|
In a recent post, I discussed research looking at foods that can influence the progression of Parkinson’s (see that post here). I am regularly asked about the topic of food and will endeavour to highlight more research along this line in future post. In accordance with that statement, today we are going to discuss Cruciferous vegetables, and why we need a clinical trial of broccoli. I’m not kidding. There is growing research that a key component of broccoli and other cruciferous vegetables – called Glucoraphanin – could have beneficial effects on Parkinson’s disease. In today’s post, we will discuss what Glucoraphanin is, look at the research that has been conducted and consider why a clinical trial of broccoli would be a good thing for Parkinson’s disease. |
Cruciferous vegetables. Source: Diagnosisdiet
Like most kids, when I was young I hated broccoli.
Man, I hated it. With such a passion!
Usually they were boiled or steamed to the point at which they have little or no nutritional value, and they largely became mush upon contact with my fork.
The stuff of my childhood nightmares. Source: Modernpaleo
As I have matured (my wife might debate that statement), my opinion has changed and I have come to appreciate broccoli. Our relationship has definitely improved.
In fact, I have developed a deep appreciation for all cruciferous vegetables.
And yeah, I know what you are going to ask:
What are cruciferous vegetables?
Cruciferous vegetables are vegetables of the Brassicaceae family (also called Cruciferae). They are a family of flowering plants commonly known as the mustards, the crucifers, or simply the cabbage family. They include cauliflower, cabbage, garden cress, bok choy, broccoli, brussels sprouts and similar green leaf vegetables.
Cruciferous vegetables. Source: Thetherapyshare
So what have Cruciferous vegetables got to do with Parkinson’s?
Well, it’s not the vegetables as such that are important. Rather, it is a particular chemical that this family of plants share – called Glucoraphanin – that is key.
What is Glucoraphanin?
Glucoraphanin is a glucosinolate.
Glucosinolates are a naturally occurring class of organic compounds that contain both sulfur and nitrogen. They are derived from glucose and an amino acid. You know that pungent smell that fills the kitchen when you are cooking cabbage or brussels sprouts? This is partly due to the release of glucosinolates from the vegetables being cooked.
Cruciferous plants release glucosinolates when they are attacked or damaged (or steamed/boiled). These glucosinolates are rapidly released when the plant tissue senses danger: freezing or boiling cruciferous plants for 10 minutes reduces glucosinolate concentrations by about 50%. Glucosinolates are believed to be part of a plant’s defence system against pests and diseases.
And being a glucosinolate, Glucoraphanin is one component of this defence system.
The structure of Glucoraphanin. Source: Wikipedia
When cruciferous vegetables are consumed, an enzyme called myrosinase transforms glucoraphanin into raphanin and sulforaphane. Raphanin is useful in that it inhibits activity of viruses, some fungi and various bacteria. A general anti-bacterial/viral agent.
And what about sulforaphane?
Sulforaphane is what I would like to focus on in this post, because it is something of a miracle worker.
Above all else, it is a potent activator of the NRF2 pathway.
What is NRF2?
Nuclear Factor Erythroid 2-Related Factor 2 (or NRF2) is a ‘transcription factor’, which is a protein that is involved in the process of converting DNA into RNA (an event called transcription) .
You may remember from your high school science class one of the central dogmas of biology: DNA gives rise to RNA, RNA gives rise to protein. Transcription is the process of making RNA.

The basic of biology. Source: Youtube
Now, a transcription factor can be an ‘activator’ of transcription – that is, it initiates or helps the process of generating RNA from DNA.

An example of a transciptional activator. Source: Khan Academy
Or it can be a repressor of transcription – blocking the machinery (required for generating RNA) from doing its work.

An example of a transciptional repressor. Source: Khan Academy
NRF2 is an activator of transcription. When it binds to DNA to aids in the production of RNA, which then results in specific proteins being produced.
And this is where NRF2 gets REALLY interesting.
You see, NRF2 binds to antioxidant response elements (ARE).
What are ARE?
Antioxidant response elements (ARE) are regions of DNA commonly found in the regulatory area of genes encoding powerful antioxidant and protective proteins (if you are wondering what antioxidants are – click here).
The regulatory region of a gene is the section of DNA where transcription is initiated. They are pieces of DNA that a transcription factor – like NRF2 – attach to and activates the production of RNA.
Given that these antioxidants and protective genes are generally considered a good thing for sick/dying cells (and that NRF2 is known to activate more than 200 of them), you can see why NRF2 is an interesting protein to investigate.
Source: NRF2 science
By binding to ARE, NRF2 is directly encouraging the production of naturally occurring antioxidant and protective proteins. And this is why a lot of people are excited about NRF2.
It is also why I am interested in sulforaphane: it is a naturally-occurring, potent activator of NRF2.
And broccoli have high levels of sulforaphane?
