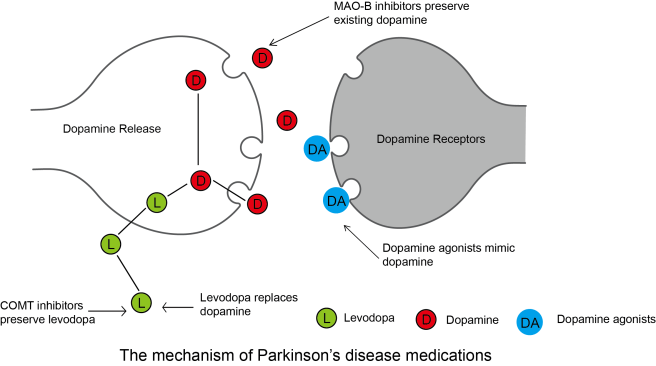
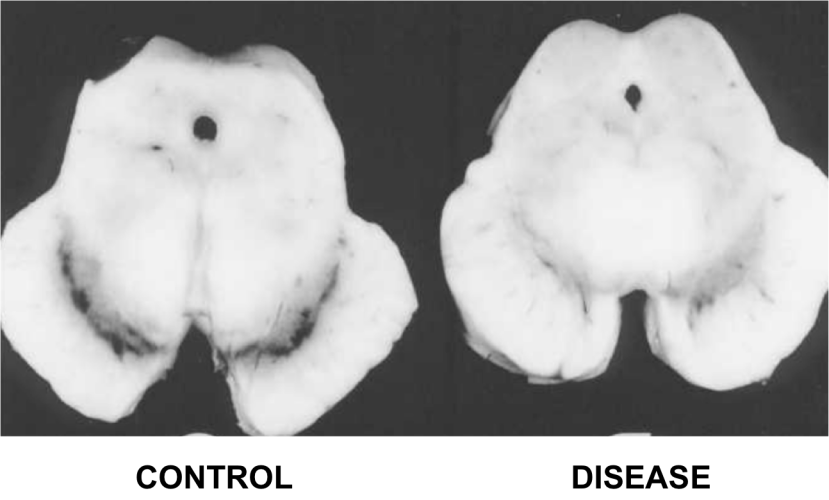
|
Being a patriotic ‘kiwi’, I am always very pleased to write about interesting Parkinson’s research originating from the homeland. And recently the results of an interesting clinical study that was designed and conducted in New Zealand have been published.
The clinical study was focused on whether a diet manipulation could influence motor and non-motor symptoms/features of Parkinson’s.
Specifically, the researchers were looking at the low-fat versus ketogenic diets.
In today’s post, we will discuss what is meant by a ketogenic diet, we will assess the results of the study, and consider what they might mean for the Parkinson’s community.
|

The All Blacks. Source: Newshub
Aotearoa (also known as New Zealand) is a remarkable little country (and yes, I know I’m slightly biased).
It flies under the radar for most folks who are not interested in rugby, amazing scenery, great quality of life, or hobbits, but historically this tiny South Pacific nation of 4.6 million people has punched well above its weight on many important matters.
For example, on the 19th September 1893, the governor, Lord Glasgow, signed a new Electoral Act into law. And as a result of that simple act, New Zealand became the first self-governing country in the world in which all women (over the age of 21) had the right to vote in parliamentary election (Australia followed in 1902, the US in 1920, & the Britain in 1928). That achievement, it should be said, was the result of years of dogged effort by suffrage campaigners, led by Kate Sheppard who is today acknowledged with her portrait on the $10 note:

The NZ $10 note. Source: Whaleoil
A New Zealander made the first trans-global radio transmission on the 18th October 1924. Invalided during WWI, Frank Bell revived his childhood interest in wireless communication, and after being the first kiwi to have two-way radio contact with Australia and North America, he achieved something far more impressive. From the family sheep station in ‘Shag Valley’ (East Otago – bottom of the South Island), he sent a Morse code transmission (“Greetings from New Zealand, signed Bell Z4AA”) which was received and replied to by amateur operator Cecil Goyder at Mill Hill School (London).

Frank and his older sister Brenda. Source: NZhistory
New Zealand was also where jet boats was first invented by Sir Bill (William Hamilton). His first jet boat was a 3.6 meter (12 foot) plywood hull with a 100 E Ford engine, and the jet a centrifugal type pump. This craft was tested on the Irishman Creek dam, before it ran successfully upriver in 1953. And from there it kind of went viral. In 1960, three Hamilton jet boats (the Kiwi, Wee Red and Dock), became the first and only boats to travel all the way up through the Grand Canyon.

Sir Hamilton and his first jet boat (1958). Source: ipenz
And the list doesn’t stop there. We could go on with other great firsts:
- Sir Ed (Hillary) – first to summit Everest (to be fair, it was a team effort)
- Sir Ernest (Rutherford) – first to split the atom
- Sir Peter (Blake) – first to sail around the world in less than 75 days (again, a bit of a team effort)
- Sir John (Walker) – first to run the mile in under 3:50 (now a member of the Parkinson’s community)
- Georgina Beyer – first openly transsexual mayor, and then the world’s first openly transsexual Member of Parliament
- AJ Hackett & Henry van Asch – set up the first commercial bungy jump on the Kawarau Bridge, near Queenstown
- Helen Clark, Dame Siliva Cartwright & Sian Elias – first country to have women in the top three senior public roles (Prime Minister, the Governor General, & the Chief Justice, respectively)
- Rocket Lab – first private company in the Southern Hemisphere to reach space (in 2009)
And I guess we better stop there (if only out for fear of making larger nations feel somewhat inadequate), but you get the idea – small nation, doing lots of great stuff.
There is also a very proactive Parkinson’s community – with groups like Parkinson’s New Zealand organising and running support groups across the country, and helping to fund some of the great local Parkinson’s research.

And some of that local Parkinson’s research has been discussed previously on this website (Click here, here and here for examples of previous posts), and it is also the focus of today’s post:
Continue reading “The Kiwi Ketogenic study” →
 Source: Herantis
Source: Herantis