|
The great ice hockey player Wayne Gretzky once said “A good hockey player plays where the puck is. A great hockey player plays where the puck is going to be” (the original quote actually came from his father, Walter). At the start of each year, it is a useful practise to layout what is planned for the next 12 months. This can help us better anticipate where ‘the puck’ will be, and allow us to prepare for things further ahead. 2017 was an incredible year for Parkinson’s research, and there is a lot already in place to suggest that 2018 is going to be just as good (if not better). In this post, we will lay out what we can expect over the next 12 months with regards to the Parkinson’s-related clinical trials research of new therapies. |
Charlie Munger (left) and Warren Buffett. Source: Youtube
Many readers will be familiar with the name Warren Buffett.
The charming, folksy “Oracle of Omaha” is one of the wealthiest men in the world. And he is well known for his witticisms about investing, business and life in general.
Warren Buffett. Source: Quickmeme
He regularly provides great one liners like:
“We look for three things [in good business leaders]: intelligence, energy, and integrity. If they don’t have the latter, then you should hope they don’t have the first two either. If someone doesn’t have integrity, then you want them to be dumb and lazy”
“Work for an organisation of people you admire, because it will turn you on. I always worry about people who say, ‘I’m going to do this for ten years; and if I really don’t like it very much, then I’ll do something else….’ That’s a little like saving up sex for your old age. Not a very good idea”
“Choosing your heroes is very important. Associate well, marry up and hope you find someone who doesn’t mind marrying down. It was a huge help to me”
Mr Buffett is wise and a very likeable chap.
Few people, however, are familiar with his business partner, Charlie Munger. And Charlie is my favourite of the pair.
Charlie Munger. Source: Upminded
A college drop-out who served as a meteorologist in the US Army Air Corps, before going to Harvard to study Law. At age 31, he met Warren at a dinner and convinced him to go into investing. And the rest is history.
When it comes to the one lines, Charlie gets to the point colder and quicker than Warren. And it was Charlie who provided one particular pearl that I hold on to:
The secret to happiness is to lower your expectations.
Easy to say for a billionaire, do I hear you mutter?
Source: Keepcalmandposters
Charlie is not for a moment suggesting that one should not be ambitious. Rather he is simply saying aim for the stars, but lower your expectations so that if you only reach the moon you won’t be disappointed. It would still be an amazing achievement.
I have taken Mr Munger’s quote one step further: I have no expectations.
My cup is completely empty. Thus, whenever anything happens, it’s magic! This particular strategy has served me well in scientific research where (if we are honest) few things ever seem to meet expectations.
Source: Unitystone
Now, having said all this: I do have some expectations for 2018
They are based on things that we know are coming. For example:
- Two new countries will emerge in the form of New Caledonia and Bougainville as they both hold independence referendums from France and Papua New Guinea.
- The FIFA football world cup will be held in Russia
- NASA will deploy the James Webb Space Telescope (which will be more powerful than the Hubble telescope)
- The results of the phase II GDNF clinical trial from Bristol will be published (see below).
In this post we will outline the things we can expect from Parkinson’s clinical research in 2018 :
I am going to frame this discussion around a common theme that I had in many of my public talks last year with regards to what a ‘cure for Parkinson’s’ will look like, for example:
That theme was: any ‘cure for Parkinson’s’ is going to require three components:
- A disease halting mechanism
- A neuroprotective agent
- Some form of cell replacement therapy
In this post we will explore each of these components individually and discuss what we should learn in 2018. Let’s look at the first component of any “Cure for Parkinson’s”:
1. A disease halting mechanism
Source: MyThaiLanguage
Parkinson’s is a progressive neurodegenerative condition. Thus, the first and most critical component of any ‘cure’ for Parkinson’s involves a treatment that will slow down/halt that progression of the condition.
This can be done either directly or indirectly.
The direct approach
You can think of the direct approach in military terms as a full frontal assault on the enemy. Going straight at the condition and trying to punch a hole through those enemy lines, which gets at the heart of the problem. A direct approach in halting Parkinson’s disease, however, requires a fundamental understanding of how the condition is actually progressing.
And if we are completely honest, we “are not there yet”. We do not know the mechanisms that underly the gradual progress of this condition.
But fear not.
As Prof George Church recently pointed out in an excellent interview, “we developed effective smallpox prevention back when we had close to zero understanding of virology and immunology”.
Prof George Church. Source: Biospace
Within the Parkinson’s field, we do have some good theories about how the condition may be spreading in the brain and they are worth testing while new ideas are being investigated.
One of those theories is the idea that a toxic form of the Parkinson’s-associated protein, alpha synuclein, could be being passed from cell to cell, and as it is absorbed by each new healthy cell it starts causing trouble which results in clustering of protein (leading to the appearance of Lewy bodies).
And there is some evidence for this as individuals who received cell transplants back in the 1990s have since passed away and their brains have been examined post-mortem. One very interesting finding from those brains is that some of the cells in the transplants (up to 10% in one case – Click here to read the research report on that case) have Lewy bodies in them. This suggests that condition is being passed on from the Parkinson’s affected brain to the healthy transplanted cells in some way. Researchers have proposed that the alpha synuclein protein may be the guilty party in this process.
Above are photos of neurons from the post-mortem brains of people with Parkinson’s that received transplants. White arrows in the images above indicate lewy bodies inside transplanted cells. Source: The Lancet
One way of ‘directly’ dealing with this problem is to grab the toxic form of alpha synuclein as it is being passed between cells, and remove it from the body. This idea has given rise to a series of ongoing clinical trials that are using a process called immunotherapy – which involves boosting the body’s immune system to target toxic forms of the Parkinson’s associated protein, alpha synuclein.

