|
The title of this post probably reads like the mad, drug-fuelled scream of a drunk Saturday night party animal, but the elements of it may be VERY important for a particular kind of Parkinson’s disease. Mutations in a gene called DJ-1 can cause an early onset form of Parkinson’s disease. The protein of DJ-1 plays an important role in how cells handle oxidative stress – or the increase in damaging free radicals (explained below). This week researchers announced that they have found an interesting new therapeutic target for people with DJ-1 associated Parkinson’s disease: A chemical called Isocitrate. In this post, we will discuss what DJ-1 is involved with Parkinson’s disease, how isocitrate helps the situation, and what the results of new research mean for future therapeutic strategies. |

Source: Listchallenge
In 2017, we are not only observing the 200 year anniversary of the first description of Parkinson’s disease (by one Mr James Parkinson), but also the 20th anniversary of the discovery of the first genetic variation associated with the condition (Click here to read more about that). Our understanding of the genetics of Parkinson’s disease since 1997, has revolutionised the way we look at Parkinson’s disease and opened new doors that have aided us in our understanding.
During the last 20 years, we have identified numerous sections of DNA (these regions are called genes) where small errors in the genetic coding (mutations or variants) can result in an increased risk of developing Parkinson’s disease. As the graph below indicates, mutations in some of these genes are very rare, but infer a very high risk, while others are quite common but have a low risk of Parkinson’s disease.
The genetics of PD. Source: Journal of Parkinson’s disease
Some of the genetic mutation need to be provided by both the parents for Parkinson’s to develop (an ‘autosomal recessive‘ mutation – the yellow circles in the graph above); while in other cases the genetic variant needs only to be provided by one of the parents (an ‘autosomal dominant’ mutation – the blue circles). Many of the genetic mutations are very common and simply considered a region of increased risk (green circles).
Importantly, all of these genes provide the instructions for making a protein – which are the functional parts in a cell. And each of these proteins have specific roles in biological processes. These functions tell us a little bit about how Parkinson’s disease may be working. Each of them is a piece of the jigsaw puzzle that we are trying to finish. As you can see in the image below, many of the genes mentioned in the graph above give rise to proteins that are involved in different parts of the process of autophagy – or the waste disposal system of the cell. You may notice that some proteins, like SCNA (otherwise known as alpha synuclein), are involved in multiple steps in this process.
The process of autophagy. Source: Nature
In today’s post we are going to look at new research regarding just one of these genes/proteins. It is called DJ-1, also known as Parkinson disease protein 7 (or PARK7).
What is DJ-1?
In 2003, this research report was published regarding DJ-1:

Title: Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism
Authors: Bonifati V, Rizzu P, van Baren MJ, Schaap O, Breedveld GJ, Krieger E, Dekker MC, Squitieri F, Ibanez P, Joosse M, van Dongen JW, Vanacore N, van Swieten JC, Brice A, Meco G, van Duijn CM, Oostra BA, Heutink P.
Journal: Science. 2003 Jan 10;299(5604):256-9.
PMID: 12446870
The researchers had been presented with two families from genetically isolated communities (in the Netherlands and Italy) that had members displaying an early-onset form of Parkinson’s disease. They had previously identified the region of DNA on chromosome 1 of DNA that they called PARK7 (Click here to read that report), and they had also validated that result on an independent database of DNA from people with Parkinson’s disease (Click here to read more about that study). But it was not until 2003 that they identified the specific gene within the PARK7 region on chromosome 1. And that gene was DJ-1.
The researchers immediately started looking for more examples young-onset Parkinson’s disease to see how common the DJ-1 mutation was. And that effort resulted in this research report which was published later that same year:
Title: Early-onset Parkinson’s disease caused by a compound heterozygous DJ-1 mutation.
Authors: Hague S, Rogaeva E, Hernandez D, Gulick C, Singleton A, Hanson M, Johnson J, Weiser R, Gallardo M, Ravina B, Gwinn-Hardy K, Crawley A, St George-Hyslop PH, Lang AE, Heutink P, Bonifati V, Hardy J, Singleton A.
Journal: Ann Neurol. 2003 Aug;54(2):271-4.
PMID: 12891685
The investigators collected DNA from 107 early-onset (that is age at diagnosis being below 50 years of age) Parkinson’s disease and conducted DNA sequencing on the entire DJ-1 gene. They identified several different mutations in the DJ-1 gene that were shared between independent subjects in their 107 subjects.
With subsequent research, investigators have identified more than 25 mutations within the PARK7 region of DNA that can cause early-onset form of Parkinson’s disease. Some PARK7 gene mutations lead to an abnormally small DJ-1 protein which is unstable and does not function properly. Other mutations delete a large portion of the PARK7 gene which prevents the production of any functional DJ-1 protein.
What does DJ-1 protein do?
DJ-1 is present in almost all cells, including those of the brain. And it is a busy protein, involved in many different roles required for normal biological activity. As I mentioned above, one of the primary functions of DJ-1 is to help protect cells from oxidative stress.
What is oxidative stress?
Oxidative stress is the damage done to a cell by the process of oxidation.
And yes, I know what you are going to ask next: What is oxidation?
Oxidation is the loss of electrons from a molecule, which in turn destabilises that particular molecule. Think of iron rusting. Rust is the oxidation of iron – in the presence of oxygen and water, iron molecules will lose electrons over time. Given enough time, this results in the complete break down of objects made of iron.

