|
Novel methods for treating neurodegenerative conditions are being proposed on a weekly (sometimes daily) basis. Recently researchers from the University of Cambridge have presented an intriguing new method of removing proteins from inside of cells which involves small proteins called antibodies. Antibodies are an important part of the immune systems response to infection. But their function usually only applies to objects floating around outside of cells. In today’s post, we will look at what antibodies are, explain how this new system works, and discuss some of the issues we face with taking this new technique forward. |
A brain cell from a person with Alzheimer’s. The red tangles in the yellow cell body are toxic misfolded “TAU” proteins next to the cell’s green nucleus. Source: NPR
Here at the SoPD, we often talk about the clustering (or aggregation) of proteins.
Densely packed aggregates of a protein are a common feature of many neurodegenerative conditions, including Parkinson’s.
In fact, the aggregation of a protein called alpha synuclein are one of the cardinal features of the Parkinsonian brain.
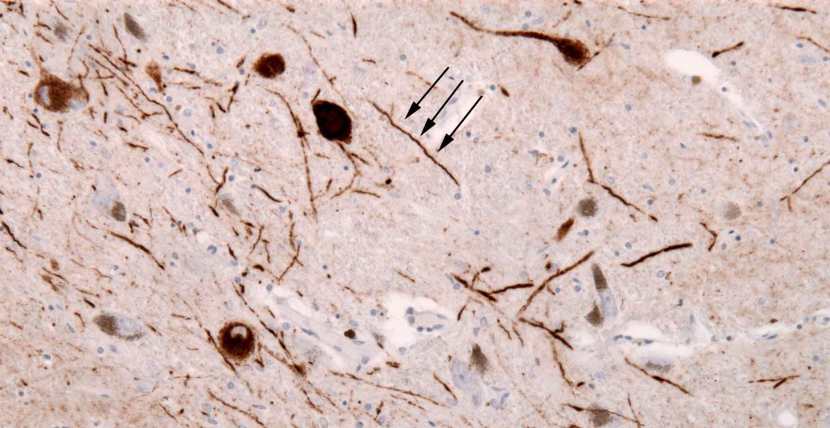
Aggregated alpha synuclein protein in the Parkinsonian brain (stained in brown). Source: Wikimedia
Researchers have long been devising new ways of trying to reduce the amount of alpha synuclein collecting in the brain cells of people with Parkinson’s.
In most cases, their efforts have focused on utilising the cell’s own waste disposal systems.
How do cells dispose of waste?
There are two major pathways by which the cells in your body degrade and remove rubbish:
- The ubiquitin–proteasome system (UPS)
- A process called autophagy
The Ubiquitin Proteasome Pathway is the route via which the majority of proteins are usually degraded.
Source: 2bscientific
A protein labelled with a lot of ubiquitin will be identified and sent off for disposal and be broken down in a structure called the proteasome. For a more thorough explanation of the Ubiquitin Proteasome pathway, please watch this video kindly provided by the Scottish Enterprise:
A second process by which cells dispose of rubbish is called autophagy, and this method is used for removing larger proteins/objects from cells.
Autophagy (from the Ancient Greek αὐτόφαγος autóphagos, meaning “self-devouring”) is an absolutely essential function in a cell. Without autophagy, old proteins would pile up making the cell sick and eventually causing it to die. Through the process of autophagy, the cell can break down the old protein, clearing the way for fresh new proteins to do their job.

The process of autophagy. Source: Wormbook
Waste material inside a cell is collected in membranes that form sacs (called vesicles). These vesicles then bind to another sac (called a lysosome) which contains enzymes that will breakdown and degrade the waste material – the same way enzymes in your washing powder break down muck on your dirty clothes). The degraded waste material can then be recycled or disposed of by spitting it out of the cell.
Interesting. Can you give an example of how researchers are using these waste disposal to target aggregated proteins?
Absolutely.
Late last year (2017), researchers at the University of Cambridge published this report:

Title: A Method for the Acute and Rapid Degradation of Endogenous Proteins
Authors: Clift D, McEvan WA, Labzin LI, Mogessie B, James LC
Journal: Cell, 2017, 171(7):1692-1706.
PMID: 29153837 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers described a method by which aggregated protein could be rapidly degraded.
It involved antibodies.
What are antibodies?
Antibodies are Y-shaped proteins that the immune system naturally and continuously produces to identify anything in the body that is ‘not self’ (that is, not a normally occurring part of you – think of viruses, bacteria, etc).

Monoclonal antibodies. Source: Astrazeneca
Antibodies act like alert flags for the immune system.
