
|
In 2017, Parkinson’s UK – the largest charitable funder of Parkinson’s disease research in Europe – took a bold step forward in their efforts to find novel therapies. In addition to funding a wide range of small and large academic research projects and supporting clinical trials, they have also decided to set up ‘virtual biotech’ companies – providing focused efforts to develop new drugs for Parkinson’s, targeting very specific therapeutic areas. In today’s post we will look at the science behind their first virtual biotech company: Keapstone. |

A virtual world of bioscience. Source: Cast-Pharma
I have previously discussed the fantastic Parkinson’s-related research being conducted at Sheffield University (Click here to read that post). Particularly at the Sheffield Institute for Translational Neuroscience (SITraN) which was opened in 2010 by Her Majesty The Queen. It is the first European Institute purpose-built and dedicated to basic and clinical research into Motor Neuron Disease as well as other neurodegenerative disorders such as Parkinson’s and Alzheimer’s disease.
The research being conducted at the SITraN has given rise to multiple lines of research following up interesting drug candidates which are gradually being taken to the clinic for various conditions, including Parkinson’s.
It’s all very impressive.
And apparently I’m not the only one who thought it was impressive.
On the 7th March, the University of Sheffield and Parkinson’s UK launched a new £1 million virtual biotech company (Click here to read the press release). The company is called Keapstone Therapeutics.
Source: Parkinsonsvirtualbiotech
The Keapstone project is being led by Dr Richard Mead of the Sheffield Institute for Translational Neuroscience at Sheffield University.
Dr Richard Mead. Source: Sheffield
Sounds interesting. What do they mean by ‘virtual biotech’?
The virtual biotech approach means that there are no fixed premises, no staff, and no expensive bits of kit. Just a very focused approach to drug development.
Oooookay, but how do they get anything done with no staff???
The company will work in partnership with a range of other organisations that have the facilities and staff to carry out scientific work on a contract basis. This approach provides Keapstone with the ability to work flexibly and creatively with leading research organisations around the world to develop and commercialise new treatments for Parkinson’s. This will hopefully lower the cost of developing drugs and speed up the process.
Right, so how is it doing this?
Thus far the company has identified a range of compounds that can activate a particular biological pathway in cells that could be beneficial in Parkinson’s. They identified these compounds in the Innovative Medicines Initiative funded project that was conducted at the European Lead Factory.
European Lead Factory. Source: IMI
The European Lead Factory is a pan-European platform for drug discovery that is connecting innovative drug targets to high-quality compounds. The platform involves a large screening centre that can test a particular drug target against a collection of over half a million compounds. This short video explains the basic idea behind the concept:
Researchers in academia, small and medium-sized enterprises (SMEs) and patient organisations can submit a novel drug target to the European Lead Factory and if it is assessed they can receive a list of the known compounds that can bind to that drug target. This second video explains this idea:
Keapstone Therapeutics have completed this screening process and they are now funding a chemistry specialist company, called Sygnature Discovery, to further develop these molecules.
Source: Sygnaturediscovery
Sygnature Discovery, based out of Nottingham (UK) is a leading centre of excellence for drug discovery, offering significant expertise across a wide range of therapeutic areas and biological targets. They are now trying to improve upon the compounds that Keapstone Therapeutics identified in their screening.
How can they improve them?
Well, some of the compounds may not be able to enter the brain due to their inability to cross the blood-brain-barrier (a protective membrane that surrounds your brain). There are some small tricks of chemistry that groups like Sygnature Discovery can do on compounds that help them to cross this barrier, thus improving the potential use of the molecule.
The compounds may also not be stable inside the body – breaking down before they get to the brain. Again, chemistry can come to the rescue here as well.
Sygnature Discovery is attempting to improve some of these aspects of the Keapstone Therapeutics compounds, while not affecting their ability to perform the task of activating the biological pathway of interest.
And what is the biological pathway of interest?
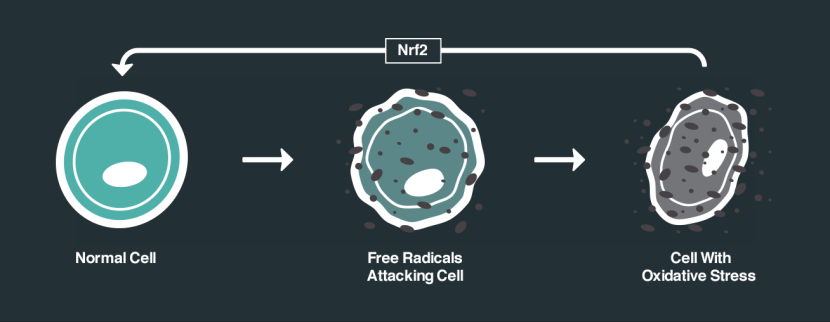
The Keapstone Therapeutics compounds have been selected for their ability to trigger a cellular defence system that helps protect brain cells from oxidative stress.
What is oxidative stress?
Oxidative stress is the damage done to a cell by the process of oxidation.
And yes, I know what you are going to ask next: What is oxidation?
Oxidation is the loss of electrons from a molecule, which in turn destabilises that particular molecule. Think of iron rusting. Rust is the oxidation of iron – in the presence of oxygen and water, iron molecules will lose electrons over time. Given enough time, this results in the complete break down of objects made of iron.

