
When people in England think of the city of Sheffield, quite often images of a great industrial past will come to mind.
They usually don’t think of the flies, fish and (yes) a Tigar (no, not a typo!) that are influencing Parkinson’s disease research in the city.
In today’s post we will look at how the re-invention of a city could have a major impact on Parkinson’s disease.

The industrial heritage of Sheffield. Source: SIMT
It is no under statement to say that the history of Sheffield – a city in South Yorkshire, England – is forged in steel.
In his 1724 book, “A tour thro’ the whole island of Great Britain“, the author Daniel Defoe wrote of Sheffield:
“Here they make all sorts of cutlery-ware, but especially that of edged-tools, knives, razors, axes, &. and nails; and here the only mill of the sort, which was in use in England for some time was set up, for turning their grindstones, though now ’tis grown more common”
Sheffield has a long history of metal work, thanks largely to its geology: The city is surrounded by fast-flowing rivers and hills containing many of the essential raw materials such as coal and iron ore.
And given this fortunate circumstance and an industrious culture, the city of Sheffield particularly prospered during the industrial revolution of the mid-late 1800s (as is evident from the population growth during that period).

The population of Sheffield over time. Source: Wikipedia
But traditional manufacturing in Sheffield (along with many other areas in the UK) declined during the 20th century and the city has been forced to re-invent itself in the early 21st century. And this time, rather than taking advantage of their physical assets, the city is focusing on its mental resources.
Great. Interesting stuff. Really. But what does this have to do with flies, fish and Parkinson’s disease???
Indeed. Let’s get down to business.

The Sheffield Institute for Translational Neuroscience (SITraN) was officially opened in 2010 by Her Majesty The Queen. It is the first European Institute purpose-built and dedicated to basic and clinical research into Motor Neuron Disease as well as related neurodegenerative disorders such as Parkinson’s and Alzheimer’s disease.
Since its opening, the institute has published some pretty impressive research, particularly in the field of Parkinson’s disease.
And here is where we get to the flies:

Pink flies. Source: Wallpapersinhq
We have previously discussed “Pink” flies and their critical role in Parkinson’s research (Click here to read that post).
Today we are going to talk about Lrrk2 flies.
What is Lrrk2?
This is Sergey Brin.

He’s a dude.
One of the founders of the search engine company “Google”. Having changed the world, he is now turning his attention to other projects.
One of those other projects is close to our hearts: Parkinson’s disease.
In 1996, Sergey’s mother started experiencing numbness in her hands. Initially it was believed to be RSI (Repetitive strain injury). But then her left leg started to drag. In 1999, following a series of tests, Sergey’s mother was diagnosed with Parkinson’s disease. It was not the first time the family had been affected by the condition: Sergey’s late aunt had also had Parkinson’s disease.
Both Sergey and his mother have had their DNA scanned for mutations that increase the risk of Parkinson’s disease. And they discovered that they were both carrying a mutation on the 12th chromosome, in a gene called PARK8 – one of the Parkinson’s disease associated genes. Autosomal dominant mutations (meaning if you have just one copy of the mutated gene) in the PARK8 gene dramatically increase one’s risk of developing Parkinson’s disease.
PARK8 provides the instructions for making an enzyme called Leucine-rich repeat kinase 2 (or Lrrk2).

