|
Nuclear receptor related 1 protein (or NURR1) is a protein that is critical to the development and survival of dopamine neurons – the cells in the brain that are affected in Parkinson’s disease. Given the importance of this protein for the survival of these cells, a lot of research has been conducted on finding activators of NURR1. In today’s post we will look at this research, discuss the results, and consider issues with regards to using these activators in Parkinson’s disease. |
Comet Hale–Bopp. Source: Physics.smu.edu
Back in 1997, 10 days after Comet Hale–Bopp passed perihelion (April 1, 1997 – no joke; perihelion being the the point in the orbit of a comet when it is nearest to the sun) and just two days before golfer Tiger Woods won his first Masters Tournament, some researchers in Stockholm (Sweden) published the results of a study that would have a major impact on our understanding of how to keep dopamine neurons alive.
Dopamine neurons are one group of cells in the brain that are severely affected by Parkinson’s disease. By the time a person begins to exhibit the movement symptoms of the condition, they will have lost 40-60% of the dopamine neurons in a region called the substantia nigra. In the image below, there are two sections of brain – cut on a horizontal plane through the midbrain at the level of the substantia nigra – one displaying a normal compliment of dopamine neurons and the other from a person who passed away with Parkinson’s demonstrating a reduction in this cell population.
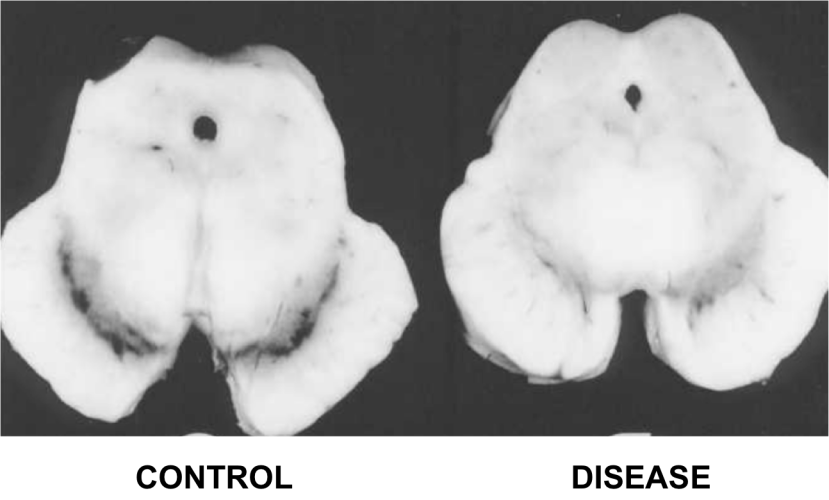
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinson’s disease brain (right). Source:Memorangapp
The researchers in Sweden had made an amazing discovery – they had identified a single gene that was critical to the survival of dopamine neurons. When they artificially mutated the section of DNA where this gene lives – an action which resulted in no protein for this gene being produced – they generated genetically engineered mice with no dopamine neurons:
 Title: Dopamine neuron agenesis in Nurr1-deficient mice
Title: Dopamine neuron agenesis in Nurr1-deficient mice
Authors: Zetterström RH, Solomin L, Jansson L, Hoffer BJ, Olson L, Perlmann T.
Journal: Science. 1997 Apr 11;276(5310):248-50.
PMID: 9092472
The researchers who conducted this study found that the mice with no NURR1 protein exhibited very little movement and did not survive long after birth. And this result was very quickly replicated by other research groups (Click here and here to see examples)
So what was this amazing gene called?
Nuclear receptor related 1 protein (or NURR1; it is also known as NR4A2 – nuclear receptor subfamily 4, group A, member 2)
And what is NURR1?
NURR1 is a transcription factor.
Remind me again, what is a transcription factor?
Remember those high school science class when you were half asleep and the adult at the front of the class was explaining biology 101? They were saying something about DNA giving rise to RNA, RNA giving rise to protein? The central dogma of biology? Does any of this sound familiar?

The basic of biology. Source: Youtube
Ultimately this DNA-RNA-Protein mechanism is a circular cycle, because the protein that is produced using RNA is required at all levels of this process. Some of the protein is required for making RNA from DNA, while other proteins are required for making protein from the RNA instructions.
A transcription factor is a protein that is involved in the process of converting (or transcribing) DNA into RNA.
Importantly, a transcription factor can be an ‘activator’ of transcription – that is initiating or helping the process of generating RNA from DNA.