Rusting iron. Source: Thoughtco
The exact same thing happens in biology. Molecules in your body go through a similar process of oxidation – losing electrons and becoming unstable. This chemical reaction leads to the production of what we call free radicals, which can then go on to damage cells.
What is a free radical?
A free radical is an unstable molecule – unstable because they are missing electrons. They react quickly with other molecules, trying to capture the needed electron to re-gain stability. Free radicals will literally attack the nearest stable molecule, stealing an electron. This leads to the “attacked” molecule becoming a free radical itself, and thus a chain reaction is started. Inside a living cell this can cause terrible damage, ultimately killing the cell.
Antioxidants are the good guys in this situation. They are molecules that neutralize free radicals by donating one of their own electrons. The antioxidant don’t become free radicals by donating an electron because by their very nature they are stable with or without that extra electron.

How free radicals and antioxidants work. Source: h2miraclewater
One of the ways in which DJ-1 responds to oxidative stress is by interacting with different proteins. One of the proteins that DJ-1 has a particular fancy for is called Kelch-like ECH-associated protein 1 (or Keap1). Under normal conditions, Keap1 acts as an anchor to another protein called nuclear factor erythroid-2-related factor 2 (Nrf2). Nrf2 is a major activator of antioxidant factors and I have discussed its activities in a previous post (Click here to read that post).
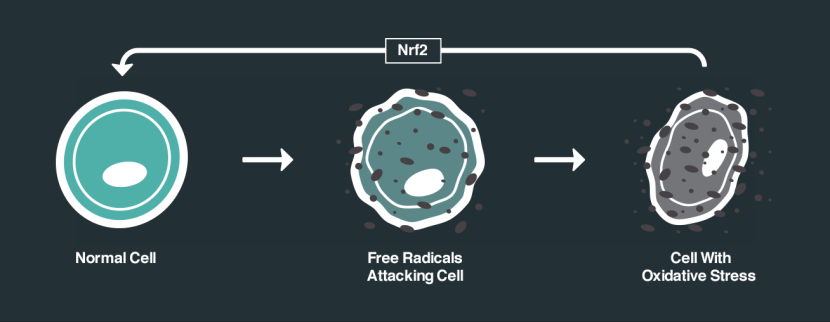
How Nrf2 works. Source: NRF2 science
So without any stress on a cell, Nrf2 is held in place by Keap1. But if oxidative stress suddenly appears, DJ-1 is activated and it grabs Keap1. This results in Nrf2 becoming free to do what it wants, and when Nrf2 is free there is only one thing it wants to do: head for the nucleus of the cell.
In the nucleus, Nrf2 activates various genes that have antioxidate properties, and by turning on these genes Nrf2 helps to decrease the level of oxidative stress. This antioxidant feature of Nrf2 has a lot of researchers excited and focused on finding drugs that help activate Nrf2.
What else does DJ-1 do?
DJ-1 is also involved in the production of the chemical dopamine. It grabs proteins that block the production of other proteins involved in making dopamine, and when DJ-1 is not functioning properly this means that less dopamine is produced (Click here and here to read more about this).
DJ-1 protein may helps fold newly produced proteins into their proper 3-dimensional shape and also helps re-fold damaged proteins. DJ-1 is known to prevent the clustering (or aggregation) of Parkinson’s disease associated protein alpha synuclein. Some studies have shown that when the PARK7 gene is mutated, the loss of DJ-1protein function results in a toxic buildup of misfolded or damaged proteins, eventually leading to cell death.
So as you can see, DJ-1 is a busy little protein with lots of jobs. For a very good review of DJ-1 in Parkinson’s disease – Click here.
So what was the new research about DJ-1 published this week?
This week, this research report was published:

Title: Isocitrate protects DJ-1 null dopaminergic cells from oxidative stress through NADP+-dependent isocitrate dehydrogenase (IDH)
Authors: Yang J, Kim MJ, Yoon W, Kim EY, Kim H, Lee Y, Min B, Kang KS, Son JH, Park HT, Chung J, Koh H.
Journal: PLoS Genet. 2017 Aug 21;13(8):e1006975.
PMID: 28827794 (This article is OPEN ACCESS if you would like to read it)
In this study the researchers conducted a screening study that was looking for activators of antioxidant activity which are themselves activated by DJ-1. They did this experiment in two types of flies: one normal group of flies and a second group in which the DJ-1 gene had been removed. They then exposed the flies to a toxin that causes oxidative stress (Rotenone) and compared the biological reaction. In the normal flies, the researchers found an increase in levels of a protein called isocitrate dehydrogenase, which was reduced in the DJ-1 flies.