The structure of Lrrk2. Source: Wikipedia
Also known as ‘Dardarin‘ (from the Basque word “dardara” which means trembling), Lrrk2 has many functions within a cell – from helping to move things around inside the cell to helping to keep the power on (involved with mitochondrial function).
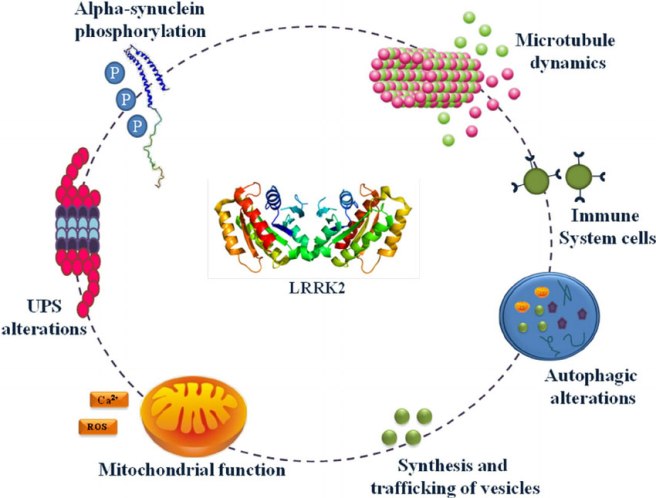
Source: Researchgate
NOTE: Curiously, mutations in the PARK8 gene are also associated with Crohn’s disease (Click here and here for more on this) – though the mutation is in a different location for PD.
Now, not everyone with this particular mutation will go on to develop Parkinson’s disease, and Sergey has decided that his chances are 50:50. But he does not appear to be taking any chances though. Being one of the founders of a large company like Google, has left Sergey with considerable resources at his disposal. And he has chosen to focus some of those resources on Lrrk2 research (call it an insurance policy). He has done this via considerable donations to groups like the Michael J Fox foundation.
Actor Michael J Fox was diagnosed at age 30. Source: MJFox foundation
So just as Pink flies derive their name from mutations in the Parkinson’s associated Pink1 gene, Lrrk2 flies have mutations in the Lrrk2 gene.
So what have the researchers at Sheffield done with the Lrrk2 flies?
In 2013, the Sheffield researchers published an interesting research report:

Title: Ursocholanic acid rescues mitochondrial function in common forms of familial Parkinson’s disease
Authors: Mortiboys H, Aasly J, Bandmann O.
Journal: Brain. 2013 Oct;136(Pt 10):3038-50.
PMID: 24000005
In this study, the investigators took 2000 drugs (including 1040 licensed drugs and 580 naturally occurring compounds) and conducted a massive screen to identify drugs that could rescue mitochondrial dysfunction in PARK2 (Parkin) mutant cells.
Mitochondria are the power house of each cell. They keep the lights on. Without them, the lights go out and the cell dies.

Mitochondria and their location in the cell. Source: NCBI
In certain genetic forms of Parkinson’s disease (such as those associated with mutations in the PARK2 gene), the mitochondria in cells becomes dysfunctional and may not be disposed of properly (Click here to read our previous post related to this).
In their huge screen of 2000 drugs, the researchers in Sheffield identified 15 drugs that could rescue the mitochondria dysfunction in the PARK2 skins cells. Of those 15 compounds, two were chosen for further functional studies. They were:
- Ursocholanic acid
- Dehydro(11,12)ursolic acid lactone
Neither ursocholanic acid nor dehydro(11,12)ursolic acid lactone are FDA-licensed drugs. We have little if any information regarding their use in humans. Given this situation, the researchers turned their attention to the chemically related bile acid ‘ursodeoxycholic acid’, which has been in clinical use for more than 30 years.
What is Ursodeoxycholic Acid?
Ursodeoxycholic Acid (or UDCA) is a drug that is used to to improve bile flow and reduce gallstone formation. In the USA it is also known as ‘ursodiol’.

Ursodiol. Source: Wikimedia
Bile is a fluid that is made and released by your liver, and it stored in the gallbladder. Its function is to help us with digestion. UDCA occurs naturally in bile – it is basically a bile acid and can therefore be useful in dissolving gallstones. UDCA has been licensed for the treatment of patients since 1980. UDCA also reduces cholesterol absorption.
So what did the Sheffield researchers find with UDCA?
The researchers tested UDCA on mitochondrial function in PARK2 skin cells, and they found that the drug rescued the cells. They then tested UDCA on skin cells from people with Parkinson’s disease who had mutations in the PARK8 (Lrrk2) gene (G2019S).
The researchers had previously found impaired mitochondrial function and morphology in skin cells taken from people with PARK8 associated Parkinson’s disease (Click here to read more about this), and other groups had reported similar findings (Click here for more on this).
And when they treated the Lrrk2 cells with UDCA, guess what happened?
UDCA was able to rescue the mitochondrial effect in those cells as well!
Obviously these results excited the Sheffield scientists and they set up a collaboration with researchers at York University and from Norway, to look at the potential of UDCA in rescuing the fate of Lrrk2 flies. The results of that study were published two years ago:

Title: UDCA exerts beneficial effect on mitochondrial dysfunction in Lrrk2 (G2019S) carriers and in vivo.
Authors: Mortiboys H, Furmston R, Bronstad G, Aasly J, Elliott C, Bandmann O.
Journal: Neurology. 2015 Sep 8;85(10):846-52.
PMID: 26253449 (This article is OPEN ACCESS if you would like to read it).
The researchers tested UDCA on flies (or drosophila) with specific Lrrk2 mutations (G2019S) display a progressive loss of photoreceptor cell function in their eyes. The mitochondria in the photoreceptor are swollen and disorganised. When the investigators treated the flies with UDCA, they found approximately 70% rescue of the photoreceptor cells function.
The researchers in Sheffield concluded that UDCA has a marked rescue effect on cells from a Parkinson’s disease-associated gene mutation model, and they proposed that “mitochondrial rescue agents may be a promising novel strategy for disease-modifying therapy in Lrrk2-related PD, either given alone or in combination with Lrrk2 kinase inhibitors” (for more information about the Lrrk2 inhibitors they refer, click here).
And the good news regarding this line of research: other research groups have also observed similar beneficial effects with UDCA in models of Parkinson’s disease:

Title: Ursodeoxycholic acid suppresses mitochondria-dependent programmed cell death induced by sodium nitroprusside in SH-SY5Y cells.
Authors: Chun HS, Low WC.
Journal: Toxicology. 2012 Feb 26;292(2-3):105-12.
PMID: 22178905
This research group also demonstrated that UDCA could reduce cell death in a cellular model of Parkinson’s disease.
And this study was followed by another one from a different research group, which involved testing UDCA in animals:

Title: Ursodeoxycholic Acid Ameliorates Apoptotic Cascade in the Rotenone Model of Parkinson’s Disease: Modulation of Mitochondrial Perturbations.
Authors: Abdelkader NF, Safar MM, Salem HA.
Title: Mol Neurobiol. 2016 Mar;53(2):810-7.
PMID: 25502462
These researchers found UDCA rescued a rodent model of Parkinson’s disease (involving the neurotoxin rotenone). UDCA not only improved mitochondrial performance in the rats, but also demonstrated anti-inflammatory and anti-cell death properties.
Given all this research, the Sheffield researchers are now keen to test UDCA in clinical trials for Parkinson’s disease.
Has anyone tested UDCA in the clinic for Parkinson’s disease?
Not that we are aware of, but two groups are interested in attempting it.
Firstly, the University of Minnesota – Clinical and Translational Science Institute has registered a trial (Click here to read more about this). This trial will not, however, be testing efficacy of the drug on Parkinson’s symptoms. It will focus on measuring UDCA levels in individuals after four weeks of repeated high doses of oral UDCA (50mg/kg/day), and determining the bioenergetic profile and ATPase activity in those participants. Basically, they want to see if UDCA is safe and active in people with Parkinson’s disease.
The CurePD trust (in the UK) is also currently seeking to run a clinical trial for UDCA (Click here for more on this). The group are currently organising the funding for that trial.

EDITOR’S NOTE HERE: Before we move on, the team at the SoPD would like to say that while UDCA is a clinically available drug, it is still experimental for Parkinson’s disease. There is no indication yet that it has beneficial effects in people with Parkinson’s disease. In addition, UDCA is also is known to have side effects, which include flu symptoms, nausea, diarrhea, and back pain. And individuals have been known to have allergic reactions to UDCA treatment (Click here and here for more on the side effects of UDCA). Thus we must impress caution on anyone planning to experiment with this drug. Before attempting any kind of change in a current treatment regime, PLEASE discuss your plans with a medically qualified physician who is familiar with your case history.
Ok, so that was the flies research, what about the fish? And the… uh, tigar?
Yes. The fish are called Zebrafish (or Danio rerio).
They are a tropical freshwater fish that is widely used in biological research.