Rusting iron. Source: Thoughtco
The exact same thing happens in biology. Molecules in your body go through a similar process of oxidation – losing electrons and becoming unstable. This chemical reaction leads to the production of what we call free radicals, which can then go on to damage cells.
What is a free radical?
A free radical is an unstable molecule – unstable because they are missing electrons. They react quickly with other molecules, trying to capture the needed electron to re-gain stability. Free radicals will literally attack the nearest stable molecule, stealing an electron. This leads to the “attacked” molecule becoming a free radical itself, and thus a chain reaction is started. Inside a living cell this can cause terrible damage, ultimately killing the cell.
Antioxidants are the good guys in this situation. They are molecules that neutralise free radicals by donating one of their own electrons. The antioxidant don’t become free radicals by donating an electron because by their very nature they are stable with or without that extra electron.

How free radicals and antioxidants work. Source: h2miraclewater
How are free radicals generated in cells?
Free radicals can be generated in cells in different ways. One source of free radicals in cells are mitochondria.
I have previously spoken a lot about mitochondria and Parkinson’s on this website (for example – click here).
For the uninitiated, mitochondria are small bean-shaped structures that act as the power stations of each cell. They help to keep the lights on. Without them, the party is over and the cell dies.

Mitochondria and their location in the cell. Source: NCBI
They convert nutrients from food into Adenosine Triphosphate (or ATP). ATP is the fuel which cells run on. Given their critical role in energy supply, mitochondria are plentiful (some cells have thousands) and highly organised within the cell, being moved around to wherever they are needed.
The generation of ATP, however, causes the production of free radicals.
Oh!
But fear not. Across millions of years of hosting mitochondria, cells have developed numerous methods of reducing these free radicals that arise. There are specific biological pathways that become activated in the presence of free radicals and those processes reduce the levels of free radicals before they do to much damage.
So Keapstone Therapeutics compounds are activating biological pathways that reduce free radicals and oxidative stress?
Exactly.
Specifically, they are targeting a protein called Kelch-like ECH-associated protein 1 (or Keap1).
Under normal conditions, Keap1 acts as an anchor to another protein called nuclear factor erythroid-2-related factor 2 (Nrf2). Nrf2 is a major activator of antioxidant factors and I have discussed its activities in a previous post (Click here to read that post).
How Nrf2 works. Source: NRF2 science
Without any stress on a cell, Nrf2 is held in check by Keap1.
If oxidative stress suddenly appears, however, another protein called DJ-1 is activated and it grabs Keap1 and pulls it away from Nrf2 (Click here to read more on the Parkinson’s-associated protein DJ-1). This results in Nrf2 becoming free to do what it wants, and when Nrf2 is free there is only one thing it wants to do: head for the nucleus of the cell.
Inside the nucleus, Nrf2 activates various genes that have antioxidative properties, and by turning on these genes Nrf2 helps to decrease the level of oxidative stress in the cell. This antioxidant feature of Nrf2 has a lot of researchers excited and focused on finding drugs that help activate Nrf2.
How does NRF2 activate these genes?
NRF2 is a transcription factor.
What is a transcription factor?
A transcription factor is a protein that is involved in the process of converting (or transcribing) DNA into RNA.

An example of a transcriptional activator. Source: Khan Academy
Nrf2 is an activator of transcription.
When it binds to DNA to aids in the production of RNA, which then results in specific proteins being produced. And this is where NRF2 gets interesting.
You see, NRF2 binds to antioxidant response elements (ARE).
What are ARE?
Antioxidant response elements (ARE) are regions of DNA is commonly found beside genes that provide the instructions for various antioxidant and protective proteins. The ARE region of genes is the section of DNA where transcription is initiated for each gene. They are pieces of DNA that a transcription factor like Nrf2 binds to and activates the production (or transcription) of RNA.
Given that antioxidants and protective proteins are generally considered to be a good thing for sick/dying cells, you can see why Nrf2 is an interesting protein to investigate (Click here to read a previous post on this topic).
And you can also see why Keapstone Therapeutics are interested in compounds that block Keap1, which allows Nrf2 to be free to have its beneficial effects.
Now, if nothing I have written makes any sense, you can watch this video that Parkinson’s UK have kindly produced of Dr Mead explaining all of the various aspects of the Keapstone project:
Cool. Is there any evidence that activating this Nrf2 pathway helps in Parkinson’s?
There is a lot of pre-clinical research indicating that it can help.
One year ago, for example, researchers at the Gladstone Institute of Neurological Disease (San Francisco) published this study found that Nrf2 actually has some additional beneficial features that may be completely unrelated to the antioxidant properties:

Title: Nrf2 mitigates LRRK2- and α-synuclein-induced neurodegeneration by modulating proteostasis.
Authors: Skibinski G, Hwang V, Ando DM, Daub A, Lee AK, Ravisankar A, Modan S, Finucane MM, Shaby BA, Finkbeiner S.
Journal: Proc Natl Acad Sci U S A. 2016 Dec 27. pii: 201522872.
PMID: 28028237
The researchers wanted to determine what effect introducing high levels of Nrf2 into cell culture models of Parkinson’s would have on the behaviour and survival of the cells. There were two types of cell culture models of Parkinson’s disease used in the study: one produced a lot of the Parkinson’s associated protein alpha synuclein (the normal un-mutated version) and the other cell culture model involved two mutations in the LRRK2 gene (we have previously discussed LRRK2 – Click here and here to read those posts).
The researchers had previously demonstrated that both of these cell culture models of Parkinson’s disease exhibited increased levels of cell death when compared with normal cells. In the current study, when the researchers artificially exaggerated the amounts of Nrf2 in both sets of cell cultures, they found that not only did Nrf2 reduce LRRK2 and alpha synuclein toxicity in cell culture, but it also influenced alpha synuclein protein regulation, by increasing the degradation of the protein. This means that Nrf2 increased the disposal of the unnecessary excess of alpha synuclein – a property that could be beneficial in a condition like Parkinson’s where we see clustering and aggregation of alpha synuclein.
In addition, Nrf2 also promoted the collection of free-floating mutant LRRK2 and bundling it up into dense ‘inclusion bodies’ – dense clusters which are similar to the Lewy bodies of Parkinson’s disease but inclusion bodies are not associated with cell death. The scientists concluded that high levels of Nrf2 help to make the cells healthier and that this could represent a new candidate for future therapies of Parkinson’s.
Even better! So is it all going to work?
This is the big question. My fingers are most certainly crossed that it will.
Parkinson’s UK are hedging their bets though, by setting up additional virtual biotechs which we will hear more about early in the year. These other companies will focus on different biological pathways that are involved in Parkinson’s.
If nothing else, the approach is very innovative and exciting.
In addition, Dr Arthur Roach – Director of research at Parkinson’s UK – has spent 25 years in research, discovery and development for drug companies and universities – leading more than 100 drug discovery partnerships as Senior Director at Merck Serono.
Dr Arthur Roach. Source: Parkinson’s UK
He certainly has a great deal of experience with this kind of project, and he is playing a key role in the development of these virtual biotechs. Here is a video of Dr Roach explaining some of the ways that Parkinson’s UK is looking to fast track the development of new therapies for Parkinson’s:
With all of his experience, Dr Roach is also well aware of the nature of drug discovery and taking novel compounds to the clinic. He is very au fait with the risks of the drug development process and strategies for reducing those risks.
Risks? What risks?
The scary part of the whole equation is that 97% of drugs in preclinical tests never make it being approved for use in the clinic. They fail to pass through the 3 phases of clinical trials. To make matters worse, 95% of the drugs in phase 1 clinical trials and 88% of molecules in phase 2 are never approved. In other words there is a VERY high rate of failure in clinical trial process.
But by using the virtual biotech model of drug development, Parkinson’s UK can work with the best groups in each field – taking full advantage of their skills and knowledge – in an effort to reduce the overall risk and hopefully increase the chances of a drug candidate making it through to clinical approval.
So what does it all mean?
Summing up: Parkinson’s UK are trying something new.
The charitable organisation taking a very focused approach to the development of new therapies. By setting up virtual biotech companies that can target specific biological pathways and access the best people in the industry without the cost of hiring them or buying expensive equipment, this effort will hopefully stream line the drug development process.
It will be interesting to see how this endeavour plays out. All going well, it will provide not only novel therapeutics, but also a new way of drug development that other groups can replicate.
We watch and wait.
Editorial Note: This post highlights the activities of a biotech company. The folks at SoPD have no equity in the company, nor were we asked or paid by the company to write this post. We simply believe that what they are doing is really interesting (the science, the business model, the ultimate goal), and we thought we would make readers aware of them and their mission. SoPD initiated the production of this post, and we were very grateful to Dr Mead and Parkinson’s UK for providing us with answers some questions when we reached out to them.
The banner for today’s post was sourced from Parkinson’s UK







3 thoughts on “A virtual reality for Parkinson’s: Keapstone”