No, broccoli have high levels of glucoraphanin which is converted into sulforaphane (see above). Broccoli has approximately 44-170 mg of glucoraphanin for every 100g of dry weight.
Even more impressive are Broccoli sprouts, which have 1100 mg of glucoraphanin for every 100g of dry weight (but I’d still like to focus on a broccoli clinical trial because broccoli sprouts are associated with a great deal of childhood dinner-time trauma! I simply hate them. Still to this day. With a very deep passion).
Brocolli sprouts. Source: Organiclifestylemagazine
For a very good explanation of glucoraphanin/sulforaphane I can recommend that you watch this video:
Interesting, but what Parkinson’s-related research has been done on glucoraphanin/sulforaphane?
Most of the research that has been conducted has focused on sulforaphane. Firstly, there were cell culture experiments, such as this one:
Title: Protective effect of sulforaphane against dopaminergic cell death
Authors: Han JM, Lee YJ, Lee SY, Kim EM, Moon Y, Kim HW, Hwang O
Journal: J Pharmacol Exp Ther. 2007 Apr;321(1):249-56.
PMID: 17259450 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in investigating the potential neuroprotective effects of Sulforaphane. They had previously found that exposing dopamine neurons to a protein called quinone reductase (which functions as an enzyme that reduces levels of quinones – an oxidative species) helped protect the cells from oxidative stress. By reducing quinones in the cells there was less oxidative stress.
Now, sulforaphane is a potent activator of quinone reductase in various non-neuronal cells, so the investigators decided to see if sulforaphane had the same effect in dopamine neurons. And they discovered that it did. Sulforaphane protected the dopamine neurons against neurotoxins (such as 6-hydroxydopamine) which are known to induce quinone production.
And these cell culture results have been replicated in other studies which have highlighted other activities of sulforaphane (Click here and here to see examples).
Subsequent research on sulforaphane has been conducted in animal models of Parkinson’s disease. Studies such as this one:
Title: Neuroprotective effect of sulforaphane in 6-hydroxydopamine-lesioned mouse model of Parkinson’s disease.
Authors: Morroni F, Tarozzi A, Sita G, Bolondi C, Zolezzi Moraga JM, Cantelli-Forti G, Hrelia P.
Journal: Neurotoxicology. 2013 May;36:63-71.
PMID: 23518299
In this study, the investigators modelled Parkinson’s disease in mice using a neurotoxin (6-OHDA). One hour after the neurotoxin was administered, the researchers started treating the mice with sulforaphane (5mg/kg), and they continued to give this treatment to the mice twice per week for four weeks. This regime was found to reduce the behaviour and motor problems associated with this model and it also rescued the dopamine cells from dying.
Interestingly, this neuroprotective effect may have been due to an increase in glutathione levels. I have previously written about glutathione (Click here to read that post), which is a potent anti-oxidant. In this particular study, while sulforaphane had no effect on glutathione levels in normal mice, it produced a significant increase in the disease model animals (see graph below).
Sulforaphane (SFN) increases glutathione (GSN) levels. Source: Sciencedirect
The researchers concluded their report suggesting that sulforaphane “may potentially be effective in slowing down the progression of idiopathic Parkinson’s disease by the modulation of oxidative stress and apoptotic machinery”. And subsequent research in other animal models of Parkinson’s disease appears to support this idea:
Title: Sulforaphane protects against rotenone-induced neurotoxicity in vivo: Involvement of the mTOR, Nrf2, and autophagy pathways.
Authors: Zhou Q, Chen B, Wang X, Wu L, Yang Y, Cheng X, Hu Z, Cai X, Yang J, Sun X, Lu W, Yan H, Chen J, Ye J, Shen J, Cao P.
Journal: Sci Rep. 2016 Aug 24;6:32206.
PMID: 27553905 (This article is OPEN ACCESS if you would like to read it)
The investigators who conducted this study gave mice a neurotoxin (rotenone) which is used to model Parkinson’s. Some of these mice received sulforaphane at the same time while other were treated with a neutral placebo solution. The neurotoxin caused locomotor problems and dopamine neuron cell loss in the animals that did not receive sulforaphane. In mice that did receive sulforaphane, however, the researchers observed a near complete rescue. Additionally, sulforaphane treatment inhibited oxidative stress and resulted in increased levels of glutathione.
Sulforaphane treatment increased the levels of NRF2 and this activation by sulforaphane appears to be quite direct as it has been reported that sulforaphane has no effect on mice that have been genetically engineered to not produce NRF2 protein (Click here to read more about this).
Sulforaphane treatment in this study was also associated with significant inhibition of cell death (‘apoptosis’) pathways and restoration of the cellular waste disposal system (‘autophagy’). And given these impressive results, the researchers concluded that “Sulforaphane appears to be a promising compound with neuroprotective properties that may play an important role in preventing Parkinson’s disease”.