An example of a transcriptional activator. Source: Khan Academy
Or it can be a repressor of transcription – blocking the machinery (required for generating RNA) from doing it’s work.

An example of a transcriptional repressor. Source: Khan Academy
NURR1 is a really interesting protein because it can function as either a transcriptional activator OR a transcriptional repressor, depending on the situation in the cell. Here we will look solely at it as a transcriptional activator.
So what does NURR1 activate?
NURR1 is very important for activating the transcription of a lot of genes that are required for making and maintaining a dopamine neuron.
These NURR1 activated genes include many involved in the processing of dopamine, such as a gene called tyrosine hydroxylase cause turns the chemical tyrosine into DOPA. DOPA is then turned into dopamine by another NURR1 activated gene called Aromatic L-amino acid decarboxylase (or AADC). Thus, no NURR1, no dopamine.
NURR1 also activates genes associated with the survival of the dopamine neurons, such as the receptor c-RET. cRET is the receptor for the neurotrophic factor GDNF (or Glial cell-derived neurotrophic factor) which has had a long history associated with Parkinson’s disease (Click here to read a previous post on GDNF and PD).
And this close relationship between NURR1 and important survival factors is why when there is no NURR1, there is no dopamine neuron.
How does NURR1 work then?
NURR1 has two regions of interaction:
- The DNA binding domain (DBD)
- The ligand binding domain (LBD)
The transcriptional activity of NURR1. Source: PNAS
With regards to the DNA binding domain, this is the part of the protein that actually makes contact with DNA>
Nurr1 is known to activate transcription of certain genes by binding to a specific section of DNA called the NGFI-B response element (or NBRE). It does this by itself (or as a monomer). NURR1 can also bind to another section of DNA called the Nur response element (or NurRE). But it does this by pairing up with another NURR1 protein (two of them together is called a homodimer). And there is actually a third way Nurr1 can activate transcription: it combines with a different protein called retinoid X receptor (or RXR). This pairing is called a heterodimer and this pair bind to a region of DNA called the DR5 site.
Now, the heterodimer partnering is very important.
This is because NURR1, by itself, does not have a natural ligand. A ligand is a small molecule that binds to a protein and has biological function. Like a key that can activate the protein (in some cases). And, YES I know that NURR1 has a ‘ligand binding domain’ – the region of the protein where ligands bind, but after two decades of looking, we are yet to find a NURR1 ligand. The absence of a ligand suggests that NUUR1 by itself is always active.
But in the heterodimer situation – where NURR1 and RXR combine – things are different. RXR does have ligands, and this has lead many researchers to go looking for ligands that can activate this pairing and cause more transcription of target genes.
Have researchers found any ligands for NURR1/RXR?
Yes (and from this point on we’ll simply refer to these ligands as activators of NURR1).
In 2013, a research report was published that described one such ligand:

Title: Low dose bexarotene treatment rescues dopamine neurons and restores behavioral function in models of Parkinson’s disease.
Authors: McFarland K, Spalding TA, Hubbard D, Ma JN, Olsson R, Burstein ES.
Journal: ACS Chem Neurosci. 2013 Nov 20;4(11):1430-8.
PMID: 24117438 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers at a company called Acadia Pharmaceuticals conducted a large screening of possible NURR1/RXR ligands, and they discovered that the cancer drug bexarotene (also known as Targretin) preferentially bound to the NURR1/RXR heterodimer (and interestingly, not the RXR/RXR homodimer).
To test the effectiveness of this cancer drug in a model of Parkinson’s, the investigators gave rats a neurotoxin that models Parkinson’s disease (6-OHDA) and 3 days later they started treating the animals daily with bexarotene (or a placebo control) for 28 days. Remarkably, at doses as much as 100x lower than those effective in rodent cancer models, bexarotene treatment rescued the dopamine neurons and the associated motor problems.
Dopamine neurons are stained in green. Source:
The researchers concluded that “low oral doses of bexarotene may provide an effective and tolerated therapy for Parkinson’s disease”.
Sounds amazing right?
…
Almost too good to be true?
…
…
Well, actually it may be too good to be true.
Some independent research groups in Sweden also tested bexarotene in models of Parkinson’s disease and they got….different results:

Title: Nurr1 and Retinoid X Receptor Ligands Stimulate Ret Signaling in Dopamine Neurons and Can Alleviate α-Synuclein Disrupted Gene Expression.
Authors: Volakakis N, Tiklova K, Decressac M, Papathanou M, Mattsson B, Gillberg L, Nobre A, Björklund A, Perlmann T.