When antibodies bind to something, they alert the immune system to investigate and potentially remove. Each antibody targets a very specific structure (called an antigen), while ignoring everything else. An antigen is defined as any substance or molecule that is capable of causing an immune response in an organism.
When a pathogen (an agent that causes disease or damage) is detected in your body, it will quickly be determined to be not ‘self’. This judgement will be made by the identification of antigens on the surface of the pathogen. If an molecule on the surface of the pathogen is not familiar to the immune system, it will be considered an antigen and an immune response will be initiated to have the pathogen removed.
In this fashion, antibodies are a very powerful method of removing items from the body that are causing trouble or not wanted.

Antibodies binding to a virus. Source: Biology-questions-and-answers
We have previously discussed how biotech companies are trying to use antibodies to remove the toxic form of alpha synuclein from the brains of people with Parkinson’s. And there are ongoing clinical trials that are utilising antibody-based approaches to capture this protein as it is being passed from cell to cell (Click here to learn more about these efforts).
Of particular interest with regards to today’s post is a company called Prothena which has designed antibodies that specifically target the toxic aggregated form of alpha synuclein.
We’ll come back to Prothena later in this post, but please understand that it has always been assumed that naturally occurring antibodies are only used by the body to target stuff floating around in your blood or other fluids in your body.
It was believed that antibodies did not target and help remove proteins floating around inside your cells.
(Note the past tense in that previous sentence)
But all of this changed a couple of years ago.
What happened?
A few years ago, the Cambridge researchers behind the new report discovered something amazing:
Title: Antibodies mediate intracellular immunity through tripartite motif-containing 21 (TRIM21).
Authors: Mallery DL, McEwan WA, Bidgood SR, Towers GJ, Johnson CM, James LC.
Journal: Proc Natl Acad Sci U S A. 2010 Nov 16;107(46):19985-90.
PMID: 21045130 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported the discovery of an interesting new function for an intracellular protein (that is, a protein floating around inside of cells) called tripartite motif-containing 21 (or TRIM21).
And what made TRIM21 really interesting was that it binds to antibodies.
TRIM21 bound to an antibody. Source: Cell
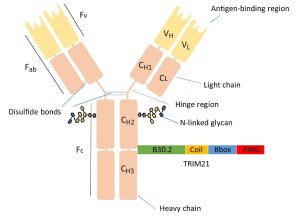
The researchers found that TRIM21 bound itself to the constant region of the ‘heavy chain’ of antibodies.
What is the ‘constant region of the heavy chain’?
Your typical antibody is composed of two immunoglobulin heavy chains (blue in the image below) and two immunoglobulin light chains (pink in the image below):
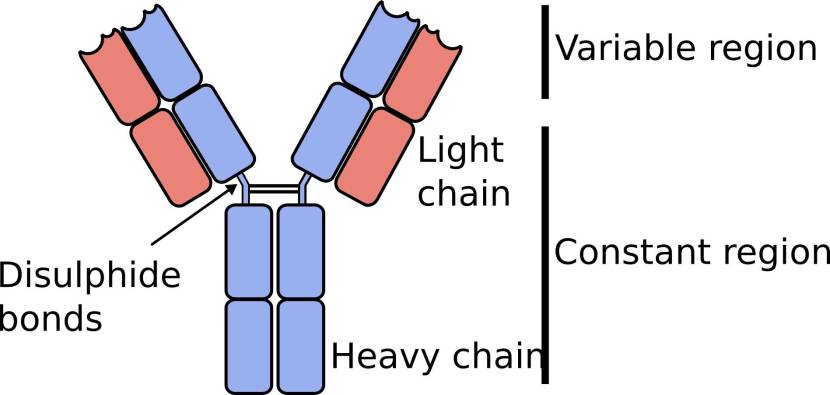
The light chains are in pink. Source: Bitesizebio
Most of the structure of an antibody is constant (always the same), but the top of the Y arms are always variable (or different) from antibody to antibody. These variable parts are the antigen identifying and binding regions, while the bottom stalk of the Y is the part the rest of the immune system interacts with.
This ‘bottom stalk of the Y’ is where TRIM21 also binds to.
And this is where things get really interesting.
