|
A new research report has been published this week which may point not only towards a new understanding of the biology of Parkinson’s, but also to potentially novel therapies which are clinically available. These exciting new findings involve a DNA repair mechanism called ‘poly ADP ribose polymerase’ (or simply PARP) and a process of cell death called Parthanatos. Biotech companies have developed PARP inhibitors which have been reported to rescue models of Parkinson’s. With a bit of tweaking, this class of drugs could potentially be re-purposed for Parkinson’s. In today’s post, we will look at what PARP is, explain how PARP inhibitors work, review what previous PD research has been conducted on this topic, evaluate the new report, and consider what it means for the Parkinson’s community (Spoiler alert: this will be a long post!).
|

Source: Quotefancy
Ah, the good old days!
Remember them. Way back before Netflix. When life was sooo much easier.
You know what I’m talking about.
Back when biology was simple. Remember when DNA gave rise to RNA and RNA gave rise to protein, and that was it. Simpler times they were. Now, everything is so much more complicated. We have all manner of ‘regulatory RNA’, epigentics, splice variants, and let’s not get started on the labyrinthian world of protein folding.
Oh, how I long for the good old days.
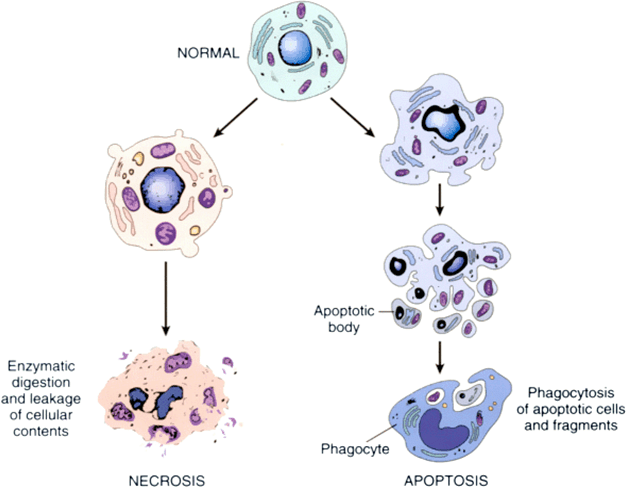
Back when a cell could only die one of two ways: apoptosis (a carefully controlled programmed manner of death) and necrosis (cell death by injury):
Source: Researchgate
Now life is too complicated and complex beyond reason or imagination.
Let’s just take the example of cell death that I mentioned above: over the past decade, the Nomenclature Committee on Cell Death (or NCCD – I kid you not there is actually a committee for this) has written up guidelines for the definition/interpretation of ‘cell death’. And as part of that effort they have decided that there are now at least 12 (yes, 12) different ways a cell can die:
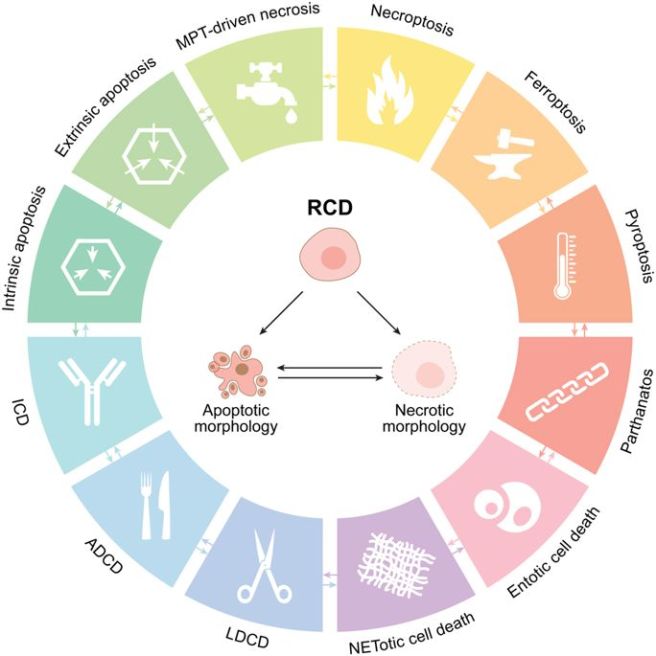 Source: Nature
Source: Nature
For those of who are interested in reading more about all of these different kinds of cell death, click here to read NCCD committee’s most recent recommendations which were updated this year (2018). Some riveting betime reading.
Which form of cell death applies to Parkinson’s?
Now that’s a really good question!
One that has been studied and the source of debate for a very long time.
To be fair, we don’t really know. But fascinating new research published this week suggests that the Parthanatos pathway could be involved in the cell death associated with Parkinson’s.
What is Parthanatos?
Continue reading “We’re re-branding: It’s now called PARPinson’s”