Biology researchers love these little guys because their genome has been fully sequenced and they has well characterised and testable behaviours. In addition, their development is very rapid (3 months), and its embryos are large and transparent.
And the researchers at Sheffield are using these fish to study Parkinson’s disease.
How did they do that?

Title: TigarB causes mitochondrial dysfunction and neuronal loss in Pink1 deficiency
Authors: Flinn LJ, Keatinge M, Bretaud S, Mortiboys H, Matsui H, De Felice E, Woodroof HI, Brown L, McTighe A, Soellner R, Allen CE, Heath PR, Milo M, Muqit MM, Reichert AS, Köster RW, Ingham PW, Bandmann O.
Journal: Ann Neurol. 2013 Dec;74(6):837-47.
PMID: 24027110 (This article is OPEN ACCESS if you would like to read it)
Firstly, the group at Sheffield generated zebrafish that had a mutation in the Parkinson’s associated gene ‘PARK6’. This gene provides the plans for the production of a protein called Pink1 (we have previously discussed Pink1 – click here to read more on this).
In normal healthy cells, the Pink1 protein is absorbed by mitochondria and eventually degraded as it is not used. In unhealthy cells, however, this process becomes inhibited and Pink1 starts to accumulate on the outer surface of the mitochondria. Sitting on the surface, it starts grabbing another Parkinson’s associated protein called Parkin. This pairing is a signal to the cell that this particular mitochondria is not healthy and needs to be removed.

Pink1 and Parkin in normal (right) and unhealthy (left) situations. Source: Hindawi
The process by which mitochondria are removed is called mitophagy. Mitophagy is part of the autophagy process, which is an absolutely essential function in a cell. Without autophagy, old proteins and mitochondria will pile up making the cell sick and eventually it dies. Through the process of autophagy, the cell can break down the old protein, clearing the way for fresh new proteins to do their job.
Think of autophagy as the waste disposal/recycling process of the cell.

The process of autophagy. Source: Wormbook
Waste material inside a cell is collected in membranes that form sacs (called vesicles). These vesicles then bind to another sac (called a lysosome) which contains enzymes that will breakdown and degrade the waste material. The degraded waste material can then be recycled or disposed of by spitting it out of the cell.
In the case of a PARK6 mutations, Pink1 protein can not function properly with Parkin and the autophagy process breaks down. As a result, the old or unhealthy mitochondria start to pile up in the cell, resulting in the cell getting sick and dying.
Now back to the Zebrafish.
When the Sheffield researchers mutated PARK6 in the zebrafish, they noticed that the fish had a very early and persistent loss of dopamine neurons in their brains. These fish also had enlarged, unhealthy mitochondria and reduced mitochondrial activity.
Given this result, the investigators next wanted to identify which genes have increased or decreased levels of activity as a result of this genetic manipulation. They identified 108 genes that were higher in the PARK6 mutant, and 146 genes had lower activity.
One gene in particular had activity levels 12 times higher in the PARK6 mutant fish than the normal zebrafish.
The name of that gene? TP53-Induced Glycolysis And Apoptosis Regulator (or Tigar).
What is Tigar?
Tigar is a gene that provides the instructions for making a protein that is activated by p53 (also known as TP53).
What does that mean?
p53 is a protein that has three major functions: controlling cell division, DNA repair, and apoptosis (or cell death). p53 performs these functions as a transcriptional activator (that is a protein that binds to DNA and helps produce RNA (the process of transcription) – see our previous post explaining this).
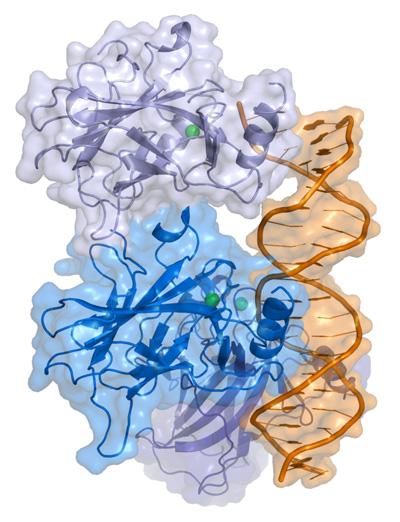
p53 protein structure, bound to DNA (in gold). Source: Wikipedia
In regulating the cell division, p53 prevents cells from dividing too much and in this role it is known as a tumour suppression – it suppresses the emergence of cancerous tumours. Genetic mutations in the p53 gene result in run away cell division, and (surprise!) as many as 50% of all human tumours contain mutations in the p53 gene.

