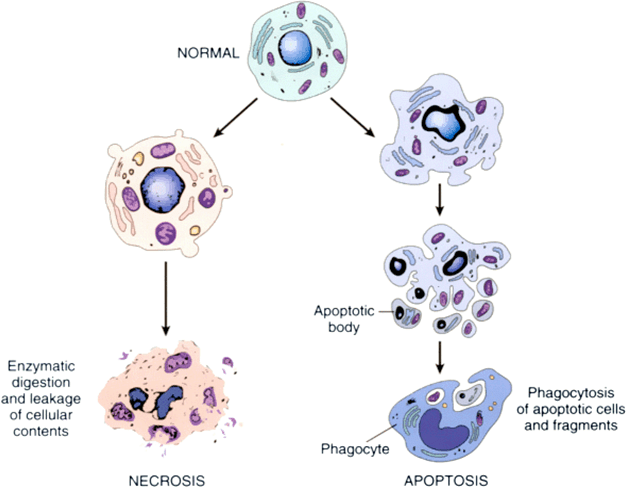
|
# # # # You may not realise it, but the DNA in your cells is under constant attack. All kinds of stressors (like the damaging effects of oxidative stress resulting from cellular processes) are constantly bombarding this precision molecule that contains the genetic blueprint for making and maintaining you. Luckily, millions of years of evolution has led to a complex and comprehensive DNA repair system that never takes holidays…. but might become a little slower as we age. Recently researchers have reported that certain aspects of this DNA repair system could be playing a role in Parkinson’s. In today’s post, we will review some new research in this area and consider the implications of the findings. # # # # |
My daughter is entering the pre-teen years, and I am struggling with all the horrors that that age brings.
Having survived the ‘Terrible Twos’ and the ‘Three-nager’ phase, I have absolutely adored innocence and magic of years 4 to 8. They were delightful. The ninth year, however, has brought with it the ominous arrival of (for lack of a better word) sass.
It has also involved a departure from the childhood songs (think Disney’s Lion King, Frozen, or Moana hits), and the introduction of more modern music, like her current favourite Meghan Trainor’s All about that Bass (see video above).
The next decade $£%#!& terrifies me.
But Meghan’s song provides an appropriate background for the subject matter of today’s post: Base excision repair
(Yeah, I know that’s a strange segway, but I’m tired and lacking imagination tonight)
What is Base excision repair?