the characteristic ‘hot cross bun’ shape of the pons between image C (2004) and two years in image D (2006):
Serial MRI of brain of a person with MSA-C over a 2-year period. Source: Openi
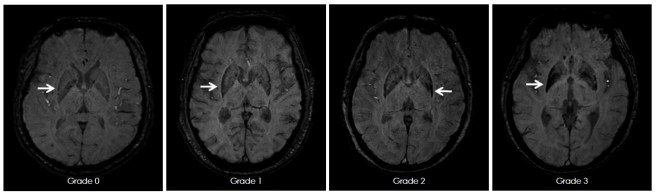
And in MRI brain scan images from individuals affected by MSA-P, there is a gradual darkening of the region of the brain called the putamen (indicated on the left side of the brain by a white arrow in the image below). You can see that over time as the condition progresses (from grade 0 to grade 3), the putamen becomes darker and darker.
MRI of brain of a person with MSA-P over time. Source: e-jmd
What causes Multiple system atrophy?
I think most researchers would agree that the correct answer to this question is:
Source: Wellbeing365
Having said that, there is now a growing pile of evidence that a prion form of the Parkinson’s associated protein Alpha Synuclein may have a causal influence in this condition.
What does ‘prion’ mean?
A prion is an infectious agent that is entirely protein based.
There is no DNA/RNA as in the case of an infectious virus, prions are simply proteins that have gone rogue.
Proteins ‘go rogue’ when they don’t fold properly. In order to do their particular job correctly, each protein must fold itself in particular ways to achieve a specific shape. Proteins are tiny machines and each type of protein has a different shape (many of them have several different shapes that they can adopt). The take-home message, however, is that if they do not fold correctly, they can not do their job.
For a good explanation of protein folding, watch this video:
Prions are proteins that present alternative configurations to their normal shape, but the critical distinguishing feature of prions is that the protein becomes self-propagating. That is to say, the alternative folding is transmissible to other prion-prone proteins. One protein that has gone rogue (and become a prion) can encourage another protein to go rogue. And this ultimately leads to a cascade that very much resemble the spread of a viral infection.
How does this work?
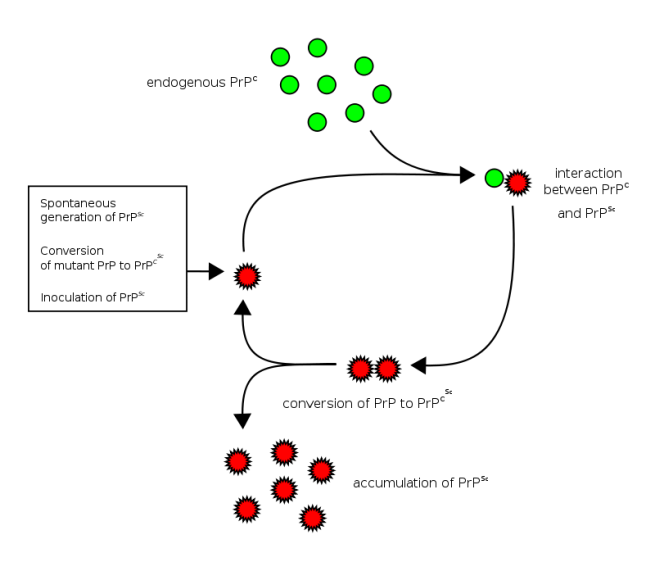
The basic idea is that a Prion prone Protein (PrP) will sit inside a cell having various functions which it conducts in a normal set of folded configurations. This normal state of the protein is called PrPC (C refers to ‘cellular’). Then one of two things could happen: 1. the PrPC will spontaneous take on the infectious form of the protein, which is called PrPSc (Sc refers to ‘scrapie’, the prototypic prion disease occurring in sheep); or 2. the PrPC will bump into a PrPSc which will then induce the PrPC to fold into the PrPSc configuration, thus causing the disease to spread.
Prion life-cycle. Source: Wikipedia
The most well researched example of a prion condition is Creutzfeldt-Jakob disease. This video will explain for you what CJD is:
So researchers think that alpha synuclein could be a prion-prone protein?
There is certainly evidence to suggest that alpha synuclein can act like a prion.
When a protein is produced (by stringing together amino acids in a specific order set out by the RNA instructions), it will then be folded into a functional shape that do a particular job (as we have discussed above).
Alpha synuclein is slightly different in this respect. It is normally referred as a ‘natively unfolded protein’, in that is does not have a defined structure. Alone, it will look like this:
Alpha synuclein. Source: Wikipedia
By itself, alpha synuclein is considered a monomer, or a single molecule that will bind to other molecules to form an oligomer (a collection of a certain number of monomers in a specific structure). In Parkinson’s disease, alpha-synuclein also aggregates to form what are called ‘fibrils’.