Monoclonal antibodies. Source: Astrazeneca
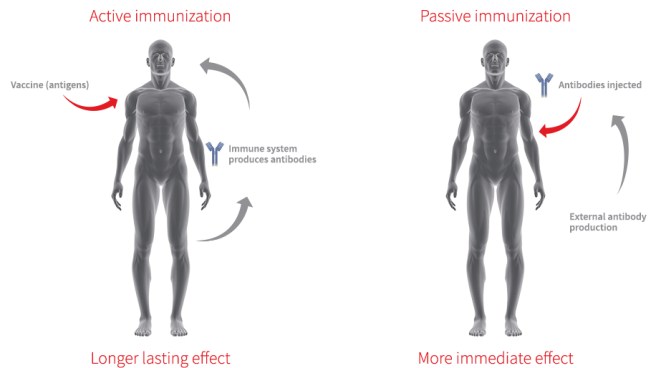
This immunotherapy approach uses antibodies, which are Y-shaped proteins that act like alert flags for the immune system. Antibodies target very specific structures, while ignoring everything else. Immunotherapy can be conducted in two way
- The body’s immune system can be encouraged to target the toxic form of alpha synuclein (using active immunisation in the form of a vaccine); or
- Researchers can design antibodies themselves that specifically target the toxic form of alpha synuclein (while leaving the normal version of the protein alone), and then inject those antibodies into the body (passive immunisation)
Immunotherapy. Source: Acimmune
Both of these approaches are being tested in two clinical trial programmes are being run by two biotech companies. The vaccine approach is being tested by an Austrian biotech firm called AFFiRiS.
In June 2017, AFFiRiS announced that the results of Phase I clinical study of their product AFFITOPE® PD03A. The company reported that the vaccine is causing an immune response (the immune system is generating antibodies against the toxic form of alpha synuclein) and that the vaccine was safe in people with Parkinson’s (Click here to read the press release, and click here to read more about the trial).
AFFiRiS has two vaccines for Parkinson’s that have been studied in phase I studies, and thus far 98 people with Parkinson’s or multiple system atrophy (MSA, a condition very similar to Parkinson’s – Click here to read a SoPD post on this topic) have participated in studies investigating either AFFITOPE® PD01A or PD03A. During these studies, participants were observed for up to 48 months (AFFITOPE® PD01A) or 12 months (AFFITOPE® PD03A), respectively. These observations have focused on long-term safety, immunological and clinical parameters, and the vaccine appears to be relatively safe.
Next, the company will be seeking to determine if they actually work. AFFiRiS is currently planning a Phase II efficacy trial, and we expect to learn more about that trial in 2018. (It would also be nice to hear about the follow up analysis of the 90+ individuals who have received the vaccine – have they demonstrated any benefits? – but this may be expecting too much).
Vaccination. Source: WebMD
Meanwhile, the passive immunisation (or ‘design your own antibodies’) approach to immunotherapy is being tested by an American biotech company called Prothena.
In 2016, Prothena reported the Phase I safety clinical trial results of their primary drug PRX002 (also known as RG7935; Click here to read the research report on this study), and they have rapidly moved on to Phase II testing of the drug, which is called the PASADENA study (listed on the Clinicaltrials.gov as NCT03100149).
That study involves two parts. Part 1 is a randomised, double-blind, placebo-controlled, three-arm study which will enrol approximately 300 patients (less than 2 years since diagnosis) to evaluate the efficacy and safety of PRX002 in people with Parkinson’s over 52 weeks. Participants will be randomly assigned to one of three groups (1500 mg or 4500 mg of PRX002, or placebo treatment). The treatments will be administered via intravenous infusion once every 4 weeks. One complicating aspect of this first part of the study is that eligible participants must not be on any dopaminergic therapy, and they must not be expected to require dopaminergic therapy for the 52 weeks of Part 1 of the study – identifying subjects that fit this criteria may slow recruitment.
Part 2 of this Phase II clinical trial is a 52-week blinded extension phase in which participants from the placebo group of the study will be re-randomly assigned into one of two active doses on a 1:1 basis. This means that all participants will be on active treatment. Participants who were originally assigned to an active dose will continue at that same dose level for the additional 52 weeks. In part 2 of the study, participants will be allowed to use dopaminergic therapies (if required).
We are not expecting the results of this trial until 2020/21.
AFFiRiS and Prothena are not the only companies investigating the immunotherapy approach for Parkinson’s. There are other companies with early-stage programs in this area, including Biogen (BIIB054), AC Immune (ACI-870), Proclara (NPT088), NeuroPore (NPT200-11; in collaboration with UCB) and BioArctic Neuroscience (BAN0805; in collaboration with AbbVie).
And in addition to this direct approach to slowing the condition, there is also the indirect approach to tackling the progression of Parkinson’s:
The indirect approach
In keeping with the war analogy I used above, if the direct approach is a full frontal assault on the enemy front lines, you can consider the indirect approach to slowing/halting Parkinson’s as employing guerilla warfare tactics, attacking the rear supply lines if you like.
Source: Flickr
Researchers are indirectly trying to slow the spread of Parkinson’s using multiple techniques, one of which involves enhancing cells ability to recycle or dispose of waste.
The first of these clinical trials involves a clinically available drug called Ambroxol:

Ambroxol. Source: Skinflint
Ambroxol is a commonly used treatment for respiratory diseases (the respiratory system being the lungs and related components required for breathing). Ambroxol promotes the clearance of mucus and eases coughing. It also has anti-inflammatory properties, reducing redness in a sore throat. It is the active ingredient of products like Mucosolvan, Mucobrox, and Mucol.
Researchers believe that Ambroxol could help with Parkinson’s in two ways:
1. Ambroxol is believed to triggers exocytosis of lysosomes (Source). Exocytosis is the process by which waste is exported out of the cell. Lysosomes are the bags of digestive that rubbish and waste is put into inside a cell for recycling. by encouraging lysosomes to undergo exocytosis and spit their contents out of the cell – digested or not – Ambroxol allows the cell to remove waste effectively and therefore function in a more normal fashion.