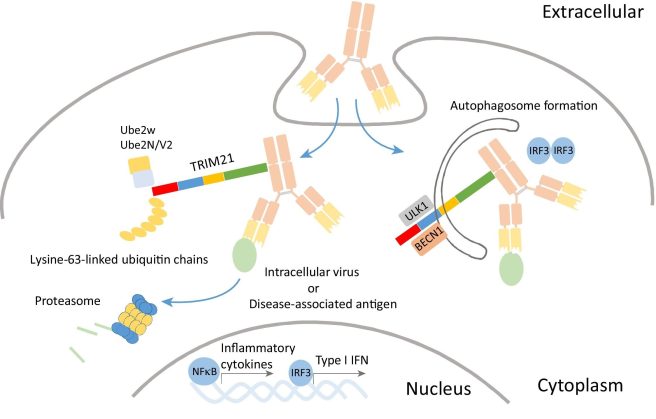
The Cambridge researchers found that not only did TRIM21 bind to antibodies, but it also helped ship them off to the proteasome for degradation.
And if the antibody in question was attached to something bad (like a virus), that would get moved to the proteasome as well.
TRIM21 binding to an antibody inside of a cell. Source: Cell
And yes, for the eagle eyed readers who noticed it in the image above: TRIM proteins have also been associated with the initiation of autophagy (the other waste disposal pathway – Click here to read more about this).
In all of the experiments the investigators conducted, the binding and removal of antibodies by TRIM21 was directly associated with the levels of TRIM21 protein. And they found that cells which produce the highest levels of TRIM21 were more resistant to viral infections than those with the least amount of TRIM21.
This interesting activity of TRIM21 appears to be conserved across mammals, and may simply serve as a system of cleaning up antibodies that somehow find their way into a cell.
OR it could represent an intracellular arm of the adaptive immune system. By this I mean that the protection provided by antibodies does not stop at the cell membrane, but rather it continues inside the cell – offering a very useful defense against viral or bacterial infections.
And the researchers replicated and expanded on these findings in a follow up study (Click here to read the report of that study).
That’s really interesting. What did the researchers do next?
Well, this initial (amazing) discovery got the researchers thinking:
If TRIM21 can be used to dispose of invading viruses, etc, could it also be used to remove other ‘nasties’? Such as,…oooh, I don’t know,…maybe an aggregated protein that is associated with a neurodgenerative condition???
Unfortunately for the Cambridge researchers, other investigators around the world were asking the same question as them, in 2015 a research group in Boston (USA) published this report:
Title: Antibody against early driver of neurodegeneration cis P-tau blocks brain injury and tauopathy.
Authors: Kondo A, Shahpasand K, Mannix R, Qiu J, Moncaster J, Chen CH, Yao Y, Lin YM, Driver JA, Sun Y, Wei S, Luo ML, Albayram O, Huang P, Rotenberg A, Ryo A, Goldstein LE, Pascual-Leone A, McKee AC, Meehan W, Zhou XZ, Lu KP.
Journal: Nature. 2015 Jul 23;523(7561):431-436.
PMID: 26176913 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers chose a protein called TAU as the first aggregating protein to try and target for removal using TRIM21.
What is TAU?
TAU is protein that is very abundant in the brain. It is generated from the RNA (the instructions for making protein) of the MAPT gene, located on chromosome 17.
TAU protein is a microtubule-associated protein, which functions by stabilising the highways and byways inside a cell. Basically, these proteins hold together the roads which allows vesicles (the small bags of stuff) to be moved around the cell (by transporter proteins like kinesin).

TAU: a stabilising presence. Source: Hindawi
So why is TAU interesting to researchers?
Because TAU is associated with both Parkinson’s AND Alzheimer’s.
In both conditions there is a build up of TAU protein, which aggregates in a similar fashion to our old friend the Parkinson’s-associated protein alpha synuclein (Click here for a good review article on this topic).
So what happened when the researchers conducted the experiment on aggregated TAU?
As part of a much larger study, the researchers in Boston demonstrated that when they removed TRIM21 from cells, those cells had no defense against TAU aggregation and there was significantly more build up of antibody-bound TAU in those cells (compared to control cells with normal levels of TRIM21).
And in early 2017, the researchers in Cambridge published the results of their study which replicated and expanded on those findings:
Title: Cytosolic Fc receptor TRIM21 inhibits seeded tau aggregation
Authors: McEwan WA, Falcon B, Vaysburd M, Clift D, Oblak AL, Ghetti B, Goedert M, James LC.
Journal: Proc Natl Acad Sci U S A. 2017 Jan 17;114(3):574-579.