Journal: J Neurosci. 2015 Oct 21;35(42):14370-85.
PMID: 26490873 (This article is OPEN ACCESS if you would like to read it)
The researchers who conducted this study had previously demonstrated that the Parkinson’s associated protein alpha synuclein can reduce levels of NURR1 (Click here to read more about this). In this follow-up study, they were interested to know if treating cells with NURR1 activators could rescue the cells in models of Parkinson’s. By forcing NURR1 protein production in cells that had too much alpha synuclein, they were able to restored the normal activity of hundreds of dysregulated genes.
Next they tested whether Nurr1 can be pharmacologically targeted with bexarotene (the ligand for RXR mentioned above). They found that bexarotene was ineffective in providing neuroprotection in rat model of Parkinson’s (the daily treatment of bexarotene was started on the day of 6-OHDA neurotoxin administration). But their results did reveal that bexarotene could restore subsets of NURR1 target genes, particularly the GDNF receptor c-Ret. This result led the investigators to conclude that while bexarotene may not be potent enough for use in the clinical treatment of Parkinson’s disease, other NURR1/RXR targeting agents could be considered.
This result may not be the end of bexarotene, however.
More recently, a group of researchers Poland and Vietnam (what a cool collaboration!) found something interesting about Bexarotene:

Title: Bexarotene Does Not Clear Amyloid Beta Plaques but Delays Fibril Growth: Molecular Mechanisms.
Authors: Huy PDQ, Thai NQ, Bednarikova Z, Phuc LH, Linh HQ, Gazova Z, Li MS.
Journal: ACS Chem Neurosci. 2017 Sep 20;8(9):1960-1969.
PMID: 28689412
In this study, the research found that bexarotene delays the assembly of the toxic form of the Alzheimer’s protein beta-amyloid. Beta-amyloid has been found to aggregate in the brains of people with Alzheimer’s, and these research found that at high concentrations bexarotene can slow down that clustering process. It does this through reduction of the beta-content of the individual beta-amyloid proteins (called monomers). It is the first compound to display such an unusual behaviour. One could question whether it could do the same thing in Parkinson’s?
Bexarotene is being tested in a phase 2 clinical trial in Alzheimer’s disease. In this clinical trial, bexarotene was tested on people with Alzheimer’s disease, but the interesting part of the study was that some of those people had a specific genetic mutation (APOE4 allele) that increases the risk for Alzheimer’s, while others did not. The study found that bexarotene had no effect on the beta amyloid protein burden in people with the APOE4 allele mutation, but did reduce beta amyloid protein in those who did not have the mutation. The study did not have enough subjects to make a conclusion with regards to cognitive ability, but the initial result is still interesting (Click here to read more on this trial).
All of that said, there is a health warning here with regards to bexarotene: it has effects on blood lipids which may increase the risk of stroke and heart attack. Thus, until this drug has been clinically investigated further it should not be used off label for clinical treatment.
Ok, but have other NURR1 activating drugs been identified?
Again, yes.
Title: Nuclear receptor Nurr1 agonists enhance its dual functions and improve behavioral deficits in an animal model of Parkinson’s disease.
Authors: Kim CH, Han BS, Moon J, Kim DJ, Shin J, Rajan S, Nguyen QT, Sohn M, Kim WG, Han M, Jeong I, Kim KS, Lee EH, Tu Y, Naffin-Olivos JL, Park CH, Ringe D, Yoon HS, Petsko GA, Kim KS.
Journal: Proc Natl Acad Sci U S A. 2015 Jul 14;112(28):8756-61.
PMID: 26124091 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers successfully screened a large US Food and Drug Administration (FDA)-approved drug library (holding 960 different compounds) to identify NURR1-activating compounds. They identified three:
- amodiaquine (anti-malaria drug)
- chloroquine (anti-malaria drug)
- glafenine (a pain-relieving drug)
All three of these drugs were able to enhance the transcriptional activation of many of the genes specific to dopamine neurons. Remarkably, they were also able to de-activate genes involved in the inflammatory activities of microglia.
Microglia are cells in the brain that form the main part of active immune defence to injury or disease. When any kind of trouble kicks off in the brain, microglia become activated and start dealing with the source of the trouble as well as removing any damaged/dying cells. They communicate with other cells (particularly cells of the immune system, such a T-cells and macrophage) by releasing chemicals which is part of the process of inflammation.