Exocytosis. Source: Socratic
2. Ambroxol has been shown to increase levels of an enzyme called glucocerebrosidase (or GCase) in the brain (Source). GCase is involved with the breaking down of proteins like alpha synuclein in the lysosome. GCase is generated from instructions provided by a gene called GBA1. Mutations in this gene are one of the most common genetic risk factors for Parkinson’s.
So by administering Ambroxol to people with Parkinson’s, researchers are hoping to raise levels of GCase to help digest proteins and increase the excretion of this waste from cells. This would ideally keep cells healthier for longer and slow down the progression of Parkinson’s.
There are two clinical trials testing Ambroxol in Parkinson’s in two different “Londons” (London, UK and London, Canada).
London, Canada. Source: Pinterest
The first study (named AiM-PD) is a phase IIA Prospective, Single-Centre, Open Label Clinical Trial to evaluate the safety, tolerability and pharmacodynamic effects of Ambroxol in Parkinson’s (Click here to read more about this trial). This trial, which is funded by the Cure Parkinson’s Trust and the Van Andel Research Institute(USA), is being conducted at the Royal Free Hospital in London (UK). It is testing the safety of slowly increasing doses of Ambroxol in Parkinson’s.
The researchers are also looking to see if Ambroxol can increase levels of glucocerebrosidase (GCase) activity and whether this has any beneficial effects in the subjects. The study has been conducted over a 6 month period, on 20 people with Parkinson’s (critically 10 people with the GBA variant and 10 people with out a GBA mutation – Click here to read more about it).
We are expecting results from this study in 2018.
The second study is being conducted by the Lawson Health Research Institute (and the Weston Foundation) in Canada, and it is a phase II, 52 week trial of Ambroxol in 75 people with Parkinson’s Disease Dementia (Click here to read more about this trial). In this randomised, double blind study, two doses of Ambroxol will be tested – a high dose (1050 mg) and a low dose (525 mg) – as well as a placebo treated group.
While this second trial will finish in the second half of 2018, we are not expecting results from this study until 2019.
Another drug that may provide an indirect means of slowing Parkinson’s is Nilotinib.
Nilotinib. Source: William-Jon
Nilotinib is a small-molecule tyrosine kinase inhibitor, that has been approved for the treatment of imatinib-resistant chronic myelogenous leukemia (CML).
In certain types of leukemia, a specific genetic abnormality occurs in chromosome 22. This is called the Philadelphia chromosome and it results from part of chromosome 9 and 22 actually swapping places.

Philadelphia chromosome. Source: Wikipedia
The result of this mix up is that a fusion gene on chromosome 22, which is created by shifting of the ABL1 gene from chromosome 9 (region q34) to a part of the BCR gene on chromosome 22 (region q11). This leads to a weird fusion protein called bcr-abl, and this protein undertakes various functions, but it is always turned on. And this constant activity helps to drive cancer cell growth in blood cells.
Initially a cancer wonder drug called Imatinib (also known as Gleevec – maybe you’ve heard of it) was developed to block the bcr-abl fusion protein from binding to other proteins that help to encourage the cancer growth. It competitively binds to a small region of the bcr-abl fusion protein, not allowing (or blocking) it to fulfill its function.

How Imatinib (aka Gleevec) works in cancer. Source: Wikipedia
Basically, Nilotinib is very similar to Gleevec – in shape and function. Think of Nilotinib as the new and improved version of Gleevec.
abl protein binds to and interacts with the Parkinson’s associated protein Parkin. But this interaction is not good: abl basically stops Parkin from having doing its job – that is, grabbing old proteins and other rubbish for disposal or recycling in a process called autophagy. Autophagy is a process that clears waste and old proteins from inside cells, preventing them from accumulating and possibly causing the death of the cell.

The process of autophagy. Source: Wormbook
When Parkin is inhibited by abl, normal autophagy does not take place and old proteins and faulty mitochondria start to pile up, making the cell sick (ultimately leading to the death of the cell). By administering Nilotinib to people with Parkinson’s, researchers are hoping the drug will block the abl protein from inhibiting Parkin and allow cells to dispose of waste and function better.
There are currently two Phase II double blind clinical trials of Nilotinib being conducted:
- PD Nilotinib, which is being conducted at Georgetown University in Washington DC (Click here for the more details about this study). This study involves 75 participants and will involve two parts:In the first part of the study, one third of the participants receiving a low dose (150mg) of Nilotinib, another third receiving a higher dose (300mg) of Nilotinib and the final third will receive a placebo drug (a drug that has no bioactive effect to act as a control against the other two groups). The outcomes will be assessed clinically at six and 12 months by investigators who are blind to the treatment of each subject. These results will be compared to clinical assessments made at the start of the trial.In the second part of the study, there will be a one-year open-label extension trial, in which all participants will be randomised given either the low dose (150mg) or high dose (300mg) of Nilotinib. This extension is planned to start upon the completion of the first part (the placebo-controlled trial) to evaluate Nilotinib’s long-term effects.