Cancer vs no cancer. Source: Khan Academy
In DNA repair, p53 is sometimes called “the guardian of the genome” as it prevents mutations and helps to conserve stability in the genome. This function also serves to prevent the development of cancer, by helping to repair potentially cancer causing mutations….and in this role it is known as a tumour suppression. Obviously, if there is a mutation in the p53 gene, less DNA repair will occur – increasing the risk of cancer occurring.
And finally, in cell death, p53 plays a critical role in telling a cell when to die. And (continuing with the cancer theme), if there is a mutation in the p53 gene, fewer cells will be told to die – increasing the risk of cancer occurring. And in this role p53 is known as a tumour suppression.
In normal cells, the levels of p53 protein are usually low. When a cell suffers DNA damage and stress, there is often an increase in the amount of p53 protein. If this increases past a particular threshold, then the cell will be instructed to die.
If you haven’t guessed yet, p53 is a major player inside most cell, and it controls the activity of a lot of genes.
And one of those genes is Tigar.
But what does Tigar actually do?
So we have explained the “TP53-Induced” part of the “TP53-Induced Glycolysis And Apoptosis Regulator” name, let’s now focus on the “Glycolysis And Apoptosis Regulator”
Tigar is an interesting protein because it is an enzyme that primarily functions as a regulator of the breaking down of glucose (“Glycolysis” involves the conversion of glucose into a chemical called pyruvate). In addition to this role, however, Tigar acts in preventing cell death (or apoptosis).
Increased levels of Tigar protects cells from oxidative-stress induced apoptosis, by decreasing the levels of free radicals. In this way, it promotes anti-oxidant activities.
But hang on a second, anti-oxidant activity should be good for the cell right? Why are the dopamine cells are dying if Tigar levels are increasing in the PARK6 mutants?
Fantastic question!
The answer: TIGAR is also a negative regulator of a process called mitophagy. As we discussed above, mitophagy is the process of removing mitochondria by autophagy. Increases in the levels of TIGAR blocks mitophagy in a cell, and results in an increased number of swollen and unhealthy mitochondria in those cells (Click here to read more about this). These swollen mitochondria are comparable to the enlarged mitochondria identified the PARK6 zebrafish by the Sheffield researchers.
And the researchers believe that this may be the cause of the cell death in the PARK6 zebrafish – the double impact of PARK6 and Tigar induced problems with mitophagy.
NOTE: Problems with mitophagy is believed to be an important mechanism in the development of early-onset Parkinson’s disease (Click here for a recent review on this)
Ok, and what did the Sheffield researchers do next?
Given that there was such a huge increase in Tigar levels in the PARK6 zebrafish, the investigators decided to reduce Tigar levels in the PARK6 zebrafish to see what impact this would have on the fish (and their mitochondria).
Remarkably, reductions of Tigar levels resulted in complete rescue of the dopamine neurons in the PARK6 fish. It also increased mitochondrial activity in those cells, and reduced the activation of the microglia cells, which can also play a role in the removal of sick cells in the brain.
The researchers concluded that the results demonstrate that TIGAR is “a promising novel target for disease‐modifying therapy in Pink1‐related Parkinson’s disease”.
And what are the researchers planning to do next with Tigar?
Prof Oliver Bandmann, the senior scientist who ran the study, has said that they “need to finish studying TIGAR levels in the brains of people with Parkinson’s and want to better understand how this protein is involved in maintaining the cell batteries – called ‘mitochondria'” (Source).
Our guess is that the group will also be conducting studies looking at Tigar reduction in rodent models of Parkinson’s disease to determine if this is a viable target in mammals. If Tigar reduction in rodents is found to be effective, the researchers will probably turn their attention to drug screening studies to identify currently available drugs that can reduce the activity of Tigar. Such a drug would provide us with yet another potential treatment for Parkinson’s disease.
We’ll be keeping an eye out for these pieces of research.
This is all very interesting. What does the future hold for Parkinson’s research in Sheffield?
Well, in a word: Keapstone.
Que?
In March, the University of Sheffield and Parkinson’s UK have launched a new £1 million virtual biotech company called “Keapstone Therapeutics” (see the press release by clicking here).

