|
In American slang, to ‘nix‘ something is to ‘put an end to it’. Curiously, a protein called NIX may be about to help us put an end to Parkinson’s disease, at least in people with specific genetic mutations. In today’s post we will look at what NIX is, outline a new discovery about it, and discuss what this new information will mean for people living with Parkinson’s disease. |
Sydney harbour. Source: uk.Sydney
Before we start, I would like the reader to appreciate that I am putting trans-Tasman rivalry side here to acknowledge some really interesting research that is being conducted in Australia at the moment.
And this is really interesting.
I have previously spoken a lot about mitochondria and Parkinson’s on this website. For the uninitiated, mitochondria are the power house of each cell. They help to keep the lights on. Without them, the party is over and the cell dies.

Mitochondria and their location in the cell. Source: NCBI
You may remember from high school biology class that mitochondria are tiny bean-shaped objects within the cell. They convert nutrients from food into Adenosine Triphosphate (or ATP). ATP is the fuel which cells run on. Given their critical role in energy supply, mitochondria are plentiful (some cells have thousands) and highly organised within the cell, being moved around to wherever they are needed.
Like you and I and all other things in life, however, mitochondria have a use-by date.
As mitochondria get old and worn out (or damaged) with time, the cell will recycle them via a process called mitophagy (a blending of the words mitochondria and autophagy – the waste disposal system of each cell).
What does this have to do with Parkinson’s disease?
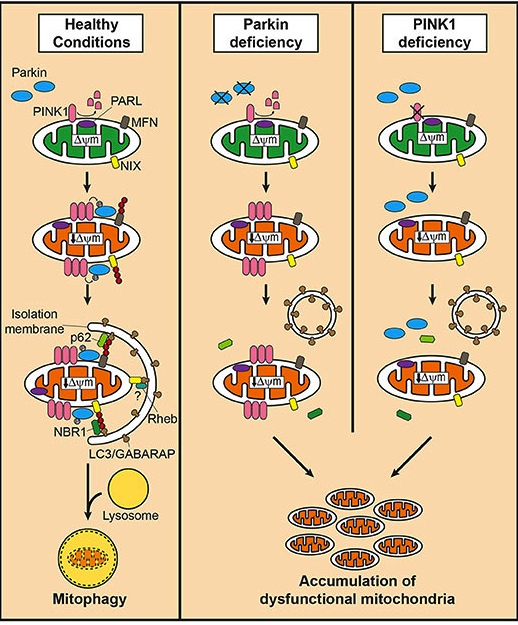
Well, about 10% of Parkinson’s cases are associated with particular genetic variations that render people vulnerable to developing the condition. Some of these mutations are in sections of DNA (called genes) that provide the instructions for proteins that are involved in the process of mitophagy. Two genes, in particular, are the focus of a lot of Parkinson’s-related research – they are called PARKIN and PINK1.
What do PARKIN and PINK1 do?
Both proteins appear to have many different functions, but their roles in the process of mitophagy are well understood.
PINK1 acts like a kind of handle on the surface of mitochondria. In normal, healthy cells, the PINK1 protein attaches to the surface of mitochondria and it is slowly absorbed until it completely disappears from the surface and is degraded. In unhealthy cells, however, this process is inhibited and PINK1 starts to accumulate on the outer surface of the mitochondria. Lots of handles poking out of the surface of the mitochondria.
Now, if PINK1 a handle, then PARKIN is a flag that likes to hold onto the PINK1 handle. While exposed on the surface of mitochondria PINK1 starts grabbing the PARKIN protein. This pairing is a signal to the cell that this particular mitochondrion (singular) is not healthy and needs to be removed.

Pink1 and Parkin in normal (right) and unhealthy (left) situations. Source: Hindawi
In the absence of normal PINK1 or PARKIN proteins, there is no handle-flag system and sick/damaged mitochondria start to pile up. They are not disposed of appropriately and as a result the cell getting sick and ultimately dying.
Mitophagy. Source: Frontiersin
People with particular mutations in the PINK1 or PARKIN genes are vulnerable to developing an early onset form of Parkinson’s disease. It is believed that the dysfunctional disposal of (and accumulation of) old mitochondria are part of the reason why these individuals develop the condition at such an early age.