- NILO-PD, which is being conducted at the Feinburg School of Medicine at Northwestern University (Click here for the more details about this study). This study will involve two groups (or cohorts) of participants.The treatment will firstly be given to a first cohort of 75 people everyday for six months, during which the participants will be closely monitored. Following the treatment phase there will be an 8 week follow up period (during which all treatments are stopped), in order to determine Nilotinib’s safety and tolerability. Trial participants will be tested on their motor and cognitive abilities, and they will give sampled (i.e., blood and spinal fluid) for biological testing. All of these tests will provide the investigators with data with which to evaluate the potential impact of Nilotinib on Parkinson’s symptoms and the disease progression.If the drug is shown to be safe in this first cohort, a second trial is planned to be immediate initiated which would test Nilotinib in a second cohort of 60 people with early Parkinson’s disease. These subjects would be given the highest tolerated daily dose of Niltotinib (based on the results of the first study) or a placebo for 12 months. The same measurements (eg. biological samples) would be applied to this study as well.
Click here to read an SoPD post about Nilotinib and these clinical trials. Both of these studies are expected to provide results in 2020.
2. A neuroprotective agent
Source: Multiplesclerosisnewstoday
Once a drug or a treatment has been determined to slow down the progression of Parkinson’s, it will be necessary to protect the remaining cells and provide a nurturing environment for the third part of the ‘cure’ (Cell replacement therapy). This is where the second component of any curative approach for Parkinson’s comes into effect.
Neuroprotection is the area of research that has had the most attention. Drug companies employ a lot of resources in this area in the hope that a treatment which works for one condition (such as Alzheimer’s) will work for another (like Parkinson’s).
In 2018 we should get a lot of information regarding this area of research.
Of particular interest are the results of the Phase II clinical trial of GDNF from Bristol.
In July 2017, we learned that this trial had failed to meet the primary efficacy end point (Click here to read the SoPD post about that announcement). The initial results suggested that the drug had not worked, in 2018 we can expect to see the final report of the study.

GDNF was pumped into the striatum (green area). Source: Bankiewicz lab
Glial cell-derived neurotrophic factor (or GDNF) is a neurotrophic factor (neurotrophic = Greek: neuron – nerve; trophikós – pertaining to food/to feed). It is a chemical that is produced in the brain. GDNF has previously been found to have miraculous effects on some of the neurons in the brain that are most affected by Parkinson’s disease (particularly the dopamine neurons – Click here to read a SoPD post about this).
The structure of GDNF. Source: Wikipedia
Given the amazing results in laboratories around the world, clinical trials were set up for people with Parkinson’s disease. The first study had astounding results, but a larger follow up study failed to replicate those results and so a third GDNF clinical trial was initiated: the Bristol GDNF study
Bristol. Source: Megabus
The Bristol GDNF study was run by the the North Bristol NHS Trust and funded by Parkinson’s UK with support from The Cure Parkinson’s Trust. The company MedGenesis Therapeutix supplied the GDNF and additional project resources/funding. MedGenesis Therapeutix itself has funding support from the Michael J. Fox Foundation for Parkinson’s Research.
The study involved participants having GDNF (or a placebo drug) pumped directly into their brains, into an area called the putamen. The putamen is where the greatest loss of dopamine occurs in people with Parkinson’s disease. All together there were 41 people with Parkinson’s disease enrolled in the clinical trial. The trial was divided into two phases: during the first phase 35 participants received either GDNF or a placebo drug over 9-months in a double blind fashion. In the second phase of the study, everyone received GDNF.
While it was disappointing to hear that the trial had failed, it will still be interesting to see the results of the study.
Those GDNF results are important for another Parkinson’s clinical trial that is being conducted in Finland. That study is being conducted by a biotech firm called Herantis Pharma, and they are pumping another neurotrophic factor into the brain called Cerebral Dopamine Neurotrophic Factor (CDNF). Herantis are using the same equipment as the GDNF trial to pump CDNF into the brains of people with Parkinson’s (Click here to read the press release about the initiation of the study last year, and click here to learn more about the structure of the study).
We will find out the results of the CDNF clinical trial in 2019/20.
In 2018, we are also expecting to hear about the next step in the testing of the diabetes drug, Buydureon (also known as Exenatide; a GLP-1 receptor agonist). In August of last year, the results of the Phase II clinical trial of Buydureon in Parkinson’s disease were published on the medical journal The Lancet. The trial was randomised and double blind, and the results suggested that the Buydureon delayed/improved the motor features of the treatment group (compared to the control group) over the 60 week trial.
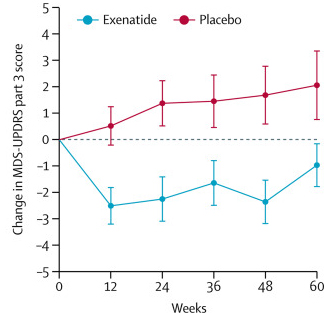
Reduction in motor scores in Exenatide group (blue line). Source: Lancet
This year we can expect to hear news about the plans for further testing of Buydureon (a Phase III trial?).
In addition, there are other GLP-1 agonists (similar to Bydureon) being tested in Parkinson’s. The Cure Parkinson’s Trust has initiated a Phase II safety and efficacy trial of Liraglutide in people with Parkinson’s disease who are also insulin-resistant at the Cedars-Sinai Medical Center in California (Click here for more information). This study will involve 52 weeks of treatment, followed by a further 2 months of no treatment. The results of that study will be available in 2019/20.
And the Cure Parkinson’s Trust has also initiating a 21 centres clinical study across France that will investigate another GLP-1 agonist called Lixisenatide in people with early stage Parkinson’s disease. The results of that study should be available in 2020/21.
Another potentially neuroprotective drug that we will be announcing results in 2018 is Ursodiol (Ursodeoxycholic Acid or UDCA). This is a drug that is used to to improve bile flow and reduce gallstone formation.