Source: Parkinson’s UK
The goal of the company – the first of its kind – is to combine world-leading research from the University with funding and expertise from the charity to help develop revolutionary drugs for Parkinson’s disease.
What is virtual about it? The biotech won’t be building its own labs, employing a team of specialist laboratory scientists, or buying any high-tech equipment (which would all be incredibly expensive). Rather they will form partnerships with groups that do specific tasks the best.
Here is a video of Dr Author Roach (director of Research at Parkinson’s UK) explaining the idea behind this endeavour:
By seeking a collaboration with Sheffield in the creation of a spin-out biotech company, Parkinson’s UK is not only acknowledging Sheffield’s track record, but also making an investment in their future research. While we cannot be entirely sure of what the long-term future holds for Parkinson’s research in Sheffield, we do know that Keapstone will be an important aspect of it in the immediate future.
Could this be a model for the future of Parkinson’s disease research? Only time will tell. We will have a closer look at Keapstone Therapeutics in an upcoming post.
Click here to learn more about the virtual biotech project.
So what does it all mean?
In 2017, we here at the SoPD have decided to begin highlighting some of the Parkinson’s disease research centres as an addition feature on the blog. We have not been approached by the research group in Sheffield or the University itself, and our selection of this city as our first case study was based purely on the fact that we really like what is happening there with regards to Parkinson’s research!
The research group in Sheffield has undertaken multiple lines of research which could potentially providing us with several novel treatment options for Parkinson’s disease. These lines of research have focused not only on clinically available drugs, but also identifying novel targets. We like what they are doing and will keep a close eye on progress there.
And over the next year we will select additional centres of Parkinson’s research based on the same criteria (us liking what they are doing). Our next case study will be the Van Andel Research Institute in Grand Rapids, Michigan (we would hate to be accused of having a UK bias).
EDITORIAL NOTE: Under absolutely no circumstances should anyone reading the material on this website consider it medical advice. The information provided here is for educational purposes only. Before considering or attempting any change in your treatment regime, PLEASE consult with your doctor or neurologist. While some of the drugs discussed on this website are clinically available, they may have serious side effects. We urge caution and professional consultation before altering any treatment regime. SoPD can not be held responsible for any actions taken based on the information provided here.
The banner for today’s post was sourced from TotalProduceLocal