Microglia become activated around sick cells and release pro-inflammatory compounds. Source: NCBI
NURR1 is present in microglia and it is important for the regulation of the process of inflammation. NURR1 represses inflammation, thus any agents that increase NURR1 levels of activity will reduce the microglia response in the brain.
Importantly, all three of these compounds significantly improved behavioural motor deficits in rodent models of Parkinson’s disease (using the neurotoxin 6-OHDA). These findings actually confirmed previously published research conducted in Parkinsonian primates that demonstrated chloroquine was neuroprotective (Click here to read more on this research). The scientists are now seeking to design better drugs by modifying chloroquine and amodiaquine with the hope of carrying out clinical trials soon.
EDITOR’S NOTE: The reason that better drugs need to be designed is because both chloroquine and amodiaquine can have very serious side effects (Click here and here to read more on this, respectively).
The scientist that led this research, Prof Kwang-Soo Kim (Harvard University), recently sat down and explained the results. If nothing I’ve written here makes any sense, I can highly recommend you watch this video:
Ok, but have other activators been found?
Yes. In addition to clinically available drugs, novel activators have also been discovered:

Title: Selective brain penetrable Nurr1 transactivator for treating Parkinson’s disease.
Authors: Wang J, Bi W, Zhao W, Varghese M, Koch RJ, Walker RH, Chandraratna RA, Sanders ME, Janesick A, Blumberg B, Ward L, Ho L, Pasinetti GM.
Journal: Oncotarget. 2016 Feb 16;7(7):7469-79.
PMID: 26862735 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in assessing the potential of their novel compound (named IRX4204) as a treatment for Parkinson’s disease. The researchers used a rodent model of Parkinson’s (6-OHDA) to test their drug, and they found that IRX4204 not only entered the brain and promoted the survival of dopamine neurons – in a dose-dependent manner – but it also restored the normal activation of many of the dopamine neuron-specific genes.
And the good news here is that a small, open label phase I clinical trial assessing the safety of IRX4204 has recently been completed by the biotech company Io Therapeutics. We are still awaiting the results of that study (Click here to read more about this trial).
In addition to this drug, others are also being tested:
Title: A Nurr1 agonist causes neuroprotection in a Parkinson’s disease lesion model primed with the toll-like receptor 3 dsRNA inflammatory stimulant poly(I:C)
Authors: Smith GA, Rocha EM, Rooney T, Barneoud P, McLean JR, Beagan J, Osborn T, Coimbra M, Luo Y, Hallett PJ, Isacson O.
Journal: PLoS One. 2015 Mar 27;10(3):e0121072.
PMID: 25815475 (This article is OPEN ACCESS if you would like to read it)
The research involves a NURR1 activator that has been identified by the pharmaceutical company Sanofi-aventis and this study was supported by the Michael J Fox Foundation. The drug (called SA00025) was found to enter the brain and regulate the transcription of dopamine neuron-specific genes. It also provided neuroprotection in a 6-OHDA neurotoxin model of Parkinson’s disease AND displayed anti-inflammatory activity.
Strangely, Sanofi-aventis has nothing about SA00025 on their website, and there are currently no planned or ongoing clinical trials for SA00025 (Source). This may relate to unpublished research that is mentioned on the Michael J Fox Foundation page about the grant associated with this drug – the results section reads:
“In the subsequent neuroprotection studies, SA00025 showed no significant effect on dopaminergic neuron degeneration in the alpha-synuclein model”
No effect in a more disease relevant model of Parkinson’s disease (alpha synuclein) may have made Sanofi-aventis weary of moving ahead with the development of this drug. We shall see what happens.
Other novel compound options are on the table though:
Title: Nurr1:RXRα heterodimer activation as monotherapy for Parkinson’s disease.
Authors: Spathis AD, Asvos X, Ziavra D, Karampelas T, Topouzis S, Cournia Z, Qing X, Alexakos P, Smits LM, Dalla C, Rideout HJ, Schwamborn JC, Tamvakopoulos C, Fokas D, Vassilatis DK.
Journal: Proc Natl Acad Sci U S A. 2017 Apr 11;114(15):3999-4004.
PMID: 28348207 (This article is OPEN ACCESS if you would like to read it)
The researchers conducting this study designed a unique Nurr1:RXR-selective activator that they called BRF110. This drug easily enters the brain of mice and prevents dopamine neuron loss in multiple models of Parkinson’s (MPTP, 6-OHDA and Alpha synuclein models). It increases the levels of activation of dopamine neuron specific genes, and does not cause any dyskinesia (abnormal involuntary movements associated with long term use of L-dopa). The researchers also tested BRF110 on dopamine neurons generated from skin cells collected from people with Parkinson’s-related genetic mutations (LRRK2-G2019S mutation) – this is done by turning the skins cells into induced pluripotent stem cell (iPSC- click here to read a previous post about this amazing technology). Remarkably, BRF110 demonstrated positive effects on these patient derived cells.
