An inconvenient truth:
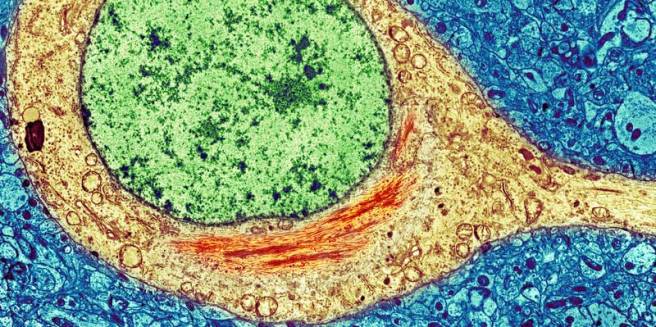
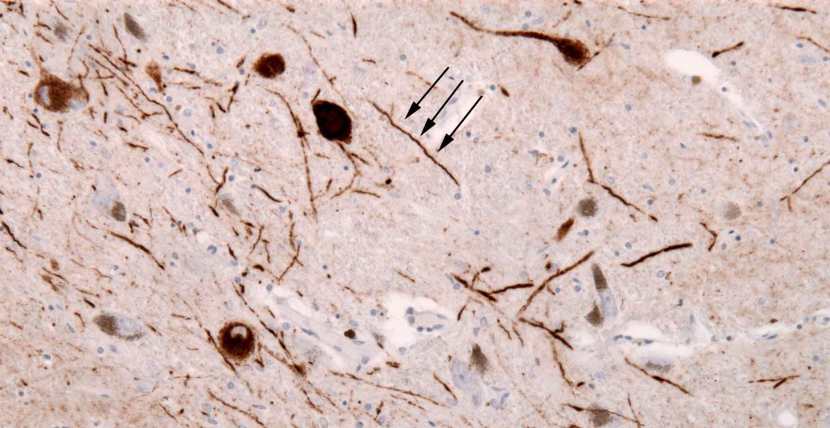
The diagnosis of Parkinson’s disease can only be definitively achieved at the postmortem stage.
There is currently no diagnostic test for this task and we are reliant on the training and skills of the neurologists making the diagnosis. Brain imaging techniques (such as DAT-scans) are great, but they can only aid physicians in their final decision.
And those decisions are not always right.
In 1992, a study looking at the brains of 100 subjects who had died with Parkinson’s disease, found that 24% of the cases did not fulfill the pathological requirements for the diagnosis of Parkinson’s disease. That study was:

Title: Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases.
Authors: Hughes AJ, Daniel SE, Kilford L, Lees AJ.
Journal: Journal of Neurol Neurosurg Psychiatry. 1992 Mar;55(3):181-4.
PMID: 1564476
Unfortunately, despite years of research, it would appear that there is still a large degree of error in the clinical diagnosis of Parkinson’s disease. A study published in 2014 in the journal Neurology that suggested that there is currently a 15% rate of misdiagnosis. That study was:

Title: Low clinical diagnostic accuracy of early vs advanced Parkinson disease: clinicopathologic study.
Authors: Adler CH, Beach TG, Hentz JG, Shill HA, Caviness JN, Driver-Dunckley E, Sabbagh MN, Sue LI, Jacobson SA, Belden CM, Dugger BN.
Journal: Neurology. 2014 Jul 29;83(5):406-12.
PMID: 24975862
It has to be said that clinicians face a very difficult task in diagnosing Parkinson’s disease. The variety of features (symptoms) that patients present with in the clinic, and the lack of diagnostic tools, leave neurologists making a judgement based largely on clinical observations.
But this degree of error ultimately has a huge impact on clinical studies and trials: if 10-20% of the participants are not Parkinsonian, are we really going to observe an accurate result?
Better diagnostic tests/tools are critically required.
In November last year, a study was published in the journal Immunology Letters which may help in this regard:

Title: Potential utility of autoantibodies as blood-based biomarkers for early detection and diagnosis of Parkinson’s disease.
Authors: DeMarshall CA, Han M, Nagele EP, Sarkar A, Acharya NK, Godsey G, Goldwaser EL, Kosciuk M, Thayasivam U, Belinka B, Nagele RG; Parkinson’s Study Group Investigators.
Journal: Immunol Letters, 168(1), 80-8.
PMID: 26386375 (this article is OPEN access if you would like to read it)
The researchers took 398 subjects, including 103 early-stage Parkinson’s disease subjects and they collected blood samples from them. They then screened the blood for 9,486 different autoantibodies that could be useful as biomarkers for Parkinson’s disease.
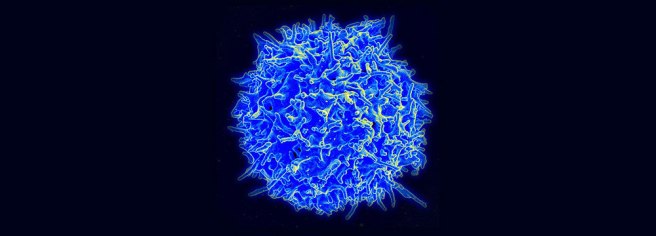
Antibodies are produced by our immune system to determine what is ‘self’ and not ‘self’. They are the foundation of our defenses against the big, bad germ/bacteria world. Autoantibodies are antibodies produced by our immune system that are directed against our own tissues. They target ‘self’.
And yeah, that is bad. Autoantibodies are associated with autoimmune diseases such as Lupus.
We are not sure why we produce autoantibodies. The causes of their production vary greatly and are not well understood. In Parkinson’s disease, however, autoantibodies may be produced as a result of the cell death in the brain. Some of the debris resulting from the dying cells will make its way into the bloodstream, to be removed from the body. Whilst in the blood, some of that debris could trigger the immune system, thus resulting in the production of autoantibodies.
De Marshall et al (the researchers who conducted this study) were hoping to take advantage of this autoantibody production and use them as biomarkers to not only differentiate between people with and without Parkinson’s disease, but also to differentiate between different stages of Parkinson’s disease (see the figure below).

Attempting to differentiate between different stages of Parkinson’s disease. Source: Immuno Letters
The researchers found that using the top 50 autoantibodies that they associated with Parkinson’s disease, they could successfully differentiate between people with and without Parkinson’s disease with 90% prediction accuracy in a blind analysis (they actually found that just the top 4 autoantibodies were enough).
Interestingly, the researchers then compared the early Parkinson’s group with a mild-moderate Parkinson’s group and they found that they could differentiate between the two groups with an overall accuracy of 97.5%!
These are very exciting results and we will be following this work with interest – not only from the standpoint of biomarkers, but also the role of autoantibodies in Parkinson’s disease.
 Winter. Source: Sky
Winter. Source: Sky Winter (Center, looking left) and friends. Source: Uchicago
Winter (Center, looking left) and friends. Source: Uchicago