PMID: 28049840 (This report is OPEN ACCESS if you would like to read it)
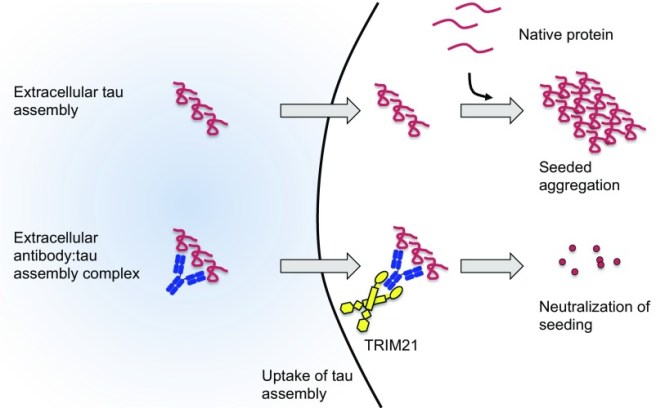
In this study, the Cambridge exposed human cells grown in cell culture to aggregated TAU (with antibodies attached), and they found that TRIM21 could intercept and potently neutralise the antibody-labeled TAU. In the absence of TRIM21, TAU protein (with antibodies attached) would simply aggregate.
Source: NCBI
And again, this process was dependent on levels of TRIM21 protein.
Now, one of the issues with TRIM21 is that the levels of TRIM21 protein in brain tissue are very low (Click here to read more about this). Thus, if this protein is some kind of intracellular defense system against invaders, it is a rather weak weapon.
But this got the researchers in Cambridge thinking again:
What if we had a method of increasing the levels of TRIM21 protein inside of cells? Could we then simply remove any protein that we have a good antibody for?
And late last year (2017), the researchers at the University of Cambridge published this report which answered this question (this is the report I mentioned near the top of this post):

Title: A Method for the Acute and Rapid Degradation of Endogenous Proteins
Authors: Clift D, McEvan WA, Labzin LI, Mogessie B, James LC
Journal: Cell, 2017, 171(7):1692-1706.
PMID: 29153837 (This article is OPEN ACCESS if you would like to read it)
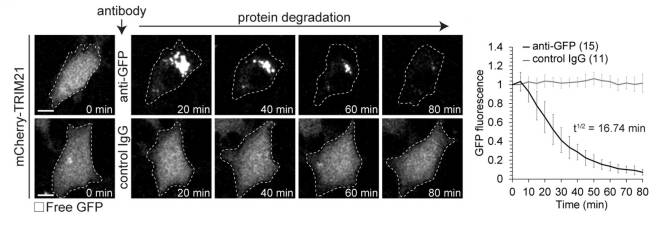
In this study, the researchers produced cells that generate huge amounts of TRIM21. They found that very high levels of TRIM21 did not affect the overall well being of the cells. The investigators then introduced a green fluorescent protein (or GFP) to those cells, which floated around inside them. Next, they exposed the cells to antibodies that targeted GFP and they observed a rapid disposal of GFP over the space of approximately 1 hour.
As you can see in the image below, cells with GFP, but no GFP antibody (control IgG) exhibited no change in their levels of GFP over time (bottom row of images), while the cells exposed to GFP antibody exhibited condensing of the GFP within 20 minutes and then the complete removal of it over the next hour (top row of images).
Source: NCBI
In a long series of experiments, the researchers found that this approach could be used against any protein for which a specific antibody was available, including intracellular proteins involved in neurodegeneration.
And they demonstrated this in a cell-based model of Huntington’s disease.
What is Huntington’s disease?
Huntington’s disease is a neurodegenerative condition which results in individuals losing all inhibition of movement, giving rise to appearance of chorea – jerky, random, and uncontrollable movements (similar to dykinesias).
Huntington’s disease is caused by an increase in a region of DNA inside the Huntingtin gene. This regions is made up of CAG repeats. In normal healthy humans, we usually have up to 30 repeats of CAG. If you have more than 40 CAG repeat, you are definitely going to develop Huntington’s disease.
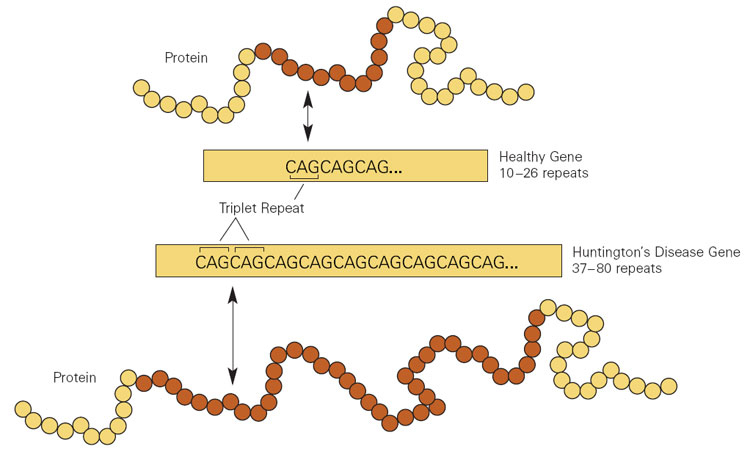
Expansion of CAGs in the Huntingtin gene. Source: NIST
The expansion of the CAGs in the Huntingtin gene results in a mutant Huntingtin protein, which is believed to be associated with the problems observed in people suffering from Huntinton’s disease.