Ursodiol. Source: Wikimedia
Bile is a fluid that is made and released by your liver, and it stored in the gallbladder. Its function is to help us with digestion. UDCA occurs naturally in bile – it is basically a bile acid and can therefore be useful in dissolving gallstones. UDCA has been licensed for the treatment of patients since 1980. UDCA also reduces cholesterol absorption.
More recently, UDCA has also been found to improve mitochondrial function in models of Parkinson’s (Click here to read a SoPD post on this topic). Mitochondria are the batteries within a cell. The power suppliers. Without them, the cell would quickly fail and die. There is a lot of evidence now suggesting that faults within normal mitochondria processing may underlie aspects of Parkinson’s. Researchers have concluded that if UDCA can help to improve mitochondrial function, then it should be clinically tested in Parkinson’s.

Mitochondria and their location in the cell. Source: NCBI
The University of Minnesota – Clinical and Translational Science Institute has been conducting a Phase I open label clinical trial of UDCA in people with Parkinson’s (Click here to read more about this). This trial has not, however, be testing efficacy of the drug on Parkinson’s symptoms.

Rather, this trial has simply focus on measuring UDCA levels in individuals after four weeks of repeated high doses of oral UDCA (50mg/kg/day), and determining the bioenergetic profile and ATPase activity in those participants. Basically, they want to see if UDCA is safe and active in people with Parkinson’s disease.
We will be expecting the results of this study in 2018 (and hopefully if those results are good, we should see the initiation of a Phase II trial).
South Korean biotech firm Kainos Medicine will be reporting the results of another Phase I clinical trial in 2018. Kainos’s new experimental treatment, called KM-819, is trying to help in a different way: it is trying to slow down the cell death that is associated with Parkinson’s.
KM-819 is an inhibitor of Fas Associated Factor 1 (or FAF1).
Fas Associated Factor 1 is a protein that interacts with and enhances the activity of a protein on the surface of cells with the ominous name: Fas Cell Surface Death Receptor…and yes, the use of the word ‘death’ in that name should give you some indication as to the function of this protein. When Fas Cell Surface Death Receptor gets activated on any given cell, things have definitely taken a turn for the worse for that particular cell. Thus, by blocking/inhibiting FAF1, Kainos’s is hoping to provide neuroprotection in Parkinson’s, and they certainly have preclinical data that suggests it works (Click here to read a SoPD post on this topic).
The company has been conducting a phase I dose-escalation clinical trial in South Korea, which has evaluated the safety, tolerability, and biochemical properties of KM-819 in 48 healthy adults (click here to read more about the trial). The study was split in two parts: Part A is a single dose of KM-819 or a placebo given in ascending doses to participants. While Part B was the same except that multiple ascending doses of the compound were given to the participants.
The trial conducted last year was only six weeks long, and we are expecting to see the results of that study in 2018 (and again, hopefully if those results are good, we should see the initiation of a Phase II trial).
One additional novel drug that I will be looking forward to hearing results for in 2018 is EPI-589.
Please don’t ask me what EPI-589 is (or what it does), because I do not know.
Edison Pharmaceuticals (no website) is a biotech firm that is working on mitochondrial functioning and they have conducted a Phase II open label, safety trial for the evaluation of EPI-589 in people with early onset genetic forms of Parkinson’s and also idiopathic Parkinson’s (Click here to learn more about this trial). While I like the focused approach, this company is very secretive about their research.
We are expecting to see the results of that study in 2018.
3. Some form of cell replacement therapy
Source: Autohouston
Once the condition has been slowed/halted (#1) and a neuroprotective/nurturing environment is in place to protect the remaining cells (#3), a curative treatment for Parkinson’s will require replacing some of the cells that have been lost.
By the time a person is presenting the motor features characteristic of Parkinson’s, and being referred to a neurologist for diagnosis, they have already lost approximately 50% of the dopamine producing neurons in an area of the brain called the midbrain. These cells are critical for normal motor function – without them, movement becomes very inhibited.
And until we have developed methods that can pick up Parkinson’s long before the motor features appear (which would require only a disease halting treatment), some form of cell replacement therapy is required to introduce new cells to take up lost function.
Cell transplantation represents the most straight forward method of cell replacement therapy.
Traditionally, the cell transplantation procedure for Parkinson’s has involved multiple injections of developing dopamine neurons being made into an area of the brain called the putamen (which is where much of the dopamine naturally produced in the brain is actually released). These multiple sites allow for the transplanted cells to produce dopamine in the entire extent of the putamen. And ideally, the cells should remain localised to the putamen, so that they are not producing dopamine in areas of the brain where it is not desired (possibly leading to side effects).