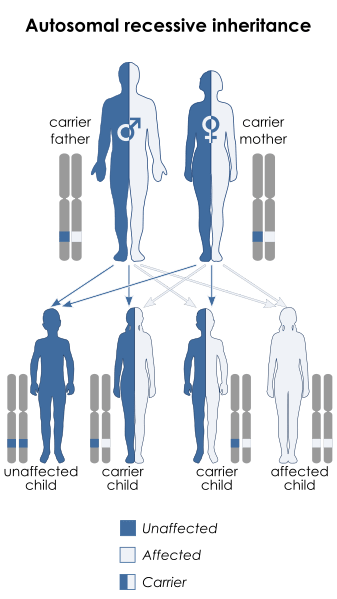
The versions of Parkinson’s associated with these two proteins involve an autosomal recessive mutation – meaning that a copy of the mutation has to be provided by both the parents in order for a condition to develop.
Autosomal recessive genetic transfer. Source: Wikipedia
For a very good review of the genetics of Parkinson’s disease – click here. Alternatively, have a look at our Genetics of Parkinson’s page.
So someone with a copy of a PARKIN or PINK1 mutation from both parents will go on to develop Parkinson’s?
Well, no.
As with all things in life, it’s more complicated than that.
And this is where the Aussies come into the picture.
This is Professor Caroline Su:
Professor Caroline Su. Source: DailyTelegraph
She is Professor of Medicine in the Kolling Institute of Medical Research at the University of Sydney.
And several years ago, she and her colleagues were confronted with a curious situation. They discussed it in this research article:
Title: Phenotypic variability of parkin mutations in single kindred.
Authors: Koentjoro B, Park JS, Ha AD, Sue CM.
Journal: Mov Disord. 2012 Sep 1;27(10):1299-303.
PMID: 22807239
The researchers were presented in the clinic with a young woman who had developed early-onset Parkinson’s. Further investigations found that the individual’s father had a single copy of the PARKIN gene mutated (remember that we have two copies of each gene) and this resulted in reduced levels of PARKIN. He exhibited a mild loss of arm swing, but nothing severe enough to warrant a diagnosis of Parkinson’s. The individual’s mother was also tested, and the results of her genetic test surprised the investigators: she had two copies of a PARKIN mutation.
The mother, who is now in her eighth decade of life, has only very mild rigidity. The investigators were left wondering the obvious question: ‘why does the mother not have early-onset Parkinson’s, while her daughter with the same genetic mutation does have the condition?’
Good mystery, huh?
But wait, there’s more.
And this story gets even stranger – PARKIN is involved with the recycling of a protein called Mitofusin 2. Disposal of this protein was impaired in both the mother and the daughter, suggesting that PARKIN activity was disrupted by the genetic mutation.
So AGAIN, why is the mother NOT exhibiting the features of Parkinson’s???
I hope you are going to tell us. Do we know the reason?
Dr Su and her colleagues have investigated this case study further and what they have found is fascinating.
They have recently published their findings in this research report:

Title: Nix restores mitophagy and mitochondrial function to protect against PINK1/Parkin-relatedParkinson’s disease.
Authors: Koentjoro B, Park JS, Sue CM.
Journal: Sci Rep. 2017 Mar 10;7:44373.
PMID: 28281653 (This article is OPEN ACCESS if you would like to read it)
The researchers took skin cells for the mother and the daughter, and they compared mitochondrial function in both with that of normal skin cells from healthy, un-affected control individuals. While the daughter had lower levels of ATP production (suggesting mitochondrial dysfunction), ATP production was normal in skin cells derived from the mother. In addition, the skin cells from the mother were more resistant to stress (similar to the control cells) than the daughter’s skins. These results suggested that mitochondrial function in the mother’s cells was relatively normal compared to the daughter’s cells which have the same Parkin deficiency.
Next the researchers exposured all of the cells to a drug called ‘carbonyl cyanide m-chlorophenyl hydrazone’ (CCCP). CCCP causes cells to initiate mitophagy, which occurred in both the control cells and the mother’s cells, but not in the daughter’s cells. This result indicated that PARKIN-independent mitophagy was occurring in the mother’s cells. The researchers also removed PINK1 protein (the handle on the surface of mitochondria) from the mother’s cells and mitophagy still occurred. So apparently in the mother’s cells, neither PARKIN or PINK1 were required for mitophagy to occur.
Que? How is this possible?
Because. Biology is complicated.
The reason folks with PARKIN or PINK1 don’t fall apart at a much younger age is because the PARKIN-PINK1 tag-team effort is not the only pathway to mitophagy.
