|
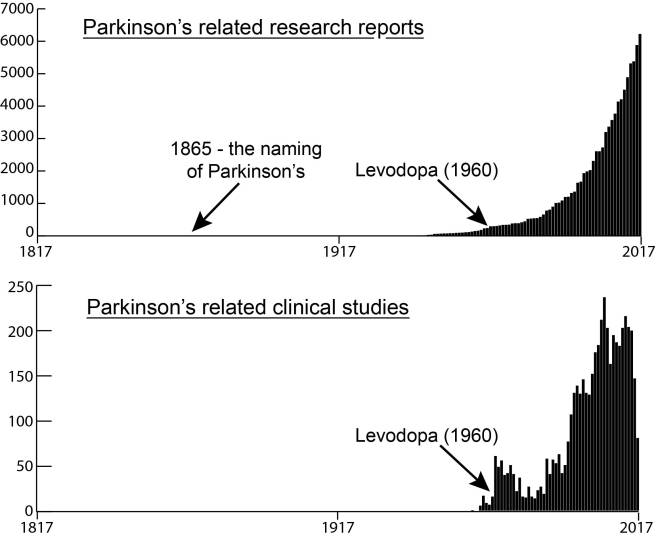
# # # # Alpha synuclein is considered to be an influential factor in Parkinson’s. It is a protein that accumulates and clumps together inside certain nerve cells in many cases of Parkinson’s. Recently, clinical trials have attempted to target alpha synuclein that is floating around outside of cells. Some of the strategies focus on an approach called ‘immunotherapy’, which involves boosting the immune system to help remove the toxic form of this protein from the body. This week, one biotech company – AC Immune – bought the Parkinson’s-associated immunotherapy assets off another biotech company – AFFiRiS – which has been developing a potential vaccine for Parkinson’s. In today’s post, we will discuss what immunotherapy is, look at how AFFiRiS has been trying to apply it to Parkinson’s, and review what AC-Immune plans to do next. # # # # |
AC Immune is a a Switzerland-based biotech company that was foundered in 2003.
They are focused on “improving the lives of patients suffering from Alzheimer’s and other neurodegenerative diseases” (Source).
Their approach has primarily centered around the development of immunotherapy approaches. And this week they made a very interesting announcement.
What is immunotherapy?