Targeting transplants into the putamen. Source: Intechopen
Postmortem analysis – of the brains of individuals who have previously received transplants of dopamine neurons and then subsequently died from natural causes – has revealed that the transplanted cells can survive the surgical procedure and integrate into the host brain. In the image below, you can see rich brown areas of the putamen in panel A. These brown areas are the dopamine producing cells (stained in brown). A magnified image of individual dopamine producing neurons can be seen in panel B:

Transplanted dopamine neurons. Source: Sciencedirect
The transplanted cells take several years to develop into mature neurons after the transplantation surgery. This means that the actually benefits of the transplantation technique will not be apparent for some time (2-3 years on average). Once mature, however, it has also been demonstrated (using brain imaging techniques) that these transplanted cells can produce dopamine. As you can see in the images below, there is less dopamine being processed (indicated in red) in the putamen of the Parkinsonian brain on the left than the brain on the right (several years after bi-lateral – both sides of the brain – transplants):

Brain imaging of dopamine processing before and after transplantation. Source: NIH
The old fashioned approach to cell transplantation involved dissecting out the region of the developing dopamine neurons from multiple donor embryos, breaking up the tissue into small pieces that could be passed through a tiny syringe, and then injecting those cells into the brain of a person with Parkinson’s.
The people receiving this sort of transplant would require ‘immunosuppression treatment’ for long periods of time after the surgery. This additional treatment involves taking drugs that suppress the immune system’s ability to defend the body from foreign agents. This step is necessary, however, in order to stop the body’s immune system from attacking the transplanted cells (which would not be considered ‘self’ by the immune system), allowing those cells to have time to mature, integrate into the brain and produce dopamine.
There is a European clinical trial currently ongoing called Transeuro that is using this procedure to test this approach.
The Transeuro trial. Source: Transeuro
The Transeuro trial is an open label study, involving 13 subjects, transplanted in different sites across Europe. They will receive immunosuppression for at least 12 months post surgery, and the end point of the study will be 3 years post surgery, with success being based on brain imaging of dopamine release from the transplanted cells (PET scans).
The European consortium behind the Transeuro trial. Source: Transeuro
In addition to testing the efficacy of the cell transplantation approach for Parkinson’s, another goal of the Transeuro trial is to optimise the surgical procedures with the aim of ultimately shifting over to an embryonic stem cells oriented technique (see below for more information about this).
FULL DISCLOSURE – The author of this blog is associated with research groups conducting the current Transeuro transplantation trials. It is also important for all readers of this post to appreciate that cell transplantation for Parkinson’s disease is still experimental. Anyone declaring otherwise (or selling a procedure based on this approach) should probably not be trusted. While I appreciate the desperate desire of the Parkinson’s community to treat the disease ‘by any means possible’, bad or poor outcomes for this technology could have serious consequences for the individuals receiving the procedure and negative ramifications for all future research in the stem cell transplantation area.
A second clinical study of cell transplantation for Parkinson’s is being conducted in Melbourne (Australia), by an American company called International Stem Cell Corporation (ISCO).

This study is taking the new approach to cell transplantation, and the company is using a different type of stem cell to produce dopamine neurons in the Parkinsonian brain.
Specifically, the researchers will be transplanting human parthenogenetic stem cells-derived neural stem cells (hpNSC). These hpNSCs come from an unfertilized egg – that is to say, no sperm cell is involved. The female egg cell is chemically encouraged to start dividing and then it becoming a collection of cells that is called a blastocyst, which ultimately go on to contain embryonic stem cell-like cells.
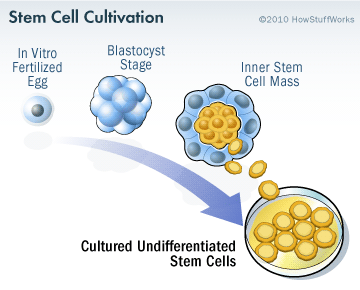
The process of attaining embryonic stem cells. Source: Howstuffworks
This process is called ‘Parthenogenesis’, and it’s not actually as crazy as it sounds as it occurs naturally in some plants and animals (Click here to read more about this). Proponents of the parthenogenic approach suggest that this is a more ethical way of generating embryonic cells as it does not result in the destruction of a viable organism.
Regular readers of this blog will be aware that I am extremely concerned about this particular trial (Click here and here to read previous posts about this). Specifically, there is limited preclinical data from the company supporting the efficacy of these hpNSC cells being used in the clinical study (for example, the company reported that the hpNSC cells that they inject spread well beyond the region of interest in the company’s own published preclinical research – not an appropriate property for any cells being taken to the clinic).
I have also expressed concerns regarding the researchers leading the study making completely inappropriate disclosures about the study while the study is ongoing (Click here to read more about this). This behaviour is unprofessional at best and bordering on unethical. Such comments only serve the interests of the company behind the study. And while I have no issues with a company taking stem cells to the clinic, such behaviour only sets this company up for criticism and ridicule from the rest of the research community.
We will find out the results of the ISCO Phase I clinical trial in early 2019.
In addition to the current Transeuro and ISCO studies, in 2018 three additional research groups – taking three slightly different approaches – will begin the process of trialling cell transplantation therapy in clinical testing.
Presenting them in no particular order: the first is group in this threesome is a company called Biotech BlueRock Therapeutics.
Bayer and healthcare investment firm Versant Ventures joined forces to invest $225 million in stem-cell therapy company BlueRock Therapeutics. This venture will be focused on induced pluripotent stem cell (iPSC)-derived therapeutics for cardiovascular disease and neurodegenerative disorders, particularly Parkinson’s (co-founders Lorenz Studer and Viviane Tabar are world renowned experts in the field of cell transplantation for Parkinson’s).
Prof Lorenz Studer. Source: Tmrwedition
Importantly, BlueRock has acquired rights to a key iPSC intellectual property from iPS Academia Japan, and with 4 years of funding they are now looking to make things happen in 2018. They are submitting an Investigational New Drug (IND) application to the FDA, seeking approval to start clinical trials in 2018 for the transplantation of dopamine neurons derived from embryonic stem cells.
Embryonic stem cells have the potential to become any type of cell in the body – this means that they are ‘pluripotent’. Embryonic stem cells can be encouraged (using specific chemicals) to become whatever kind of cell you want them to be. This feature makes them particularly appealing to research scientists and clinicians who are interested in regenerative medicine (or replacing cells that are lost due to injury or disease).