Nice article. Really. Interesting, fun and nicely explained. But… sorry to be a pedantic PITA, but could you please ease my pain by getting rid of the apostrophes in possessive “its” (4 places in the article) and being consistent about the capitalization of gene names (LRRK2 vs. Lrrk2)? Thanks!
LikeLike
Hi Bromley, thanks for you message and for pointing out the typos. All of these posts are made in the wee small hours after the little one has gone to bed, the dishes are done, etc; and as a result there are always typos. The Lrrk2 vs LRRK2 is a different matter however: in research, the capitalisation indicates the human gene/protein while the lower case is the mouse version. For simplicity sake, in future I’ll just leave it all lower case. Glad you liked the post.
Kind regards,
Simon
LikeLike
Hi Jon,
Thank you very much for your message and interest in our website. We also appreciate the extra info regarding TUDCA and ALS, we’ll look into it.
Unfortunately, we have a policy of not allowing any messages advertising products or services on the website. This is to avoid any ethical or conflict of interest situations. The policy is also in place because we are responsible for all the information present on the website. And it is for this reason that your message will not be posted. We will look into adding a section on TUDCA in a few days when we have some time, as this may be of interest to the readers.
We hope you understand.
Kind regards,
Simon
LikeLike
Hello. I was wondering if you could explain something which I am confused about- I read somewhere (but I can’t remember where) that the Lrrk2 mutation leads to a reduction in energy generated by the cell, but then in this article https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4560055/
it says that the Lrrk2 mutation causes dardarin to become overactive and this is what leads to neuronal cell loss, not the lack of energy the cell has available. Please could you tell me which is correct? Thank you.
LikeLike
Hi Isobel,
Thanks for the interesting question. There is a lot about LRRK2 that is confusing.
LRRK2 is a very busy enzyme that has lots of various functions. Those functions are controlled by different regions (or domains) of the protein. For example, there is a ‘ROC’ domain which is important for binding LRRK2 to similar protein (a process referred to as dimerising) and there is a ‘kinase’ domain, which acts as a regulator of various signal pathways. There are at least 5 genetic mutations in the gene associated with increased risk of PD (G2019S, R1441C, R1441G, I2020T, and Y1699C). And these mutations are split up across the various domains (for example R1441C and R1441G are in the ROC domain and G2019S is in the kinase region). The effect of a particular LRRK2 mutation really depends on where that mutation is on the LRRK2 gene.
Your question deals with one particular mutation: G2019S. LRRK2 protein can generally be found floating around inside the cell serving various functions, but it is also present on the outer surface of the power stations of the cell called the mitochondria. Impaired mitochondrial function and morphology in cells with a LRRK2 G2019S mutation has been reported by several groups (including the folks in Sheffield – https://www.ncbi.nlm.nih.gov/pubmed/21115957/), and this results in decreased energy levels inside the cells.
The hyperactivity you mention refers to the kinase domain basically being active all the time (usually it can be turned on or off). This constant activity is not good. It results in various biological pathways not behaving as they should do, ultimately leading to dysfunction within the cell. The cell itself will realise that something is wrong (both with its under performing mitochondria and with the various other processes that are regulated by the hyperactive LRRK2 kinase domain) and if it can’t correct the problems, then it will shut down and die (via a process called apoptosis). And this is why research reports talk about either (or both) lower energy levels and cell loss when mentioning this particular LRRK2 mutation – they are both part of the same thing. It is also why the kinase region of LRRK2 is a potential therapeutic target and the focus of a lot of research. Inhibitors of the kinase region have been shown to correct some of the dysfunction seen in cells with LRRK2 G2019S mutatins (for an example: https://www.nature.com/articles/ncomms9255 ; also see this review: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3369800/).
Does this clear up the confusion? I’m happy to have another crack at it.
Ok, lunch break over – back to work!
Kind regards,
Simon
LikeLike
Hello Simon,
Thank you very much for your reply, I understand it now. Thank you also for the links.
Kind regards,
Isobel
LikeLike
Hello Simon,
I was wondering how the discovery about the PARK6 gene might be put into practice? Would hospitals have to offer mass screenings to find people with a mutated copy of the gene who would be at risk, and put them on the treatment right away? Or would it be more economical to screen once symptoms begin to develop, to see if the patient would benefit from a Tigar inhibitor?
Would having a national genome bank be useful?
Thank you,
Isobel
LikeLike
Hi Isobel,
Thanks for your comment and interesting question. Genetic testing with a diagnosis of PD will probably (eventually) become a thing. As drugs become more personalised (for example, the Ambroxol trial in London UK is only accepting PD participants who have GBA mutations), it will be required. DNA sequencing technology is quickly becoming cheaper, which will make life easier for hospitals to handle the cost. I’m not sure that population wide screening would be advisable though. PD is not like Huntington’s disease (where if you have the genetic variation, you are 100% going to get the disease). Many people in the general population carry mutations associated with PD (such as GBA), but never actually develop the condition. Thus, early detection based on genetic screening is not likely to take off. We’ll need more definitive biomarkers for this idea.
Kind regards,
Simon
LikeLike