Before we continue, it is important to note that in all of these studies, sulforaphane treatment was started before or at the same time as Parkinson’s was modelled. A detail we are missing here is whether delaying sulforaphane treatment (which would be more disease-relevant) can also have beneficial effects. As far as I’m aware, this study has yet to be done (please correct me if I am wrong).
Has Sulforaphane been tested in the clinic?
Yes, but not for Parkinson’s disease (as far as I’m aware).
There are 53 clinic trials involving sulforaphane registered on the U.S. National Library of Medicine‘s website for the Clinical trials database. In addition, there are 99 clinical studies for broccoli (I kid you not!) registered on that website (Source).
The results of these clinical trials have been mixed, which may explain why there haven’t been any “Broccoli saves the world” headlines.
But there could also be a very good reason for this mixed outcome.
In 2007, this research report was published:
Title: GSTT1 genotype modifies the association between cruciferous vegetable intake and the risk of myocardial infarction.
Authors: Cornelis MC, El-Sohemy A, Campos H.
Journal: Am J Clin Nutr. 2007 Sep;86(3):752-8.
PMID: 17823442 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers noticed that the consumption of cruciferous vegetables was associated with a lower risk of a having a heart attack, but this association was only present among people with a normal fully functional glutathione S-transferases gene.
This gene gives rise to an enzyme that works alongside glutathione in efforts to reduce oxidative stress in cells. The glutathione S-transferases enzyme acts as a transport protein, binding to toxins and inactivating oxidative agents. In this study, the researchers found that people with both copies of the glutathione S-transferases gene (we have two copies of every gene) and who consumed cruciferous vegetables had less heart attacks than those with only one copy of the glutathione S-transferases gene. Taken another way, these individuals had all the cellular machinery to better use the chemicals in cruciferous vegetables to reduce their risk of having a heart attack.
Sulforaphane is known to activate glutathione S-transferase (Click here to read more about this).
Thus, the mixed clinical trial results for Sulforaphane (and broccoli) could be partly explained by genetic variations in genes involved in the response to oxidative stress. And these types of genetic variations are very common, with between 30-50% of the population having glutathione S-transferase variants (Click here to read more about this). If half the people enrolled in all of the broccoli clinical trials are not going to respond to the treatment, it will result in mixed results.
Are glutathione S-transferase genetic variations associated with Parkinson’s disease?
This a good question, and there does appear to be some kind of association:

Title: Null genotype of GSTT1 contributes to increased Parkinson’s disease risk in Caucasians: evidence from a meta-analysis.
Authors: Wang D, Zhai JX, Zhang LM, Liu DW.
Journal: Mol Biol Rep. 2014 Nov;41(11):7423-30.
PMID: 25086621
The researchers who conducted this study looked at 18 studies involving genetic information from 3,963 people with Parkinson’s disease and 5,472 healthy controls. They found a significant association between Parkinson’s and people with genetic variants in both copies of their glutathione S-transferase gene. People with errors in both copies of their glutathione S-transferase gene were 24% more likely to have Parkinson’s. Curiously, this association was only observed among Caucasian individuals and not found among Asians and Latinos.
One note of caution regarding this result: the same researchers published another research report in 2016 which questioned their initial results (Click here to read more about this). Independent replication of the research is required.
But genetic variations in other genes involved in these oxidative response pathways have also been associated with Parkinson’s (Click here and here to read about more examples). Different combinations of these variations may explain some of the susceptibility within the Parkinson’s affected population.
How does this affect your clinical trial of Parkinson’s and broccoli?
It would mean that the people being recruited for my hypothetical clinical trial would need to have their DNA sequenced before they can be part of the study. In order to determine if broccoli (or Sulforaphane) can influence Parkinson’s disease, we would need to recruit individuals to the study who have few genetic variations in their glutathione S-transferase gene. This would give broccoli (or Sulforaphane) the best chance to demonstrate any efficacy.
And this is an important part of the great shift towards more individualised treatment in Parkinson’s.
Individualised medicine. Source: Medscape
Future therapies will be oriented to each person on an individualised basis, based on what cellular machinery that they each have. Some people with variations in their glutathione S-transferase gene may have no beneficial response to broccoli, so we would focus their treatment regime on other biological pathways.
This shift won’t stop the great Broccoli clinical trial. It will simply target this vegetable towards the individuals who will potentially benefit the most.
What does it all mean?
So, summing up: I am often asked “Given everything you have read, what would you do if you were diagnosed with Parkinson’s?”. There is an ethical dilemma in answering it, but it is an interesting question, one which I often consider. Regardless, my first response to the question is always the same: “I would learn as much as I can about myself”. Everything: from am I glucose intolerant to what are my sleep patterns like; from what genetic variants do I have to what allergies affect me; from the variety of bacteria in my gut to a review of my environmental history (I grew up in the country side). All of this information would form the basis of any treatment I would consider trying in an attempt to slow down the course of the condition. For example, if I have a variation in my glutathione S-transferase gene, I would (happily) forgo delightful pleasures of broccoli sprouts and focus on alternative remedies.