Embryonic stem cells in a petri dish. Source: Wikipedia
Embryonic stem cells are derived from a fertilised egg cell. The egg cell will divide, to become two cells, then four, eight, sixteen, etc. Gradually, it enters a stage called the ‘blastocyst’, which is a small ball of cells. Inside the blastocyst is a group of cell that are called the ‘inner stem cell mass’, and it is these cells that can be collected and used as embryonic stem cells.

The process of attaining ES cells. Source: Howstuffworks
The tissue used in the old approach for cell transplantation in Parkinson’s disease was dissected from embryonic brains. Obviously there are serious ethical and moral problems with using this kind of tissue. There is also a difficult problem of supply: tissue from at least 3 embryos is required for transplanting each side of the brain (6 embryos in total). Given these issues, researchers have focused their attention on a less controversial and more abundant supply of cells: brain cells derived from embryonic stem cells.
Prof Lorenz Studer and colleagues have many years of experience converting embryonic stem cells into dopamine neurons and transplanting them in models of Parkinson’s (Click here and here to read examples of this research). They are also part of the New York State Stem Cell Science Consortia which is working hard to bring stem cell therapies to the clinic.
In 2018, we can expect to see BlueRock initiate an embryonic stem cell-based clinical trial for Parkinson’s.
Once BlueRock Therapeutics has conducted an embryonic stem cell-based trial for Parkinson’s, they will then look at shifting towards a more personalised format of cell transplantation using induced pluripotent stem cells.
Induced pluripotent stem cells (or IPS cells) are the type of stem cells that the second group of researchers will be using in their approach to clinical trials of cell transplantation in Parkinson’s
Prof Jun Takahashi of the Center for iPS Cell Research and Application (or CiRA) in Kyoto (Japan) is leading a team that is planning to use IPS cells for a clinical cell transplantation trial for Parkinson’s in 2018. In 2017, they published very impressive data using these cells in primate models of Parkinson’s (Click here to read more about that research report), and they are now planning to take this technology to the clinic.
IPS cells are stem cells that can be derived from a mature cell (like a skin cell) collected from any individual. The mature cell is coerced into changing its identity and reverting back to an immature stem cell which is capable of becoming any kind of cell in the body. This process is achieved using specific reprogramming factors (Click here to read a SoPD post on IPS cells).
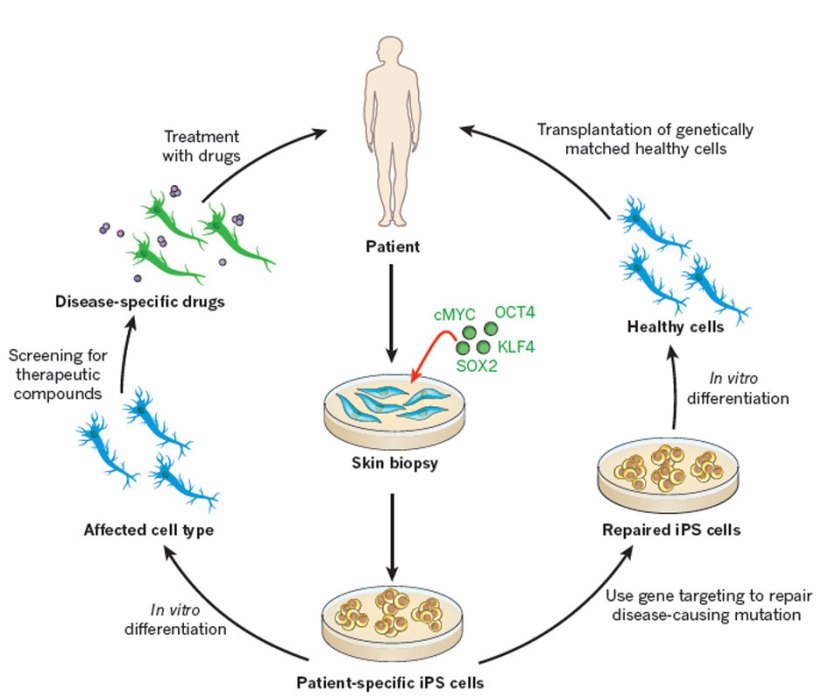
Some of the option available to Parkinson’s disease. Source: Nature
The real beauty of IPS cell technology is that it can be applied on an individual basis. Skin cells can be collected from a particular person, converted into IPS cells and then those cells can be chemically encouraged to become dopamine neurons, which can either be used for cell transplantation in that individual (avoiding the immune system rejecting the cells as being foreign objects (not self), or those dopamine cells can be used to screen drugs for that individual – identifying which drugs will and won’t work in that particular person.
It is the essence of personalised medicine.
And these are the cells that CiRA research group will be using in their clinical trial (and eventually the BlueRock Therapeutics will be using this approach as well). In 2018, we can expect to see the CiRA group initiate an IPS cell-based clinical trial for Parkinson’s.
And finally, the third group initiating a cell transplantation clinical trial in 2018, will be a research group from China led by Professor Qi Zhou, a stem-cell specialist at the Chinese Academy of Sciences Institute of Zoology.
The clinical trial (Titled: A Phase I/II, Open-Label Study to Assess the Safety and Efficacy of Striatum Transplantation of Human Embryonic Stem Cells-derived Neural Precursor Cells in Patients With Parkinson’s Disease) will take place at the First Affiliated Hospital of Zhengzhou University in Henan province.
The researchers are planning to inject neuronal-precursor cells derived from embryonic stem cell into the brains of individuals with Parkinson’s. They have 10 subjects that they have found to be well matched to the cells that they will be injecting, which will help to limit the chance of the cells being rejected by the body.
The difference between the embryonic stem cells used in this study and the ones used in the BlueRock Therapeutics study is that while BlueRock is going to be pushing the cells towards a dopamine cell fate, the Chinese researchers will be using cells that have simply been encouraged to become neurons (a much earlier step in the development of these cells). The reason for this is that the more mature cells (like dopamine neurons) are more likely to die during the physical process of transplantation, while the less mature cells have a better chance of survival.
In 2018, (if they haven’t started already) we can expect to see the Chinese research group initiate an embryonic stem cell-based clinical trial for Parkinson’s.