Microscopic images of Alpha Synuclein (AS) monomers, oligomers and fibrils. Source: Brain
Oligomer versions of alpha-synuclein are emerging as having a key role in Parkinson’s disease. They lead to the generation of fibrils and may cause damage by themselves.

Source: Nature
It is believed that the oligomer versions of alpha-synuclein is being passed between cells – and this is how the disease may be progressing – and once inside the cell, these oligomer versions of alpha synuclein are causing other ‘natively unfolded protein’ monomer versions of alpha synuclein to become oligomers, which either bind with other oligomers to form fibrils or move on to another cell.
Hopefully you can see why some researchers are suggesting that alpha synuclein has prion-like properties, causing a chain-reaction that gives rise to Parkinsonisms like MSA and idiopathic Parkinson’s.
Is Alpha Synuclein a prion? Source: PNAS
For a good review of the alpha synuclein prion theory – Click here.
Is there any evidence of this in Multiple system atrophy?
Not directly, but there is certainly evidence of lab-based experiments.
In 2013, this research report was published:
Title: Transmission of multiple system atrophy prions to transgenic mice.
Authors: Watts JC, Giles K, Oehler A, Middleton L, Dexter DT, Gentleman SM, DeArmond SJ, Prusiner SB.
Journal: Proc Natl Acad Sci U S A. 2013 Nov 26;110(48):19555-60.
PMID: 24218576 (This research report is OPEN ACCESS if you would like to read it)
In this research report, the investigators injected protein collected from the brains of people who passed away with multiple system atrophy into genetically engineered mice. The mice had one copy of mutant version of the human alpha synuclein (A53T) gene in their DNA. DNA is double stranded, meaning that there are two copies of each gene in a molecule of DNA – call this an evolutionary insurance policy, if something goes wrong with one of the copies, there is always a back up. And this system is evident in the mice with two copies of the mutant version of the human alpha synuclein (A53T) gene because they start developing neurological problems at about 10 months of age, while the mice with only one copy of mutant version of the human alpha synuclein gene remain healthy and normal…
…normal, that is, until they are injected with protein collected from the brains of people who passed away with multiple system atrophy (and this protein extracted from the brain contained various forms of alpha synuclein protein).
At approximately 100 days post injection, the single copy mice began to develop progressive signs of neurologic problems. The investigators concluded that their “results provide compelling evidence that alpha synuclein aggregates formed in the brains of MSA patients are transmissible and, as such, are prions”.
These same researchers then replicated this experiment, but this second time they compared protein extracted from the brains of people who passed away with MSA to protein extracted from the brains of people who passed away with Parkinson’s.
And guess what they found?
Title: Evidence for α-synuclein prions causing multiple system atrophy in humans with parkinsonism
Authors: Prusiner SB, Woerman AL, Mordes DA, Watts JC, Rampersaud R, Berry DB, Patel S, Oehler A, Lowe JK, Kravitz SN, Geschwind DH, Glidden DV, Halliday GM, Middleton LT, Gentleman SM, Grinberg LT, Giles K.
Journal: Proc Natl Acad Sci U S A. 2015 Sep 22;112(38):E5308-17.
PMID: 26324905 (This report is OPEN ACCESS if you would like to read it)
The researchers used the same mice that had one copy of mutant version of the human alpha synuclein (A53T) gene in their DNA, and they injected them with protein extracted from the brains of 14 cases of MSA (each case was treated as an independent sample of protein). In all of these cases, the mice developed neurological problems approximately 120 days after injection.
Then the investigators took protein extracted from the brains of 6 cases of Parkinson’s and injected them into fresh mice (that had one copy of mutant version of the human alpha synuclein gene) and…
(Drum roll please)
Nothing happened.
There were no neurological issue – out to beyond 200 days post injection.
The mice were fine.
And the researchers replicated this result in cells grown in culture: while the protein extracted from the brains of 14 cases of MSA readily caused alpha synuclein clustering in cells, the protein extracted from the brains of 6 cases of Parkinson’s did not (and nor did protein extracted from the brains of healthy control cases).
This result was interesting because a lot of the previous research on the prion theory of Parkinson’s has involved exposing cells in culture to the pre-formed alpha synuclein fibrils, and in those cases they had developed aggregated alpha synclein protein inside the cells. And in this MSA study, the researchers used pre-formed alpha synclein fibrils in their cell cultures and they also reported the clustering of alpha synuclein. But this indicates that the use of pre-formed fibrils in such experiments may be erroneous. Perhaps many of the previous prion theory of Parkinson’s experiments should be repeated using protein extracts from the brains of disease specific brains.
The researchers concluded that their “findings argue that MSA is caused by a unique strain of alpha synuclein prions, which is different from the putative prions causing PD”.
And they backed up this finding with the new research that they published this week:
Title: Familial Parkinson’s point mutation abolishes multiple system atrophy prion replication.