This research was only published earlier this year and we are yet to hear what will happen next with this drug. Not clinical trials have been registered to date (Source). I’ll let you know when I hear anything
What does it all mean?
The point of this kind of post is to make people aware of experimental classes of drugs that they have probably never heard of. And there are a lot of them. NURR1 is a very interesting target, but like LRRK2 inhibitors, we are yet to determine the scope of off-target effects with long term usage. NURR1 is not specific to dopamine neurons (for example, as we discussed above microglia have NURR1). Thus any future research should investigate what impact these NURR1 activators are having on other cell types.
The research that has been presented thus far, however, is very exciting and indicates that this class of drug (NURR1 activator) could provide a useful tool in the neuroprotective aspect of the larger “Cure for Parkinson’s” framework (any “cure” for the condition will require three components: 1. a disease halting treatment, 2. a neuroprotective agent, and 3. a cell replacement therapy – click here to read more about this). NURR1 is a very interesting target, and I will be watching this area of research very closely.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Youtube








Thank you very much Sir for putting all of this researches together but is there any new news on the cure? is the clinical trial started? and if it’s started did it succeed?
LikeLike
Hi Ahmed,
Thanks for your comment. Unfortunately, there is still no news regarding any of the drugs mentioned in this post. I have contacted Io Therapeutics, but I am yet to have a response from them regarding results from their 2015 clinical trial of IRX4204. I will post them here when I hear from the company.
Kind regards,
Simon
LikeLike
Thanks for pulling all of this info together!
I am curious about memantine/Namenda…is it a potentially effective NURR1 agonist?
LikeLike
Hi Trish,
Thanks for your question. The only study that I’m aware of that suggests a NURR1 role for memantine is Wei et al (2016) Mol. Neurobiol. (https://www.ncbi.nlm.nih.gov/pubmed/26497037). But in this study they do not demonstrate a direct relationship, so we can’t say that memantine is a ‘NURR1 agonist’. Memantine is generally considered a NMDA receptor antagonist, but it is also a affinity dopamine D2 agonist (https://www.ncbi.nlm.nih.gov/pubmed/18000814). It is difficult to determine in the Wei et al study if the increase in NURR1 is simply a result of the cells feeling better (as a result of NMDA antagonism). All of the “neuroprotective protein” levels might be increasing as a result of the NMDA receptor blockage. Memantine has been clinically tested in people with Lewy-body-related dementias (a form of Parkinson’s – https://www.ncbi.nlm.nih.gov/pubmed/20729148), but the effect was mild. I hope this answers your question.
Kind regards,
Simon
LikeLike
Thank you, Simon,
I was reading this article:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4833611/
…and wondering about some of the compounds listed in Table 2. (I see now that the memantine is cited to the same study you just shared).
FWIW, a family member of mine was recently found to have a genetic microdeletion that includes NR4A2/NURR1, so this is the reason for my research. Not PD, though there are some motor coordination issues, which could likely be attributed to a NURR1 deficit, if I am following this correctly.
If you are aware of any other compounds that might upregulate NURR1 (in a non-Parkinson’s case), please continue to update!
Best,
Trish
LikeLike
Hi Trish,
I am currently not aware of any clinically available NURR1 specific activators. I will certainly let you know when any pop up in the literature as this is a very hot area in PD research. The problem for the PD field is that a drug that activates NURR1 could have serious side effects as it will affect cell types not associated with PD. Your family member is fortunate in this regard as you are probably seeking this broad activation of NURR1 in those other cell types.
One of the drugs on the list in this article – Isoxazolo-pyridinone 7e (IP7e) – is really intriguing. It is a blood brain barrier crossing activator of the Nurr1 that was being developed by Novartis. It first appeared in 2007 (https://www.ncbi.nlm.nih.gov/pubmed/17035009), but I have seen very little about it since 2014 (https://www.ncbi.nlm.nih.gov/pubmed/25265488). It could be interesting to follow this up with Novartis. I have emailed the researchers behind the 2014 study to ask if they know of any new developments. I will let you know if they respond.
Kind regards,
Simon
LikeLike