Additional ‘expectations’
There are several other events that we can expect to occur in the world of Parkinson’s clinical research in 2018 that do not naturally fit into the “cure of Parkinson’s” format of this post. This is mainly because these events will only deal with therapeutic approaches that deal with the symptoms rather than help to cure the disease. These expectations are:
- Oxford Biomedica will initiate a new Phase II clinical trial of their gene therapy approach for Parkinson’s. Their product, OXB-102, is a virus that modifies neurons so that they produce dopamine. The company is currently working on the regulatory approval for a planned three cohort phase I/II study of OXB-102 in Parkinson’s disease which will be conducted (in the UK and France – click here for more on this trial).
- Acorda Therapeutics will finally receive FDA approval for their new inhalable form of L-dopa, called Inbrija (also known as CVT-301). Clinical trials of this product were successful and manufacturing complications have delayed regulators from giving approval.
- Acorda Therapeutics should also release the results of their Phase II clinical trial of SYN-120 in Parkinson’s dementia. SYN-120 is an oral 5-HT6/5-HT2A dual receptor antagonist (Click here to read more about that trial)
- Acorda Therapeutics will also published the results of their terminated Phase III clinical trial of their drug Tozadenant. The clinical development of this drug was cancelled due to a number of cases of sepsis being reported (five of which were fatal). Despite this unfortunate event, the company has a great deal of data from the trial and is now evaluating the results in conjunction with the FDA before deciding what to do next with this drug.
- Sunovion should be releasing results of their drug SEP363856 which has been tested in a Phase II trial of subjects with Parkinson’s associated Psychosis (Click here to read more about that trial).
- Cynapsus Therapeutics (acquired by Sunovion) will hopefully provide results on their Phase III clinical trial of APL-130277 (a fast-acting and easy-to-use thin-strip film coated with the drug apomorphine) for the treatment of OFF episodes in Parkinson’s (Click here to read more about this trial).
- Elto Pharma (a subsidiary of Amarantus Bioscience is preparing for a Phase 2b clinical study of Eltoprazine in the treatment of Parkinson’s disease Levodopa-induced dyskinesia. Eltoprazine is an agonist at the 5-HT1A and 5-HT1B receptors and as an antagonist at the 5-HT2C receptor. It has received Orphan Drug Designation from the US FDA and the company should start enrolling for a clinical trial in 2018 (Click here to read more about this trial).
Additional hopes
- Are we going to see the results of the Io Therapeutics phase I study of their brain penetrant RXR nuclear receptor agonist IRX4204 (Click here to read more about that trial)?
- Are we going to see the results from Hisamitsu Pharmaceutical‘s phase III long-term study of HP-3000 (a transdermal Ropinirole for the treatment of Parkinson’s disease – Click here to read more about this)?
- Will Lysosome therapeutics start phase I trial of their glucocerebrosidase activator called LTI-291?
- Will we hear about results from Serina Therapeutics and their Phase I study of weekly Subcutaneous Injections of SER-214 (Click here to read more about the trial)?
- Could we also hear results from Enterin Inc. on their RASMET study (a Phase I clinical trial of ENT-01 (Kenterin), a derivative of squalamine – Click here to read more about the trial)?
- Will we finally hear results from Genervon Biopharmaceuticals regarding their Phase II trial of Phase 2A Study of GM 608 (an oligopeptide of human motoneuronotrophic factor) in mild to moderate Parkinson’s (the GAP-PD study) (Click here to read more about the trial)?
- Will we ever hear any results from the Phase I clinical trial of Phenylbutyrate in Parkinson’s being conducted at the University of Colorado (Click here to read more about the trial)?
(Special thanks to Kevin McFarthing (on Twitter @InnovationFixer) for his help with pulling together these additional expectations and hopes)
What does it all mean?
In today’s post, we have outlined what we expect from the clinical research that will be conducted in 2018. The list is certainly not exhaustive, but it will hopefully give you a flavour of the many activities that we should hear about over the next 12 months.
We have not speculated on the chances of success or failure of each of the treatments/procedures that was discussed. To do so, would be raising expectations. And as I stated near the top, I am trying to lower my expectations in order to lead a happy existence.
I do think, however that 2018 is going to be a great year for Parkinson’s research.
Am I raising expectations with that statement?
Perhaps.
But only time will tell.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, many of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with any of the companies or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from Weknowyourdreams





























9 thoughts on “The road ahead: Parkinson’s research in 2018”