Flies. Source: TheConservation
Isocitrate dehydrogenase is an enzyme that converts a chemical called isocitrate into α-ketoglutarate, in a process that also produces Nicotinamide Adenine Dinucleotide Phosphate (or NADPH), which acts as an antioxidant and reduces the damaging effects of oxidative stress.
When the investigators mutated the isocitrate dehydrogenase in flies, they found that the affected flies have age-dependent mitochondrial defects and do not live as long as their normal control counterparts. These flies are also more susceptible to the oxidative stress caused by the neurotoxin Rotenone.
Interestingly, when the researchers encourage the over production of the enzyme isocitrate dehydrogenase in DJ-1 mutant flies, they find that the flies are less susceptible to the oxidative stress caused by Rotenone and they do not lose as many dopamine neurons in their brain. PINK1 is another gene in which mutations result in an increased risk of Parkinson’s disease (Click here to read more about this). But when the investigators attempted the over production of the isocitrate dehydrogenase experiment in PINK1 mutant flies, they observed no correction of Parkinson’s-like features associated with PINK1 mutation. This result suggested to the scientists that their isocitrate dehydrogenase result was specific to the DJ-1 form of Parkinson’s disease.
Next, the researchers turned their attention aware from flies and towards mammals. They conducted experiments on mouse dopamine cells that had the DJ-1 gene mutated, and they found that isocitrate dehydrogenase had the same effect in mouse cells as it did in flies (rescuing the cells from oxidative stress). To further investigate the antioxidant effect of isocitrate dehydrogenase, they looked for a compound that would increase the levels of isocitrate dehydrogenase. They did not find one (but they give very little detail as to how far and wide they searched). So the investigators simply treated the cells with isocitrate (with a slight tweak that they called TIC – the change simply helped cells absorb isocitrate more easily), and they found the same results as they observed in the previous experiments with isocitrate dehydrogenase. This result suggests that simply increasing levels of isocitrate in DJ-1 mutant cells could rescue the cells from the harmful effects of oxidative stress.
Source: PLOS
It’s a really interesting result, and especially encouraging for people affected by Parkinson’s disease with a DJ-1 mutation.
Interesting. Has anyone ever seen anything like these results associated with isocitrate dehydrogenase before?
Actually isocitrate dehydrogenase has popped up in the past. It turned up in a yeast screening experiment for molecules that suppress the toxic effects of Parkinson’s-associated protein alpha synuclein:
Title: Novel suppressors of alpha-synuclein toxicity identified using yeast.
Authors: Liang J, Clark-Dixon C, Wang S, Flower TR, Williams-Hart T, Zweig R, Robinson LC, Tatchell K, Witt SN.
Journal: Hum Mol Genet. 2008 Dec 1;17(23):3784-95.
PMID: 18772193 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers found that isocitrate dehydrogenase (and 4 other proteins) robustly reduced the toxic effects of high levels of alpha synuclein. Interestingly, isocitrate dehydrogenase did not protect the cells from mutant versions of alpha synuclein protein – further reducing the potential utility of any future therapy targeting the isocitrate dehydrogenase pathway to just people with DJ-1 associated Parkinson’s disease.
What does it all mean?
Researchers have discovered a treatment effect that is specific to a very particular form of Parkinson’s disease (DJ-1 associated mutations). The hope is to now find molecules that can cross the blood-brain-barrier and increase levels of isocitrate dehydrogenase in the brain, rescuing cells from the damaging effects of having a reduced anti-oxidant defence system. In their research report, the investigators claimed that they had looked for such a molecule, but that they had failed to find any. They provided no details on the extent of their search and one is left wondering if any ‘stones were left unturned’. Perhaps an exhaustive screening study could bring to light some molecules that could aid us in treating this particular form of Parkinson’s disease.
The researchers also found that isocitrate treatment alone could rescue cells from the effects of oxidative stress. Isocitrate is a form of isocitric acid, and while orange juice contains about 100mg/L of isocitric acid it is too soon to know whether simply increasing levels of isocitrate in the body can have any beneficial effects for people with DJ-1 mutations. The current study only looked at flies and cells in culture. Follow up studies will be required to see if the results can be replicated in mammals before we start thinking about ourselves.
The current result is very exciting though because it shows how specific we are becoming with regards to targeting and (experimentally at least) treating particular types of Parkinson’s disease. There are sure to be more post of this nature in future.
EDITORIAL NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Youtube






3 thoughts on “Hey DJ, I-so-sit-rate!”