And when the Cambridge researchers created cells that produce the mutant Huntingtin protein and high levels of TRIM21 and then they introduced mutant Huntingtin antibodies, they observed a rapid reduction in the levels of mutant protein. And remarkably the normal version of Huntingtin protein was not affected by this manipulation.
Wow, that’s amazing. When can they start clinical trials?
Yes, it is amazing, but there is a slight problem with the whole system at the moment:
Getting high levels of TRIM21 and antibody into the cells.
While the system works nicely if a virus enters a cell with antibodies attached to it, trying to target troublesome intracellular proteins is more difficult as antibodies are not normally absorbed by cells in high enough levels that allow them to find their protein of interest.
Delivery has always been the problem with medicine-based therapies. Getting the beneficial agent to the right organ (or cells) inside the body, while avoiding all other organs (particularly the liver which chews everything up) is the continual issue for biomedical researchers. And it is an issue with this method.
In the last research report, the researchers used several different methods of getting the TRIM21 protein and antibodies into cells (including electroporation). But until a better method of delivery is determined, the investigators do not foresee clinical applications for this technique, rather it is simply a powerful new research technique.
Bummer. Back to the drawing board?
It would appear to be the case, but this technique could still be of great use to us.
And this is where we come back to the biotech called Prothena which has designed antibodies that specifically target the toxic aggregated form of alpha synuclein.
It could be interesting to see whether the Prothena antibodies work with this TRIM21 system. That is to say, can the toxic alpha synuclein that is aggregating inside cells be targeted by Prothena antibodies and TRIM21.
If so, this might suggest that the Prothena approach is not simply limited to the extracellular alpha synuclein protein. If toxic alpha synuclein being passed between cells is being bound by the Prothena antibodies during that passage and then entering neurons before the immune system can clear it, perhaps low levels of TRIM21 protein are helping to clear the antibody-bound toxic protein inside of the cell.
I am just speculating here, but I think the idea is worth exploring.
So, what does it all mean?
Aggregating proteins are a common feature of many medical conditions, from diabetes to Parkinson’s. Novel methods of reducing/clearing these aggregated proteins are continually being devised. In today’s post, we have reviewed one such method, which has been given the catchy name ‘TRIM away’.
While adapting TRIM away to clinical applications is facing significant hurdles, the research being conducted on this system is teaching us a great deal about the underlying biology of cells (who knew that antibodies worked inside of cells!). And it will hopefully help us as a powerful new research tool, which will aid us in getting us to where we ultimately want to be.
If nothing else techniques such as TRIM away can help us better understand the biology of this condition.
The banner for today’s post was sourced from NCBI






Again, an impressive breakdown, Simon. Love this post. Do you think there might be potential for use of an AAD vector in introducing a gene to build up the levels of TRIM21 in cells, especially SN cells? Is there any information on the permeability of cells to antibodies, monoclonal or natural? Thanks!
LikeLike
Hi Tom,
Glad you liked the post. There is already a great deal of other research surrounding TRIM21 that I could have gone into, but decided against because the post was already getting long enough. Numerous research groups around the world are looking at TRIM21 from all sorts of different angles and medical conditions. Hopefully all of that investigative activity will be beneficial for everyone. One interesting example is this cell-penetrant antibody targeting an intracellular protein that is important for the life cycle of Hepatitis B (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5743566/).
And raising levels of TRIM21 in cells should be theoretically possible using AAV viruses as you suggest – particularly if the efficiency of the AAV-PHP vectors can be improved (https://scienceofparkinsons.com/2017/12/10/monkeys/). Such a scenario would be great because all that would be required then is to take the immunotherapy treatment every few months. A lot of in vivo research would be required before we get to that point and the big question then becomes: Is Parkinson’s just a toxic alpha synuclein condition? (the jury is still out on that one).
Kind regards,
Simon
LikeLike