Having said that, the beneficial effects of eating cruciferous vegetables are many and not simply limited to activation of NRF2 pathways. Therefore, it would be prudent for me to encourage readers of all situations to reconsider their childhood hatred of certain vegetables (I also remember not being a big fan of cabbage), and perhaps making this food group a larger part of their diet.
And, having said that, I have to acknowledge that such a change in diet would be an ‘exclusionary’ from the big clinical trial of broccoli for Parkinson’s disease (meaning that you would not be recruited to the trial if you had a past diet high in broccoli), so we better get the broccoli (or sulforaphane) trial underway immediately before too many people jump on the cruciferous vegetables band wagon.
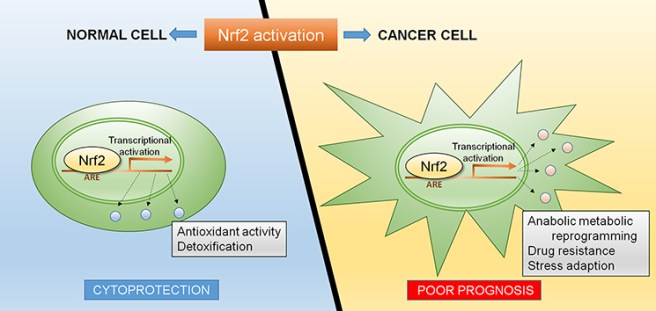
One final cautionary note before we finish: Despite all the excitement surrounding NRF2, it is important to remember that too much of a good thing can be bad.
Continuous activation of NRF2 can be pro-tumourigenic – that is, it can encourage the growth of cancerous tumours. Cancer cells generate a lot of oxidative stress, which usually helps to kill off the cancer or at least keep the growth under control. Sometimes somatic (or spontaneous) genetic mutations can occur in the NRF2 gene which results in hyperactivity of NRF2 and it continuously activates target genes. These target genes help to reduce the oxidative stress in the cancer cells, which in turns helps to keep those cells alive and multiplying (Click here to read more). Thus, one must be careful with over activating NRF2.
NRF2 activation is not always a good idea. Source: Intechopen
This is a further example of learning as much as you can about yourself in handling a disease like Parkinson’s. It would be wise to ask your doctor for their opinion/advice before considering any change to a treatment regime and asking for a full check up before undertaking an effort to increase levels of NRF2 (many cancerous biomarkers can be picked up with a simple blood test).
EDITORIAL NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. Please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Cookforyourlife













Thanks. I’ll eat more broccoli but not too much. Not sure my doc will test for variance in my genes. Maybe worth asking. Glucoraphanin the new wonder drug available without a prescription.
LikeLike
Hi Dkdc,
Yeah, the checking for genetic variants is easier said than done. But it would be wise as DNA sequencing becomes cheaper to offer this service to individuals in disease affected communities. I often wonder how many people are taking supplements that their bodies have trouble processing simply because they don’t have the right biological gear for the task. The situation could potentially get toxic – for example, my Dad nearly gave himself a heart attack by eating too many bananas (2-3 per day for him = potassium poisoning!). Destroyed his (and the family’s) faith in healthy food!
Glad you liked the post.
Simon
LikeLike
“The interesting fact about kale and other members of the Brassica family (including broccoli, green cabbage, collards, brussel sprouts and rape seed) is that they appear accumulate the heavy metal thallium from the soil[1-3], which is poisonous.” http://body.io/killer-kale-toxic-organic-farming/
LikeLike
Hi Lee,
Thanks for the interesting link.
I think to be fair, while the brassica family have been reported to be good accumulators of thalium, it is important to remember that thalium is everywhere in the environment, especially around regions of coal production and metal smeltering (this review provides an interesting overview of the research – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5080298/). Yes, it is important to be aware of these things and to check the source of one’s vegetables, but I worry about the implications of scaring folks with these sorts of articles – I note that all three of the sources for the statement come from the same research group in Brno, Czech Republic. Dr Zbiral, one of analytical chemists in Brno who conducted that research, has stated that it’s very unlikely the plants would suck up enough to poison humans. Zbiral also says that canola can be just as good as kale at absorbing thallium – which begs the question should the world stop eating that staple? I don’t think so. Being careful about the soil from which the vegetables are sourced is important, but I think we have to be careful with interpreting data from isolated studies and then presenting it to the general public as fact.
Thanks again for the comment.
Kind regards,
Simon
LikeLike
Thaank you for writing this
LikeLike