|
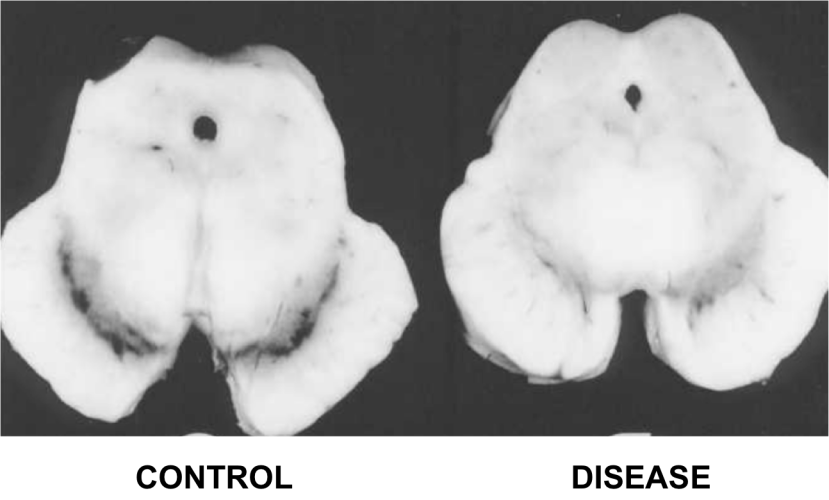
Every textbook written about the condition will tell you that the classical pathological characteristic of Parkinson’s is the loss of dopamine neurons in the midbrain region of the brain. It is the distinguishing feature that pathologists look for in order to provide a postmortem diagnosis of the condition. But what is meant by the words ‘loss of dopamine neurons’? Do the cells actually die? Recently researchers from Korea have published new data exploring this question. Interestingly, they found evidence of ‘dormant’ dopamine neurons in postmortem sections of brains from people with Parkinson’s – even those with severe forms of the condition. In today’s post, we will discuss what a dopamine neuron is, what this new research found, and what it could mean for our understanding of Parkinson’s.
|
Source: Bettys
2019 represented the centenary year for an important discovery in Parkinson’s research.
In 1919, the Uzbek neuropathologist Konstantin Tretiakoff (1892-1958) reported his findings regarding an examination of 54 human brains.
 Konstantin Tretiakoff. Source: Wikipedia
Konstantin Tretiakoff. Source: Wikipedia
Six of the postmortem brains had belonged to individuals who had suffered from Parkinson’s and three others had been diagnosed with postencephalitic Parkinsonism. In these brains he noticed something rather striking.
What did he find?