Authors: Woerman AL, Kazmi SA, Patel S, Aoyagi A, Oehler A, Widjaja K, Mordes DA, Olson SH, Prusiner SB.
Journal: Proc Natl Acad Sci U S A. 2017 Dec 26. pii: 201719369.
PMID: 29279394
In this research report, the investigators wanted to determine if genetic mutations in the alpha synuclein gene are involved in differentiating between Parkinson’s and MSA. In order to do this, they engineered cells that contained alpha synuclein which had a yellow fluorescent protein attached to it. In this way, the more alpha synuclein clustering observed in the cell, the more yellow fluorescence would be seen – this provided them with a useful, quantifiable measure of alpha synuclein aggregation.
The researchers next gave the cells different kinds of Parkinson’s associated alpha synuclein genetic mutations (such as A30P, E46K and A53T). In all there were 9 cell lines being tested (including a normal – no mutation – control cell line). They then took protein extracted from the brains of people who passed away with MSA and exposed all of the cells to these extracts.
All of the cell lines expressed robust yellow fluorescence… except one.
Cells that carried alpha synuclein with the E46K mutation did not.
This finding meant that MSA prions were unable to replicate in cells that have the E46K alpha synuclein mutation. The alpha synuclein protein that was produced by this mutated gene is not ‘prion prone’ – it could not be encouraged to go rogue and become a prion.
It also suggested that the MSA prions have a distinctive shape that will not cause problems in people with specific genetic mutations. It would be interesting to now go and have a look at the DNA of people with MSA and see if there is an absence of E46K alpha synuclein mutations in this population of people – there have been associations between genetic mutations in the alpha synuclein gene and MSA with cerebellar features (MSA-C), but I am unsure if the E46K variant was investigated in that study (Click here to read that research).
The researchers concluded that their findings confirm “that MSA alpha synuclein prions are conformationally distinct from the misfolded alpha synuclein in Parkinson’s”
It is interesting to note that the investigators finished their report by writing that “The ability to propagate a disease-relevant prion strain in cultured cells and mutant mice offers a significant approach to synucleinopathy drug discovery”, which indicates that one of their next research reports may provide a drug screen of some sort that could highlight compounds that reduce the levels of alpha synuclein aggregation following exposure to MSA prions.
That would be an extremely interesting result, with serious potential impact for the MSA community.
So what does it all mean?
How neurodegenerative conditions like Parkinson’s and Multiple System Atrophy develop is still a mystery.
New research published this week, however, provides further support for the idea that MSA could be a prion-like condition. Such information may be very useful for the MSA community as there are currently clinical trials of drugs and a vaccine that are targeting the toxic form of alpha synuclein (what the drugs target exactly is a patented secret).
The biotech companies AFFiRiS and Prothena are currently in Phase II trials for these treatments and we will hopefully be seeing some new results from those studies in the new year. These treatment approaches are trying to capture and remove the nasty version of the alpha synuclein protein, and by doing so this will hopefully slow down the progression of the condition.
If MSA is indeed a prion-like disease (these experimental results need to be independently replicated before we can conclude this), it will be interesting to see the results of the ongoing AFFiRiS clinical trial as they are testing their treatments on a cohort of people with MSA (Click here to read the latest press release from the company).
Above all else, the new results may provide us with a new method of differentiating Parkinson’s and MSA, as well as providing insight into the mechanisms behind the conditions.
Only time will tell.
One last note: Concerned readers may be asking themselves if alpha synuclein is a prion-like particle, then is MSA (or Parkinson’s) a transmissible condition? Could carers or partners be affected? To these readers, I can say, rest assured that no evidence of transmissibility in humans has ever been reported (Click here to read more about this). In addition, there is no epidemiological data suggesting a person-to-person spreading for Parkinson’s or MSA even in people with close contacts or who share a living environment as life partners (Click here to read more about this).
Yes, there have been extremely rare cases of ‘Conjugal parkinsonism’ where Parkinson’s like conditions have occurred in a married couple, but in almost every case there have been environmental elements that are believed to be the causative factors (such as multiple exposures to heavy metals or agricultural chemicals – Click here to read more about this).
All of the evidence suggests that MSA and Parkinson’s are not transmissible conditions.
The banner for today’s post was sourced from Youtube










This is a very interesting article and we were questioning that my husband had this but have now found out he has B cell follicular lymphoma a large tumour around the heart, aorta and into the spine t9-11 which explains a lot of the symptoms, but I must say that inserting A SPC was the best choice of urology management. This was only picked up by a ct angiogram as I noticed his normal pulse of 45 elevated to 76 and his normal bp Pd 90/60 went up to 110/70 and there it was wrapped around. Unfortunately PD is a complex disorder with multiple symptoms unique to the individual.
LikeLike
Dear Elaine,
Thanks you for your comment. Sorry to hear about your husband’s situation. I hope he is responding well to treatment. All the best.
Kind regards,
Simon
LikeLike