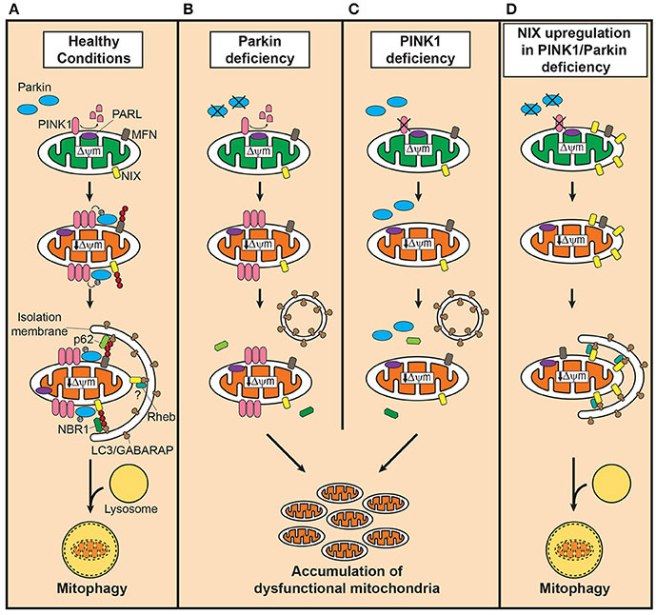
There are at least three well characterised pathways that we are aware of. The first is the PINK1-PARKIN pathway which leads to the generation of the autophagosome (the sack that will surround the mitochondria and aid in its destruction – the blue crescent in the image below). The second pathway involve the pairing of two proteins called BNIP3-NIX pathway, while the third pathway functions by the activation of a protein called FUNDC1.
The pathways to mitophagy. Source: AJPheart
Prof Su and her colleagues found that the skin cells from the mother had very high levels of NIX protein compared to the control and daughter’s skin cells. And when they removed NIX protein from the mother’s skin cells, the researcher found that no mitophagy occurred. This dependence on NIX protein in the mother’s skin cells made the investigators question whether increasing levels of NIX in cells from other PARKIN mutation affected individuals could rescue mitophagy in those cells. So they collected skin cells from other individuals affected by PARKIN mutations and they caused the over-production of NIX protein in those cells (using a specially engineered virus). The over-production of NIX restored mitophagy in the cells from folks with PARKIN mutations AND it significantly increased the mitochondrial ATP synthesis rate in the cells as well.
And here is the really amazing part of this result: The over-production of NIX protein also rescued mitophagy and ATP production in cells from people with PINK1 mutations.
As a final experiment in the study, Prof Su and her colleagues wanted to see if they could pharmacological induce NIX production using a drug. Phorbol 12-myristate 13-acetate (PMA) is a chemical that has been shown to increase NIX production and when the researchers exposed their cells from people with PARKIN mutations, they found that PMA encouraged the production of NIX and mitophagy increased. This trick also worked in cells from people with PINK1 mutations and it disappeared when the researchers reduced levels of NIX and treated the cells with PMA. Thus, the researchers confidently concluded that NIX represents a potential therapeutic target for Parkinson’s disease.
A route around the PARKIN-PINK1 problem. Source: Frontiersin
Prof Su and her colleagues have subsequently written up a nice, OPEN-ACCESS review of this research that you can read by Clicking here.
WOW! I’m going to rush out and get me some of that PMA drug stuff
Mmm, yeah, before you do that – there is a rather significant health warning that comes with Phorbol 12-myristate 13-acetate (or PMA).
PMA. Source: Wikipedia
Also known as 12-O-Tetradecanoylphorbol-13-acetate, PMA is a potent tumor promoter – that is, it encourages cancerous tumors to grow. On top of that it has some terrible side-effects. In one clinical study, that was investigating the use of this drug for relapsed/refractory malignancies, serious adverse side effects associated with the drug included individual episodes of gross hematuria (blood in the urine), a grand mal seizure, fainting, and hypotension (Source).
PMA is a dangerous drug and it should not be experimented with under any circumstances.
Efforts are being made, however, to find NIX activator drugs that do not exhibit these side effects. I will keep an eye out for them and report the research as it comes to hand.
What does it all mean?
So summing up – Aussie researchers have identified a new method of activating the waste recycling system in cells. This finding would be particularly useful for people with Parkinson’s disease-related genetic mutations in genes like PARKIN and PINK1, which are instrumental in the process of mitophagy.
The research being conducted in Sydney has highlighted an interesting case study of a person who is seemingly unaffected by a PARKIN mutation, while her relatives are directly impacted by the genetic variant with the development of early onset Parkinson’s disease. By analysing skin cells from this family, Prof Su and her colleagues have uncovered the unique role that NIX is playing in seemingly protecting an individual from Parkinson’s, and they are now following up this research by looking for activators of this gene.
EDITORIAL NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from nigms




7 thoughts on “NIX-ing the PARKIN and PINK1 problem”